We describe a previously unreported association between hyoscine hydrobromide and severe oesophagitis, with ulceration visible almost to the submucosa in the distal oesophagus. The condition resolved with cessation of tablets and 2 months’ treatment with a proton-pump inhibitor.
These symptoms developed during a boating holiday, when he had taken hyoscine tablets (an over-the-counter preparation) for motion sickness. Over a period of 4 days, these had been chewed and swallowed twice daily without food or water. He had not ingested other medications or potentially corrosive agents.
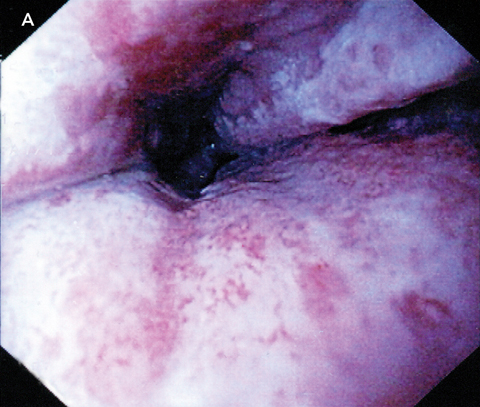
There was no other relevant medical history. The patient was a non-smoker and consumed only small amounts of alcohol. Results of a physical examination, electrocardiogram and chest x-ray were all normal. Upper gastrointestinal endoscopy revealed circumferential ulceration, with clearly visible submucosa of the distal 8 cm of the oesophagus (Box A). The stomach was normal. There was patchy erythema in the first part of the duodenum, but the second and third parts were normal. Histological examination of oesophageal biopsies demonstrated only granulation tissue. Duodenal biopsies were normal.
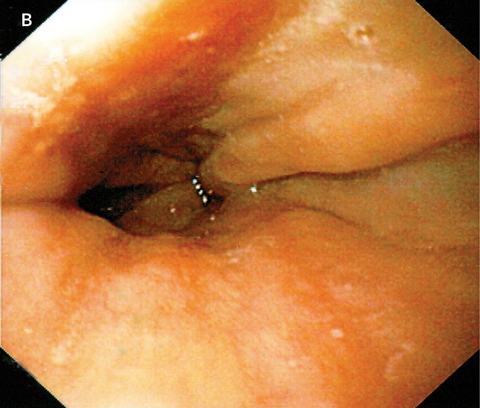
The patient was commenced on 40 mg daily of esomeprazole (a proton-pump inhibitor). Over the following week, he experienced a gradual improvement in his odynophagia and was able to return to a normal diet. Esomeprazole treatment was continued for 8 weeks and he remained asymptomatic. A repeat upper gastrointestinal endoscopy a further 2 weeks later showed complete healing of the oesophagus (Box B).
A search of the Adverse Drug Reactions Advisory Committee online database and MEDLINE database (1966 to present) uncovered no previous reports of ulcerative oesophagitis secondary to consumption of hyoscine hydrobromide. An objective causality assessment was made in accordance with the Naranjo algorithm.1 This algorithm involves summing the numeric scores for 10 standard questions to provide an overall probability index, with possible outcomes of “highly probable”, “probable”, “possible” or “doubtful”. Our score of 6 indicated that the adverse drug reaction association between hyoscine and ulcerative oesophagitis was probable.
Medication-induced oesophagitis may result from either local or systemic effects of a drug. Direct mucosal injury occurs after prolonged exposure of the oesophageal mucosa to the caustic contents of the medication.2 Examples of medications known to cause this type of injury include tetracycline, aspirin and non-steroidal anti-inflammatory drugs, as well as alendronate, potassium chloride, and iron compounds. Endoscopy often reveals a discrete ulcer with relatively normal surrounding mucosa.3 Risk factors for retention of pills in the oesophagus include a lack of adequate liquid bolus, lying down after swallowing, and older age.2,4
Although we could find no reports of hyoscine hydrobromide having a direct toxic effect on the oesophagus, the related compound hyoscine butylbromide has been shown to significantly increase the number of reflux episodes compared with placebo,5 in contrast with atropine, another anticholinergic, which has been shown to reduce reflux episodes.6 If hyoscine hydrobromide acts similarly to hyoscine butylbromide, it would be reasonable to hypothesise that the oesophagitis in our patient may be secondary to prolonged acid exposure. As the tablets were consumed without water, it is also possible that local mucosal injury may have occurred due to retention of tablets in the lower oesophagus, with subsequent pressure necrosis or caustic injury. Further studies would be required to elucidate the precise mechanism of injury.
- 1. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30: 239-245.
- 2. Carlborg B, Densert O. Esophageal lesions caused by orally administered drugs. An experimental study in the cat. Eur Surg Res 1980; 12: 270-282.
- 3. Abid S, Mumtaz K, Jafri W, et al. Pill-induced esophageal injury: endoscopic features and clinical outcomes. Endoscopy 2005; 37: 740-744.
- 4. Kikendall JW, Friedman AC, Oyewole MA, et al. Pill-induced esophageal injury. Case reports and review of the medical literature. Dig Dis Sci 1983; 28: 174-182.
- 5. Ciccaglione AF, Grossi L, Cappello G, et al. Effect of hyoscine N-butylbromide on gastroesophageal reflux in normal subjects and patients with gastroesophageal reflux disease. Am J Gastroenterol 2001; 96: 2306-2311.
- 6. Mittal RK, Holloway R, Dent J. Effect of atropine on the frequency of reflux and transient lower esophageal sphincter relaxation in normal subjects. Gastroenterology 1995; 109: 1547-1554.





Andrew Keegan is a consultant for Janssen-Cilag, which markets a competitor to esomeprazole.