Data from the Australian Institute of Health and Welfare show that there have been marked increases in the prevalence of overweight and obesity in Australia since 1980.1 The 1999–2000 Australian Diabetes, Obesity and Lifestyle Study (AusDiab) showed that 48.2% of men and 29.9% of women were overweight, and 19.3% of men and 22.2% of women were obese.2 Such studies have been conducted primarily in urban areas of Australia and there is little comparable data from rural areas.
Obesity is an important modifiable risk factor for many chronic diseases, including cardiovascular disease (CVD), type 2 diabetes, hypertension, hypercholesterolaemia, certain types of cancer, osteoarthritis, gallbladder disease and mental health problems.3 The traditional CVD risk factors of smoking, hypertension and hypercholesterolaemia have been the main focus of prevention and treatment programs for several decades, with only limited attention given to obesity.4 Recent guidelines show increasing recognition that global risk — integrating a person’s individual risk factors with age, sex and any vascular disease already present — is central to risk factor assessment and management.5
In recent years, a clustering of risk factors (hyperglycaemia, hypertension, hypertriglyceridaemia, low levels of high-density lipoprotein [HDL] cholesterol, and overweight/obesity) identified as “metabolic syndrome” (MetS) has gained widespread recognition.6-9 MetS is strongly associated with an increased risk of type 2 diabetes and CVD.10 The AusDiab study11,12 showed that the prevalence of MetS among adults aged 25 and over in Australia was 19.3% according to US National Cholesterol Education Program Adult Treatment Panel III (NCEP ATP III) criteria7 and 29.1% according to International Diabetes Federation (IDF) criteria.9 No specific data on the prevalence of MetS are available for rural Australia.
Two cross-sectional population surveys of chronic disease risk factors and related health behaviour were carried out in the Greater Green Triangle region of south-eastern Australia (Box 1).13 The first survey was conducted in August–October 2004 in the Limestone Coast (LC) region of south-eastern South Australia (including the towns of Mount Gambier, Penola, Millicent, Kingston SE, Naracoorte, Lucindale, Bordertown, Keith and Robe). The second survey was conducted in February–March 2005 in Corangamite Shire (CO) in south-western Victoria (including the towns of Camperdown, Cobden, Terang, Timboon and Lismore). These two regions are predominantly rural dairy and sheep farming areas with a mainly Caucasian population. According to the Rural, Remote and Metropolitan Areas Classification,14 all survey sites except Mount Gambier were categorised as “other rural areas” (having < 10 000 inhabitants).
Our survey methods closely followed those of the World Health Organization MONICA protocol15 and the more recent European Health Risk Monitoring Project recommendations.16 Data were collected by a self-administered questionnaire, physical measurements and laboratory tests.
In the health check, weight was measured by a beam balance scale and height by a stadiometer attached to a wall. Body mass index (BMI) was computed as weight (kg) divided by height squared (m2). Participants were classified by BMI into one of four groups: underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5–24.9 kg/m2), overweight (BMI 25.0–29.9 kg/m2) or obese (BMI > 30 kg/m2).3 Waist circumference was measured at a level midway between the lower rib margin and iliac crest with the tape all around the body in a horizontal position. The mean of two measurements was calculated. Men were classified as overweight if they had a waist circumference of 94.0–101.9 cm and obese if the measurement was ≥ 102.0 cm.3 The corresponding values for women were 80.0–87.9 cm and ≥ 88.0 cm, respectively.
Two definitions of MetS were used:
1. The most recent NCEP ATP III definition.8 Three or more of the following are present:
fasting plasma glucose level ≥ 5.6 mmol/L or subject taking medication for high blood glucose level;
systolic blood pressure ≥ 130 mmHg or diastolic blood pressure ≥ 85 mmHg or subject taking antihypertensive medication;
triglyceride level ≥ 1.7 mmol/L;
HDL cholesterol level < 1.03 mmol/L (men) or < 1.30 mmol/L (women);
waist circumference ≥ 102 cm (men) or ≥ 88 cm (women).
2. IDF definition.9 The subject has central (abdominal) obesity with waist circumference ≥ 94 cm (men) or ≥ 80 cm (women) (Caucasian criteria), plus two or more of the following:
fasting plasma glucose level ≥ 5.6 mmol/L or previously diagnosed type 2 diabetes;
systolic blood pressure ≥ 130 mmHg or diastolic blood pressure ≥ 85 mmHg or subject taking antihypertensive medication;
triglyceride level ≥ 1.7 mmol/L;
HDL cholesterol level < 1.03 mmol/L (men) or < 1.29 mmol/L (women).
We analysed the results on 806 participants for whom information on obesity and MetS was available. Statistical analyses were undertaken using SPSS software, version 14.0 (SPSS Inc, Chicago, Ill, USA). We calculated age- and sex-specific prevalences and overall prevalences. Overall prevalence rates were age-standardised according to the local populations and to the 2001 Australian population aged 25–74 years,17 as appropriate.
The background characteristics of the study population are shown in Box 2.
Overall, 24.8% (95% CI, 20.5%–29.1%) of men and 35.1% (95% CI, 30.5%–39.7%) of women were within normal BMI range. The overall prevalence of overweight and obesity combined (BMI ≥ 25.0 kg/m2) was 74.1% (95% CI, 69.7%–78.5%) in men and 64.1% (95% CI, 59.5%–68.7%) in women (Box 3, Box 4). Almost a third of participants were obese, but only 1% were underweight.
Across the age groups, about half of men were defined as overweight according to BMI (Box 4). Among women, the prevalence of overweight varied between 21.6% (95% CI, 13.5%–29.8%) in the 65–74-year age group and 42.6% (95% CI, 33.8%–51.4%) in the 55–64-year age group. The prevalence of obesity increased with age, from 21.1% (95% CI, 11.6%–30.6%) in the youngest age group to 35.0% (95% CI, 26.4%–43.7%) in the oldest age group in men, and from 25.9% (95% CI, 16.4%–35.5%) to 45.4% (95% CI, 35.5%–55.3%) in women.
The prevalence of overweight and obesity combined, defined by waist circumference (men ≥ 94 cm, women ≥ 80 cm [IDF criteria9]), was higher in women (72.4% [95% CI, 68.1%–76.7%]) than in men (61.9% [95% CI, 57.0%–66.8%]) and higher in CO residents (73.0% [95% CI, 68.5%–77.5%]) than in LC residents (64.1% [95% CI, 59.5%–68.6%]) (Box 3, Box 4). The overall prevalence of central obesity (by NCEP ATP III criteria8) was 44.7% (95% CI, 41.2%–48.1%). More than half the women and almost 40% of the men were centrally obese.
The prevalence of central obesity, whether defined by NCEP ATP III or IDF criteria, tended to increase with age in both sexes (Box 4).
By NCEP ATP III criteria,8 the prevalence of MetS was 27.1% (95% CI, 22.7%–31.6%) in men and 28.3% (95% CI, 24.0%–32.6%) in women (Box 4). By IDF criteria,9 the prevalence was 33.7% (95% CI, 29.0%–38.5%) in men and 30.1% (95% CI, 25.7%–34.5%) in women. Ninety-seven per cent of participants who had MetS by NCEP ATP III criteria were also classified as having MetS by IDF criteria, indicating a high degree of agreement between the two assessment methods.
Most of the components of MetS (central obesity, hyperglycaemia, hypertension and hypertriglyceridaemia) increased with age (Box 4), but trends in HDL cholesterol levels were less clear.
To our knowledge, our survey is the most recent risk-factor prevalence study to be conducted in rural Australia and the first to report on the prevalence of MetS and its components in rural areas. We collected objective data and used random sampling with a well defined methodology. Although the participation rate was relatively low, a comparison of the socioeconomic background — including primary occupation, rate of unemployment and total gross income of the survey participants — with population statistics available17 indicated that the participants closely represented the true populations of the areas surveyed.13,17
Of particular concern was the high rate of overweight and obesity revealed by our study (64.1% in women and 74.1% in men). These figures are substantially higher than in the 1999–2000 AusDiab survey,2,12 which showed 52% of women and 68% of men were overweight or obese. Similarly, our findings on the prevalence of central obesity (61.9% in men and 72.4% in women) are worse than those reported in the AusDiab study (55% in men and 57% in women).2,12 It appears that the prevalence of overweight and obesity, as defined by BMI, is greater in men than women, whereas the reverse is observed if a waist circumference definition is used.
In the Crossroads Undiagnosed Disease Study conducted in rural Victoria between 2001 and 2003,18 the overall prevalence of obesity (defined by BMI) ranged from 25.5% in regional centres to 30.8% in smaller towns. Using waist circumference as the criterion, obesity ranged from 46.4% in regional centres to 49.8% in smaller towns. These figures were higher than in the predominantly urban 1999–2000 AusDiab study population. It is difficult to determine whether the alarming rates of obesity found in rural survey areas are just a continuation of a national trend or highlight a specific problem in rural areas.
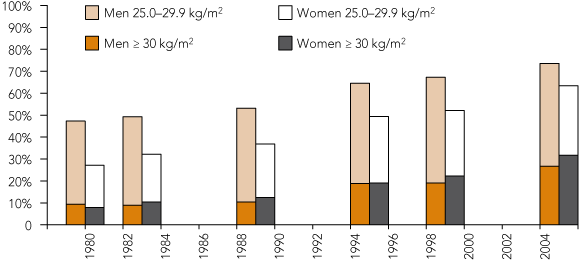
Australian trends in BMI derived from six studies done since 1980 are summarised in Box 5. Despite the limitations in comparing the different studies, there are some clear trends. In 1980, 59.2% of the adult population was of normal weight,1 compared with only 30.6% in our 2004–2005 study. The prevalence of normal weight has consistently fallen, while the prevalence of obesity has increased dramatically: the obesity prevalence of 29.3% in our survey (standardised to the 2001 Australian standard population17) was more than three times higher than in 1980.1 Although direct comparison is limited by differences in study populations and methodology, the data available show that between the 1989 National Heart Foundation of Australia (NHFA) Risk Factor Prevalence Study19 and our survey, mean height had not increased in either sex but mean weight had increased by an average of 10 kg for both sexes. Waist circumference had also increased markedly, by an average of 16 cm in women and 12 cm in men, and hip circumference had increased by 8 cm in women and 3 cm in men. This increase in waist circumference is even greater than that reported in the 1999–2000 AusDiab study,2,12 which showed an increase of 7 cm in men and 8 cm in women compared with 1989. These figures are all consistent with an increase in central obesity that has been strongly linked to the development of diabetes20 and myocardial infarction.21
Apart from AusDiab study data,11,12 there is limited information on the prevalence of MetS and its components in Australia. By NCEP ATP III criteria,7 the prevalence of MetS was 19.3% in the AusDiab study (compared with 28.6% in our study). However, we used the updated 2005 NCEP ATP III8 criteria, which incorporated a lower plasma glucose cut-off level. Using IDF criteria,9 which have lower central obesity cut-off points, the prevalence of MetS was 29.1% in the AusDiab study2,12 and 33.3% in our study. Common to both studies was an increasing prevalence of MetS and its components with age and for both sexes. The high prevalence of overweight and obesity in the areas we surveyed translates into a high prevalence of MetS and its components. As the population ages, the number of people with MetS will increase further.
The use of different definitions of MetS6-9 makes international comparisons difficult. In the 1992 FINRISK cohort,22 in which modified WHO criteria were used, MetS was found to be present in 38.8% of men and 22.2% of women. In a US study using NCEP ATP III criteria, MetS was present in 31% of men and 35% of women.7,23 Regardless of the definitions used, the prevalence of MetS is high and increasing worldwide.
As Box 5 illustrates, early warning signs of the overweight and obesity epidemic were apparent from the NHFA 1989 data,1 yet insufficient notice was taken at the time. Now that there is a major problem, with only 30% of the population within the “normal weight” range, urgent action is required at the highest level to change unhealthy lifestyle habits by improving diet, increasing physical activity and making our environments supportive of these objectives.
Major lessons can be learnt from previous successful initiatives in reducing cigarette smoking, particularly the need for population-based interventions and an ongoing monitoring and surveillance system to determine the impact of interventions. The approach in Finland is to conduct 5-yearly surveys using objective measurements and blood sampling.24 In contrast, current surveillance systems such as the Australian Bureau of Statistics national health surveys25 and statewide computer-assisted telephone interviews,26 which use self-reported height and weight data, consistently underestimate the true prevalence of overweight and obesity.
1 Limestone Coast and Corangamite Shire survey areas in the Greater Green Triangle region of western Victoria and south-eastern South Australia
3 Prevalence of overweight and obesity, by region and assessment method, expressed as percentage (95% CI)*
Overweight and obese combined (≥ 94 cm [men], ≥ 80 cm [women]) |
|||||||||||||||
4 Prevalence of overweight, obesity, metabolic syndrome (MetS) and the components of MetS, by sex and age, expressed as percentage (95% CI)*
5 The prevalence of overweight and obesity in various Australian studies conducted since 1980*†
 | |||||||||||||||
|
* Results are age-standardised to the 2001 Australian standard population.17 † Sources: Australian Institute of Health and Welfare analysis of the 1980, 1983 and 1989 Risk Factor Prevalence Surveys;1,17 1995 National Nutrition Survey;1 1999–2000 Australian Diabetes, Obesity and Lifestyle Study (AusDiab);2,12 2004–2005 Limestone Coast and Corangamite Shire risk factor prevalence surveys.13 | |||||||||||||||
Received 5 March 2007, accepted 29 May 2007
- Edward D Janus1
- Tiina Laatikainen2
- James A Dunbar3
- Annamari Kilkkinen3
- Stephen J Bunker4
- Benjamin Philpot3
- Philip A Tideman5,6
- Rosy Tirimacco6
- Sami Heistaro2
- 1 Department of Medicine, Western Health, Melbourne, VIC.
- 2 National Public Health Institute, Helsinki, Finland.
- 3 Greater Green Triangle University Department of Rural Health, Flinders University and Deakin University, Warrnambool, VIC.
- 4 Department of Epidemiology and Preventive Medicine, Monash University, Melbourne, VIC.
- 5 Cardiovascular Medicine, Flinders Medical Centre, Adelaide, SA.
- 6 Integrated Cardiovascular Clinical Network SA, Flinders Medical Centre, Adelaide, SA.
We thank Ms Anna Chapman, Dr Andrew Baird, the nurses carrying out the survey and regional hospitals providing facilities for our study. The study was supported by the Australian Government Department of Health and Ageing, the Royal Australian College of General Practitioners, Sanofi-Aventis, Pfizer and Roche Diagnostics.
Within the past 2 years, Edward Janus has received travel support from AstraZeneca and Pfizer, and honoraria and speaker fees from AstraZeneca, Sanofi-Aventis, Roche, Fournier, Servier and Pfizer. However, no pharmaceutical products were used in our study.
- 1. Australian Institute of Health and Welfare. Risk factor prevalence data. http://www.aihw.gov. au/dataonline/riskfactors/index.cfm (accessed Feb 2007).
- 2. Cameron AJ, Welborn TA, Zimmet PZ, et al. Overweight and obesity in Australia: the 1999–2000 Australian Diabetes, Obesity and Lifestyle Study (AusDiab). Med J Aust 2003; 178: 427-432. <MJA full text>
- 3. Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 2000; 894: i-xii, 1-253.
- 4. Bennett SA, Magnus P. Trends in cardiovascular risk factors in Australia. Results from the National Heart Foundation’s Risk Factor Prevalence Study, 1980–1989. Med J Aust 1994; 161: 519-527.
- 5. National Heart Foundation of Australia; Cardiac Society of Australia and New Zealand. Reducing risk in heart disease 2004. http://www.heartfoundation.com.au/downloads/RRIHD_fullguide_update_010405.pdf (accessed Feb 2007).
- 6. Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 1998; 15: 539-553.
- 7. National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002; 106: 3143-3421.
- 8. Grundy SM, Cleeman JI, Daniels SR, et al; American Heart Association; National Heart, Lung, and Blood Institute. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Circulation 2005; 112: 2735-2752.
- 9. Alberti KG, Zimmet PZ, Shaw J. Metabolic syndrome — a new world-wide definition. A consensus statement from the International Diabetes Federation. Diabet Med 2006; 23: 469-480.
- 10. Galassi A, Reynolds K, He J. Metabolic syndrome and risk of cardiovascular disease: a meta-analysis. Am J Med 2006; 119: 812-819.
- 11. Zimmet PZ, Alberti KG, Shaw JE. Mainstreaming the metabolic syndrome: a definitive definition. Med J Aust 2005; 183: 175-176. <MJA full text>
- 12. Barr ELM, Magliano DJ, Zimmet PZ, et al. AusDiab 2005. The Australian Diabetes, Obesity and Lifestyle Study. Melbourne: International Diabetes Institute, 2006. http://www.diabetes.com.au/pdf/AUSDIAB_Report_Final.pdf (accessed Jun 2007).
- 13. Heistaro S, Janus E, Dunbar J, et al, editors. Greater Green Triangle Risk Factor Study. Limestone Coast and Corangamite Shire surveys. Basic report. http://www.greaterhealth.org/media/resources/6b2c362f02722c632 baecde2d3601f46.pdf (accessed Feb 2007).
- 14. Department of Primary Industries and Energy; Department of Human Services and Health. Rural, Remote and Metropolitan Areas Classification. 1991 census edition. Canberra: AGPS, 1994. http://www.pc.gov.au/inquiry/nursehom/subs/subdr096.pdf (accessed Jun 2007).
- 15. The World Health Organization MONICA Project (monitoring trends and determinants in cardiovascular disease): a major international collaboration. WHO MONICA Project Principal Investigators. J Clin Epidemiol 1988; 41: 105-114.
- 16. Tolonen H, Kuulasmaa K, Laatikainen T, Wolf H; European Health Risk Monitoring Project. Recommendation for indicators, international collaboration, protocol and manual of operations for chronic disease risk factor surveys. Helsinki: EHRM, 2002. http://www.ktl.fi/publications/ehrm/product2/product2.pdf (accessed Jun 2007).
- 17. Australian Bureau of Statistics. Census of population and housing: basic community profiles, 2001. Canberra: ABS, 2001.
- 18. Simmons D, McKenzie A, Eaton S, et al. Choice and availability of take away and restaurant food is not related to the prevalence of adult obesity in rural communities in Australia. Int J Obes (Lond) 2005; 29: 703-710.
- 19. Risk Factor Prevalence Study Management Committee. Risk factor prevalence study survey. No. 3, 1989. Canberra: National Heart Foundation of Australia and Australian Institute of Health, 1990.
- 20. Wat NM, Lam TH, Janus ED, Lam KS. Central obesity predicts the worsening of glycaemia in southern Chinese. Int J Obes Relat Metab Disord 2001; 25: 1789-1793.
- 21. Yusuf S, Hawken S Ounpuu S, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART Study): case–control study. Lancet 2004; 364: 937-952.
- 22. Ilanne-Parikka P, Eriksson JG, Lindstrom J, et al. Prevalence of the metabolic syndrome and its components: findings from a Finnish general population sample and the Diabetes Prevention Study cohort. Diabetes Care 2004; 27: 2135-2140.
- 23. Patel A, Huang KC, Janus ED, et al. Is a single definition of the metabolic syndrome appropriate? A comparative study of the USA and Asia. Atherosclerosis 2006; 184: 225-232.
- 24. National Public Health Institute in Finland. The National FINRISK Study. http://www.ktl.fi/portal/english/research__people___programs/health_promotion_and_chronic_disease_prevention/ units/chronic_disease_epidemiology_unit/the_national_finrisk_study (accessed Feb 2007).
- 25. Australian Bureau of Statistics. National Health Survey: summary of results, 2004–05. Canberra: ABS, 2006. (ABS Cat. No. 4364.0.) http://www.ausstats.abs.gov.au/ausstats/subscriber.nsf/0/3B1917236618A042CA25711F00 185526/$File/43640_2004-05.pdf (accessed Feb 2007).
- 26. Victorian Government Department of Human Services. Victorian population health survey. http://www.health.vic.gov.au/healthstatus/vphs.htm (accessed Feb 2007).





Abstract
Objective: To measure the prevalence of overweight, obesity and the metabolic syndrome (MetS) in rural Australia.
Design, setting and participants: Cross-sectional surveys were conducted in two rural areas in Victoria and South Australia in 2004–2005. A stratified random sample of men and women aged 25–74 years was selected from the electoral roll. Data were collected by a self-administered questionnaire, physical measurements and laboratory tests.
Main outcome measures: Prevalence of overweight and obesity, as defined by body mass index (BMI) and waist circumference; prevalence of MetS and its components.
Results: Data on 806 participants (383 men and 423 women) were analysed. Based on BMI, the prevalence of overweight and obesity combined was 74.1% (95% CI, 69.7%–78.5%) in men and 64.1% (95% CI, 59.5%–68.7%) in women. Based on waist circumference, the prevalence of overweight and obesity was higher in women (72.4%; 95% CI, 68.1%–76.7%) than men (61.9%; 95% CI, 57.0%–66.8%). The overall prevalence of obesity was 30.0% (95% CI, 26.8%–33.2%) based on BMI (≥ 30.0 kg/m2) and 44.7% (95% CI, 41.2%–48.1%) based on waist circumference (≥ 102 cm [men] and ≥ 88 cm [women]). The prevalence of MetS as defined by the US National Cholesterol Education Program Adult Treatment Panel III 2005 criteria was 27.1% (95% CI, 22.7%–31.6%) in men and 28.3% (95% CI, 24.0%–32.6%) in women; based on International Diabetes Federation criteria, prevalences for men and women were 33.7% (95% CI, 29.0%–38.5%) and 30.1% (95% CI, 25.7%–34.5%), respectively. Prevalences of MetS, central (abdominal) obesity, hyperglycaemia, hypertension and hypertriglyceridaemia increased with age.
Conclusions: In rural Australia, prevalences of MetS, overweight and obesity are very high. Urgent population-wide action is required to tackle the problem.