More than one in eight women will develop breast cancer in their lifetime,1 and it is the most common cause of cancer mortality in Australian women.2 Mammography remains the mainstay of early breast cancer diagnosis, and its performance depends on image quality.3 Mammographic screening has been in use for 20–30 years,4 and reduces breast cancer mortality in women aged 40 years and over.5,6 In women aged 50–69 years, mortality decreases by 20%–35% at 14 years of follow-up; efficacy in 40–49-year-old women is slightly lower.7
A technical limitation of conventional screen-film mammography (CM) occurs when there is insufficient contrast in radiographic density between normal breast parenchyma and cancer. About 10%–20% of palpable breast cancers are not visible on CM for this reason.8 As women age, less dense breast fat replaces normally dense breast parenchyma, so density contrast limitations have a greater impact on younger women.
Digital mammography (DM) has demonstrated superior diagnostic accuracy over CM for cancer detection in women with dense breasts and women under 50 years old, and evidence exists for its superior demonstration of microcalcifications.9-11 About 8% of facilities in the United States have now converted from CM to DM,12 and the number is growing.
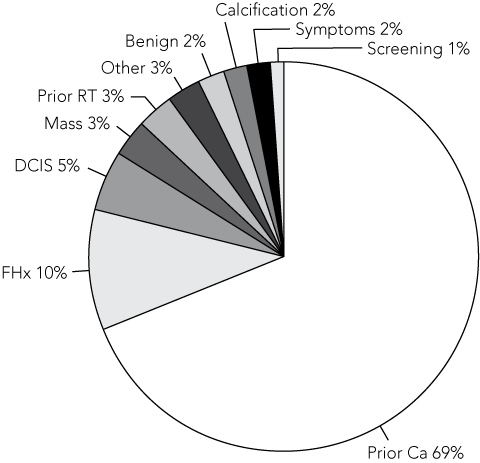
In the first year of DM operation (April 2004 – April 2005), 1208 diagnostic mammograms were performed on 1157 women. All diagnostic reading was done in “soft copy”, by viewing the mammographic images on a computer screen. The patient age range was 21–95 years (mean, 59 years; median, 59 years; interquartile range, 50–69 years). Indications for referral are shown in Box 1; 69% were for annual surveillance after successful treatment of breast cancer, and 10% were FCC referrals.
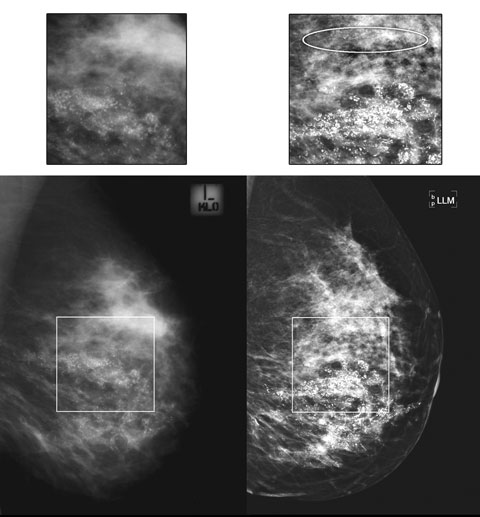
In some of our diagnostic DM studies, we found that comparison with the patient’s earlier CM imaging showed a greater number or a more extensive distribution of microcalcifications. Microcalcifications may be a manifestation of ductal carcinoma-in-situ (DCIS). They are classifiable as malignant, indeterminate or benign, depending on individual microcalcification and cluster morphology. All else being equal, progression of microcalcifications is also suspicious for malignancy. However, because DM has been shown to demonstrate microcalcifications better than CM,9-11 two explanations were possible in these women:
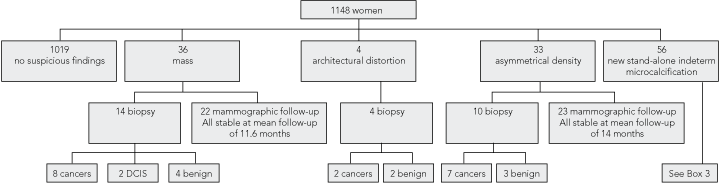
Of the 1157 women who underwent DM in our first year of operation, nine had breast cancer histologically proven before referral. Of the remaining 1148 women, 1019 had no suspicious findings on DM. Of the 129 women with positive findings, 17 new cases of invasive cancer (including three in women younger than 50 years) and six new cases of DCIS (including one found on 12-month review) were detected (Box 2).
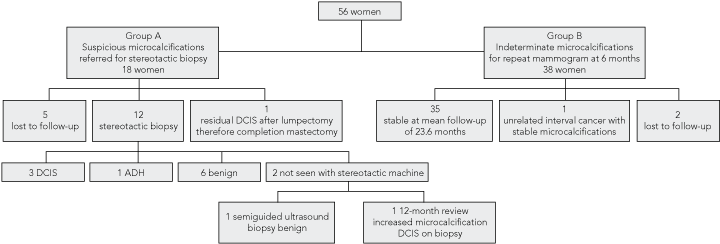
Microcalcifications adjacent to another finding were classified with that finding. New indeterminate or suspicious stand-alone microcalcifications were identified in 56 women: 18 in Group A (stereotactic biopsy) and 38 in Group B (6-month repeat mammogram). Box 3 shows the outcomes of the two groups to date. Significantly, 35 women (92%) in Group B demonstrated microcalcification stability and no adverse outcomes over a mean follow-up period of 23.6 months (range, 6–32 months). In Group A, there were only four cases of DCIS (including the case found at 12-month review) and one case of atypical ductal hyperplasia (considered premalignant).
In adopting DM, we took technical, practical and other considerations into account.
DM offers several technical advances over CM. In CM, the breast image is created directly on mammographic x-ray film, which acts as the sole vehicle for the three imaging steps: acquisition, display and storage. Overall image quality is a compromise between these steps. Once acquired, the image cannot be altered. Traditional methods of improving a reader’s viewing performance are a very dark room, a handheld loupe and a bright light.14 If greater magnification is required, the patient has to return for focal compression magnification views (with further radiation exposure).
In DM, the steps of image acquisition, display and storage are separated, allowing optimisation of every step.15 DM images are acquired in the same way as CM, but a digital detector replaces the x-ray film cassette. Unlike CM, DM allows manipulation of the stored image. The reader can adjust contrast and intensity, and magnify and move the image, all without acquiring further views (Box 4). Since a digital mammogram is a data file, it can be copied, sent via a network, burned to CD or printed on a mammographic-quality x-ray laser printer. Multiple copies can be reviewed simultaneously at different sites. The capacity for the technologist to immediately view the acquired images for quality assurance and to transmit the data files to a central reading site could make DM an excellent choice for mobile BreastScreen units operating in rural and remote areas.
It is our experience that technologist examination time is reduced with DM compared with CM, mostly due to immediate image availability. In our subjective experience, there has also been no impact on the time taken by the radiologist to review DM images, compared with CM. It has been reported that DM reading takes longer than CM reading in screening examinations.13 However, our experience is in a diagnostic workup setting, and so reflects a different workflow to screening.
All the DM-assisted HNL procedures were completed successfully. The mean time with breast in compression for 21 consecutive cases was 28.5 minutes. Our subjective experience suggests that time required for HNL procedures is less with DM; others have reported similar results.16
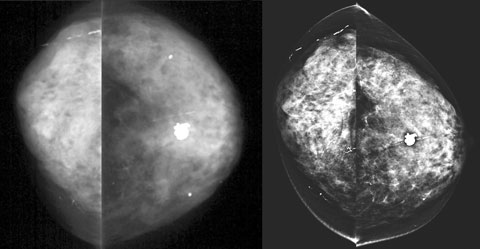
The sensitivity of DM is at least equal to CM for detection of breast cancer.17-21 A recent study of over 49 000 women showed DM to be more diagnostically accurate than CM in women under 50 years of age, women with radiographically dense breasts and pre- or peri-menopausal women.21 Underlying this clinical finding is the proven superior contrast resolution of DM, giving it maximal impact in radiographically dense breast tissue that may mask a dense cancerous mass.22 In our patient cohort, we subjectively found that imaging of dense breasts improved with DM compared with CM (Box 5). With growing debate about whether to invite women aged 40–49 years to screening by BreastScreen, the greater accuracy of DM in dense breasts is particularly pertinent.23
Further, evidence is mounting that DM may provide superior image quality at an equal or lower breast glandular dose than CM.8,21 This matters particularly for women at high genetic risk of breast cancer, who may undergo frequent mammographic screening from a relatively young age.
Evidence is also accumulating in radiological literature that DM is superior to CM for demonstration of microcalcifications.9-11 Box 6 demonstrates the example of a woman referred for HNL and wide local excision of DCIS, where DM showed a more extensive distribution of malignant calcifications, thus necessitating a change in management to mastectomy.
1 Main indications for digital mammography referral in the first year of operation at the Peter MacCallum Cancer Centre
 | |||||||||||||||
2 Diagnostic findings and outcomes for 1148 women undergoing diagnostic digital mammography studies
 | |||||||||||||||
|
DCIS = ductal carcinoma-in-situ. Indeterm = indeterminate or suspicious. | |||||||||||||||
3 Management pathways and outcomes for 56 women found to have new indeterminate or suspicious stand-alone microcalcifications
 | |||||||||||||||
|
DCIS = ductal carcinoma-in-situ. ADH = atypical ductal hyperplasia. | |||||||||||||||
- Emma Pun1
- W F Eddie Lau1
- Robin Cassumbhoy1
- Anthony J Taranto1
- Alexander G Pitman2
- 1 Department of Diagnostic Imaging, Peter MacCallum Cancer Centre, Melbourne, VIC.
- 2 Department of Medical Imaging, St Vincent’s Hospital, Melbourne, VIC.
We are grateful to BreastScreen Victoria, St Vincent’s Health (Associate Professor Jennifer Cawson, Chief Radiologist) for providing backup mammographic service during Peter Mac’s changeover from CM to DM. We acknowledge Associate Professor Cawson’s help and support as our external reference breast radiologist during the subsequent setup and validation of our DM service.
None identified.
- 1. Feuer EJ, Wun LM, Boring CC, et al. The lifetime risk of developing breast cancer. J Natl Cancer Inst 1993; 85: 892-897.
- 2. Australian Institute of Health and Welfare. BreastScreen Australia monitoring report 2002–2003. Canberra: AIHW, 2006. (Cancer Series No. 32. AIHW Cat. No. CAN 27.)
- 3. Pisano ED, Gatsonis CA, Yaffe MJ, et al. American College of Radiology Imaging Network Digital Mammographic Imaging Screening Trial: objectives and methodology. Radiology 2005; 236: 404-412.
- 4. Pisano ED. Current status of full-field digital mammography. Radiology 2000; 214: 26-28.
- 5. Humphrey LL, Helfand M, Chan BK, Woolf SH. Breast cancer screening: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med 2002; 137: 347-360.
- 6. Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med 2005; 353: 1784-1792.
- 7. Elmore JG, Armstrong K, Lehman CD, Fletcher SW. Screening for breast cancer. JAMA 2005; 293: 1245-1256.
- 8. Mahesh M. AAPM/RSNA physics tutorial for residents. Digital mammography: an overview. Radiographics 2004; 24: 1747-1760.
- 9. Fischer U, Baum F, Obenauer S, et al. Comparative study in patients with microcalcifications: full-field digital mammography vs screen-film mammography. Eur Radiol 2002; 12: 2679-2683.
- 10. Diekmann S, Bick U, von Heyden H, Diekmann F. [Visualization of microcalcifications on mammographies obtained by digital full-field mammography in comparison to conventional film-screen mammography] [German]. Rofo 2003; 175: 775-779.
- 11. Kim HS, Han BK, Choo KS, et al. Screen-film mammography and soft-copy full-field digital mammography: comparison in the patients with microcalcifications. Korean J Radiol 2005; 6: 214-220.
- 12. Dershaw DD. Film or digital mammographic screening [editorial]? N Engl J Med 2005; 353: 1846-1847.
- 13. Berns EA, Hendrick RE, Solari M, et al. Digital and screen-film mammography: comparison of image acquisition and interpretation times. AJR Am J Roentgenol 2006; 187: 38-41.
- 14. Pisano ED, Kuzmiak C, Koomen M, Cance W. What every surgical oncologist should know about digital mammography. Semin Surg Oncol 2001; 20: 181-186.
- 15. Pisano ED, Yaffe MJ. Digital mammography. Radiology 2005; 234: 353-362.
- 16. Yang WT, Whitman GJ, Johnson MM, et al. Needle localization for excisional biopsy of breast lesions: comparison of effect of use of full-field digital versus screen-film mammographic guidance on procedure time. Radiology 2004; 231: 277-281.
- 17. Lewin JM, Hendrick RE, D’Orsi CJ, et al. Comparison of full-field digital mammography with screen-film mammography for cancer detection: results of 4,945 paired examinations. Radiology 2001; 218: 873-880.
- 18. Lewin JM, D’Orsi CJ, Hendrick RE, et al. Clinical comparison of full-field digital mammography and screen-film mammography for detection of breast cancer. AJR Am J Roentgenol 2002; 179: 671-677.
- 19. Skaane P, Young K, Skjennald A. Population-based mammography screening: comparison of screen-film and full-field digital mammography with soft-copy reading — Oslo I study. Radiology 2003; 229: 877-884.
- 20. Skaane P, Skjennald A. Screen-film mammography versus full-field digital mammography with soft copy reading: randomized trial in a population-based screening program — the Oslo II study. Radiology 2004; 232: 197-204.
- 21. Pisano ED, Gatsonis C, Hendrick E, et al. Diagnostic performance of digital versus film mammography for breast-cancer screening. N Engl J Med 2005; 353: 1773-1783.
- 22. Skaane P, Balleyguier C, Diekmann F, et al. Breast lesion detection and classification: comparison of screen-film mammography and full-field digital mammography with soft-copy reading — observer performance study. Radiology 2005; 237: 37-44.
- 23. Helme S, Perry N, Mokbel K. Screening mammography in women aged 40–49: is it time to change [editorial]? Int Semin Surg Oncol 2006; 3: 4.





Abstract
In April 2004, Melbourne’s Peter MacCallum Cancer Centre, Australia’s only stand-alone dedicated cancer hospital, became the first Australian site to offer digital mammography (DM).
In the first year of DM operation, 1208 mammograms were performed on 1157 women; 17 new cases of invasive carcinoma and six new cases of ductal carcinoma-in-situ (DCIS) were detected; and 30 hook-wire needle localisations were conducted in 29 patients.
We developed a unit policy to manage indeterminate microcalcifications newly demonstrated on DM that were not previously detected by conventional screen-film mammography (CM): those believed to have malignant morphology were recommended for biopsy, and those without were recommended for 6-month DM follow-up to confirm microcalcification stability.
DM detected 56 new stand-alone microcalcifications (18 suspicious and 38 indeterminate). Tissue diagnosis of 12 suspicious microcalcifications yielded four cases of DCIS and one of atypical ductal hyperplasia. Of the indeterminate microcalcifications, 35 have demonstrated stability at DM follow-up to date, over a mean period of 23.6 months.
From our experience, we believe DM’s superior demonstration ability uncovered microcalcifications previously undetected by CM, rather than microcalcification progression. We suggest that routine review with DM, rather than biopsy, is appropriate management when new indeterminate microcalcifications without malignant characteristics are identified by DM.