Since the introduction of organised cervical screening in Australia in 1991, the National Cervical Screening Program has succeeded in reducing the incidence and mortality from invasive cervical cancer.1 The program targets sexually active women for screening at 2-yearly intervals from age 18–20 years up to 69 years. At the time the screening program was established, a major influence on policy was a 1986 review by a working group of the International Agency for Research on Cancer (IARC) on the effect of screening interval in eight countries.2 The IARC working group found that 3-yearly screening conferred a substantial level of protection against cervical cancer, but that more frequent screening afforded little further protection.
The choice of a 2-yearly screening interval in Australia was made in the context of the IARC review, while including a safety margin. This was to take into account that Pap test registers were not yet configured in all states, and that full quality assurance measures on all aspects of cervical smear testing, from the smear-taker to the laboratory, were not yet in place.3 The Australian National Cervical Screening Program, therefore, recommends cervical screening more frequently and across a wider age range than many screening programs in developed countries.3
In June 2005, the National Health and Medical Research Council (NHMRC) recommended that “the screening interval for Pap smears in Australia be reviewed to ensure that the program here is consistent with international best practice”.4 In this context, we compared policy and outcomes between Australia and the United Kingdom. As the approaches to cervical screening in the two countries have been similar in some, but not all, respects, the comparison allows the effects of key differences to be evaluated.
The primary comparison was confined to the period before 2001, partly because of data availability and partly because both programs underwent changes after this date. In 2003, the UK initiated a transition from conventional to liquid-based cytology and adopted age-dependent screening intervals. In 2004–2005, each country initiated different changes to the management of low-grade smear abnormalities.1,5
We used screening registry data to estimate the screening participation (or coverage) rate, defined as the proportion of women in the population eligible for screening who have at least one recorded cervical screening test in the time frame of interest. In Australia, the 2-yearly rate was estimated as the average national rate in women aged 20–69 years for 1997–1998 and 1999–2000,6 and the 3-yearly rate was estimated using 1998–2000 data for New South Wales (which has about a third of Australia’s population).7 In the UK, the 3-yearly rate in women aged 25–64 years was for England in 1998–2001, and the 5-yearly rate for 1996–2001.8 The lifetime rate in 2001 was estimated for Australia as the self-reported ever-screened rate in women aged 18 years or over,9 and for the UK from health authority KC53 returns for women aged 25–64 years.8 We also used registry data to assess the cumulative re-screened proportion at various times after a negative smear for women who appeared on the register.10
To compare outcomes in the two countries, changes in cervical cancer rates were examined before and after the introduction of organised screening, using data from the IARC’s Cancer incidence in five continents11 and the World Health Organization mortality database12 (for mortality rates before 2002). For additional information on incidence rates and supplementary analyses, data were obtained directly from the relevant national statistical reports.6,13,14 All analyses were performed using code C53 of the 10th revision of the International classification of diseases. Direct age-standardisation was performed using the World Standard Population.15 While reliable information on mortality is available from the 1970s, comprehensive and reliable incidence data are not available before the 1980s. Therefore, changes in incidence rates were compared only for the period after introduction of organised screening.
In both Australia and the UK, opportunistic screening was conducted from the 1960s. Organised screening was implemented in 1991 in Australia and in 1988 in the UK, at a recommended interval of 2 years for women aged from 18–20 years to 69 years in Australia, and a recommended interval of 3 or 5 years for women aged from 20 years to 64 years in the UK. Until 2003, the interval in the UK varied between 3 and 5 years for different health authorities, with more than half of those in England issuing screening invitations every 3 years.16 Even in those with a 5-yearly recall policy, a substantial proportion of general practitioners operated their own 3-yearly recall; in one such region, 62% of GPs were recalling women at 3 years, leading to the conclusion that, in England, “five-yearly ... recall systems do not have a major impact on population coverage but are acting predominantly as a failsafe mechanism”.17
Since the introduction of organised screening, both countries have implemented comprehensive laboratory quality assurance, including training, proficiency testing and performance standards for reporting.16,18 Although the system used to classify cytological abnormalities varied, the management of lesions of broadly similar severity did not differ substantially between the two countries.
It is likely that, in Australia, a higher proportion of women with low-grade abnormalities were immediately referred for colposcopy. However, relevant studies differ with respect to the follow-up period, and some report on biopsy rates rather than colposcopy referral rates, making interpretation difficult.19-22
In Australia, reminder letters to attend screening are sent at 27–36 months by the state and territory Pap test registers, which were established between 1989 (Victoria) and 1999 (Queensland). Women are entered onto the register unless they elect to opt off, with an opt-off rate in NSW of about 1%.7 A Medicare rebate is available for screening with conventional cytology. In 2001, practice incentive payments were introduced for screening eligible women who had not been re-screened for 4 years,23 and, in 2005, new incentives were introduced to allow a practice nurse to take smears in this group of women in certain regional, rural and remote areas.24
In the UK, the National Health Service (NHS) call and recall system is organised on the basis of regional health authorities (more recently restructured as Primary Care Trusts). The health authority generated invitation and reminder letters to eligible women registered with a GP in the region. The precise timing varied by region — for example, in Oxfordshire, invitation letters were issued at 34 months, and reminder letters were usually sent 6–12 months later. If a woman did not respond, follow-up was performed by the GP. To encourage high levels of 5-yearly coverage, a practice incentive scheme was introduced in 1990. This was structured so that each practice received the maximum payment if more than 80% of registered eligible women had been screened in the previous 5 years, a lower payment if 50%–79% of women had been screened, and no payment if fewer than 50% had been screened.16
Since the introduction of organised screening, the main differences in approach between the two countries lie in the specific policy recommendations for the screening interval and target age range (Box 1), and the systems for implementing these policies. Therefore, a comparative analysis of the patterns of screening uptake and cervical cancer incidence and mortality in the two countries should provide some evidence on the relative effectiveness of the different screening policies.
Box 1 shows that screening participation rates were slightly higher in Australia than in the UK over a 3-year period, but were similar over a lifetime (88% and 90%, respectively, assessed in 2001).
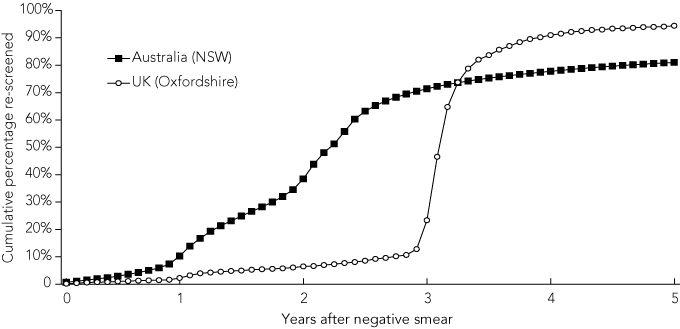
Box 2 shows the cumulative proportion re-screened by time since a negative smear for populations in Australia (NSW) and the UK (Oxfordshire). Re-screening in NSW was most likely to occur either at about 1 year after the negative index smear (mainly at 12–15 months) or after 2 years (mainly at 23–30 months), reflecting some annual screening in addition to the effective dissemination of the biennial screening message and the effect of reminder letters sent at 27 months. In NSW, the proportion of women re-screened within 21 months was 30%. This accords with the result of 32% obtained from a sample of 175 723 women from all Australian states and territories who had an index smear in February 1999.6
In the UK, most re-screening occurred at around the third year (mainly at 34–40 months) after the last negative smear. To assess the applicability of the Oxfordshire data to other regions in the UK, the re-screening patterns were compared with those obtained from a retrospective analysis of 50 915 women on 3-yearly routine recall in nine different health authority regions in the north-west of England,17 and close agreement was found. In the north-west regions, the proportions re-screened within 36 months and 60 months were 25% and 93%, compared with 23% and 94%, respectively, in Oxfordshire.
The median time to the next screening test in NSW was 27 months, compared with 38 months in Oxfordshire (Box 2). At 39 months, the re-screened proportions in the two populations were the same — 74%. However, the timing of re-screening in NSW was more variable — about the same proportion of women were re-screened in a 26-month period in NSW (13–39 months after the negative index smear), as were screened in a 4-month period in Oxfordshire (35–39 months after the negative index smear). Beyond 39 months, the cumulative re-screened proportion was lower in NSW than in Oxfordshire. By 60 months (5 years), the re-screened proportions in NSW and Oxfordshire were 81% and 94%, respectively.
After the introduction of organised screening, the average incidence rate of cervical cancer in the period 1991–1993 to 1998–2000 fell by 33% in Australia and 33% in the UK in women aged 20–69 years (Box 3).
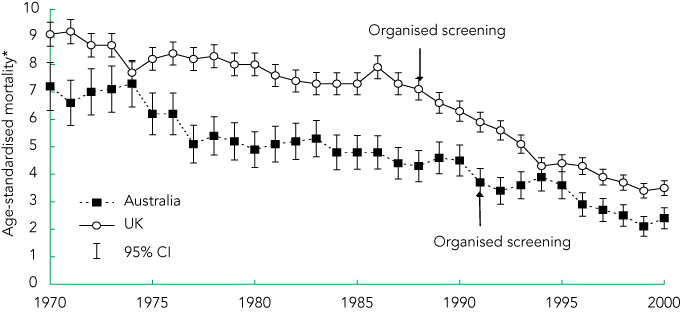
In the early 1970s, mortality rates of cervical cancer in Australia were, on average, about 20% lower than those in the UK. Since the early 1980s, mortality in Australia has been about a third lower than in the UK (Box 4). Joinpoint regression analysis was performed to detect any changes in cervical cancer mortality trends from 1965 to 2001.25 In both countries, a significant downturn in the mortality trend was detected within a few years of the introduction of organised screening.
During the period of opportunistic screening (before 1988, see Box 3), the average mortality rate in women aged 20–69 years declined by 10% in Australia and 7% in the UK. This was probably due to several factors, potentially including increased access to screening and treatment and increased survival resulting from a shift towards a greater proportion of localised cancers.26 While a fall was observed in ages 50–69 years in both countries, mortality in women aged 20–49 years stayed relatively stable in Australia and increased in the UK. During the period of organised screening (from 1991, see Box 3), the average mortality rate at age 20–69 years fell by 36% in both countries, corresponding to a fall in the average from 3.6 to 2.3 per 100 000 women in Australia and from 5.5 to 3.5 per 100 000 women in the UK.
Because organised screening was introduced in Australia 3 years later than in the UK (1991 versus 1988), it was possible that the primary comparison of changes in rates from 1991–1993 to 1998–2000 reflected initial implementation issues in Australia to a greater degree than in the UK. To check for this possibility, the reduction in average mortality in Australia was also calculated from 1994–1996 to 2001–2003 (ie, over a later period to allow for residual implementation of the organised program between 1991 and 1994). This supplementary calculation used recent data published by the Australian Institute of Health and Welfare (AIHW).6,13 (It should be noted that, for the years in which the data overlap, there are slight differences in mortality rates published by the AIHW and WHO.) At the time of writing, cervical cancer incidence data in Australia were not available after 2002 and therefore changes in incidence over the later period could not be assessed. Using the AIHW data, the reduction in mortality in women aged 20–69 years from 1994–1996 to 2001–2003 was 38% — similar to the 36% reduction observed from 1991–1993 to 1998–2000 using the WHO mortality database.
Several elements other than screening interval policy may contribute to these differences, including the timing of reminder letters, practitioner incentives, disincentives to opportunistic early re-screening, and geographic factors. There is little incentive to re-screen early in the UK, and the high re-screening rate observed at 5 years probably reflects practice incentives based on 5-yearly participation rates.16 In addition, each woman in the UK is registered with a single practice, which facilitates follow-up and tracing. In Australia, the lower 5-year re-screened proportion may partly reflect the difficulties in reaching remote and rural populations. It is known that 2-yearly screening participation rates are lower in Indigenous Australians,27 and it is likely that 5-yearly re-screening proportions are also lower in this group. With the introduction in 2001 of practice incentive payments in Australia based on 4-yearly re-screening rates, the re-screened proportion at 4 years may increase.
Both countries show a low incidence of cervical cancer for women aged 20–24 years (< 2.5 per 100 000 women), which was one reason for the 2003 increase in the age of starting screening to 25 years in the UK. In women aged 20–49 years, cervical cancer incidence was lower in Australia than in the UK, whereas in women aged 50–69 years, it was slightly higher in Australia than in the UK. Given the differences in screening uptake between the two countries, these results are consistent with a UK audit of screening histories, which found that, for women younger than 55 years, the protective effect associated with a negative smear was reduced after 3 years, but, for women 55 years and over, the protective effect remained relatively stable for at least 5 years.28 In this context, the high level of re-screening achieved by 5 years in the UK may play a role in the optimisation of screening in older women within the eligible target population.
Our comparison of national cancer rates assumes that the effect of screening is not influenced by differences between the two countries in risk factors for cervical cancer, such as the distribution of oncogenic human papillomavirus (HPV) types and established cofactors — use of oral contraceptives,29 multiparity,30 age at first full-term pregnancy30 and use of tobacco.31 A second assumption is that the population samples used to assess patterns of screening uptake in the two countries are representative of the total eligible population; as discussed, there is reasonable evidence to support this. A caveat is that estimates of the re-screening proportion obtained from screening registry data in both countries are likely to be slightly lower than the true value, as women who move or change name are not always identified as duplicate entries. A further consideration is that some women with negative smears have a history of previous abnormal smears or treatment to the cervix, such that they are recalled before the recommended screening interval in accordance with management recommendations.32 However, as the relevant management recommendations were broadly similar in the two countries, this should not substantially affect the comparison.
The primary objective of organised cervical screening is to reduce mortality from cervical cancer. Since the introduction of organised screening, Australia and the UK have experienced similar proportional reductions in cervical cancer incidence and mortality. Therefore, the 2-yearly screening policy in Australia and the predominantly 3-yearly screening policy in the UK appear to have been of broadly similar effectiveness. This supports the findings of the original 1986 IARC working group on cervical screening intervals.2
In 2004, the IARC convened an international expert working group on cervical screening33 and recommended that:
For women over the age of 50 years, a 5-year screening interval is considered appropriate; and
For women aged 25–49 years, a 3-year rather than 5-year interval might be considered in countries with the necessary resources.
1 Cervical screening policy and estimated participation rates (proportion of eligible women who had at least one Pap smear during each period)
2 Cumulative proportion of women with a negative smear who were re-screened, by time since the negative smear, in Australia (New South Wales) and the United Kingdom*
3 Changes in age-standardised cervical cancer incidence and mortality rates in Australia and the United Kingdom (England and Wales)
Per cent decrease in average annual rate |
|||||||||||||||
Received 20 February 2006, accepted 25 June 2006
- Karen Canfell1
- Freddy Sitas1
- Valerie Beral2
- 1 Cancer Epidemiology Research Unit, The Cancer Council New South Wales, Sydney, NSW.
- 2 Cancer Epidemiology Unit, University of Oxford, Oxford, UK.
We thank Professor Bruce Armstrong and Associate Professor Erich Kliewer for reviewing the manuscript and providing helpful advice; Sue Zeckendorf for providing data from the NSW Pap Test Register; Dr Winifred Gray, Pat Martingale and collaborators for providing data from the Oxfordshire National Health Service Cervical Screening Programme; Caroline Vass from the Oxford Cancer Intelligence Unit for advising on the cervical screening process in the United Kingdom; Krys Baker and her team from the Cancer Epidemiology Unit at the University of Oxford for assistance with data collection; and the Million Women Study Collaborators.
Karen Canfell was employed to September 2005 by Polartechnics Limited, a company which designs and markets cervical screening equipment. She holds no shares in this company and has not engaged in any paid consultancy or employment with them since September 2005.
- 1. National Health and Medical Research Council. Screening to prevent cervical cancer: guidelines for the management of asymptomatic women with screen detected abnormalities. Canberra: Commonwealth of Australia, 2005. http://www.nhmrc.gov.au/publications/synopses/wh39syn.htm (accessed Feb 2006).
- 2. Screening for squamous cervical cancer: duration of low risk after negative results of cervical cytology and its implication for screening policies. IARC Working Group on evaluation of cervical cancer screening programmes. Br Med J (Clin Res Ed) 1986; 293: 659-664.
- 3. Dickinson JA. Cervical screening: time to change the policy. Med J Aust 2002; 176: 547-550. <MJA full text>
- 4. National Health and Medical Research Council. Media release: new guidelines on cervical screening, acute pain management and recreational water. 2005. http://www.nhmrc.gov.au/news/media/rel05/guidelines.htm (accessed Feb 2006).
- 5. National Health Service Cancer Screening Programmes. Colposcopy and programme management: guidelines for the NHS Cervical Screening Programme. Sheffield, UK: NHS Cancer Screening Programmes, 2004. (NHSCSP Publication No. 20.)
- 6. Australian Institute of Health and Welfare. Cervical screening in Australia 1998–1999; 1999–2000; 2000–2001 and 1999–2000; 2001–2002; 2002–2003; 2003–2004. Canberra: AIHW, 2002–2006. (AIHW Cat. Nos. CAN 11, 16, 19, 22, 26, 28; Cancer Series Nos. 16, 21, 24, 27, 31, 33.)
- 7. NSW Cervical Screening Program, NSW Pap Test Register. Annual statistical report 2000. Sydney: Westmead Hospital, 2001.
- 8. Department of Health. Bulletin 2001/22, Cervical Screening Programme, England: 2000–2001. London: Department of Health, 2001.
- 9. Australian Bureau of Statistics. National Health Survey. Summary of results, Australia, 2001. Canberra: ABS, 2002. (ABS Cat. No. 4364.0.)
- 10. Canfell K, Beral V, Green J, et al. The agreement between self-reported cervical smear abnormalities and screening programme records. J Med Screen 2006; 13: 72-75.
- 11. Parkin DM, Whelan SL, Ferlay J, et al. Cancer incidence in five continents, Vol. I to VIII. Lyon, France: IARC CancerBase No. 7, 2005.
- 12. International Agency for Research on Cancer. WHO mortality database extracted from the World Health Organization Databank, 2005. http://www-dep.iarc.fr/ (accessed Feb 2006).
- 13. Australian Institute of Health and Welfare. GRIM (general record of incidence of mortality) books. Version 8. Canberra: AIHW, 2005.
- 14. Office of National Statistics, Cancer Statistics Registrations. Registrations of cancer diagnosed in 1998; 1999; 2000. England. London: ONS, 2002–2003. (Series MB1 Nos. 29, 30, 31.)
- 15. Bray F. Age-standardization. In: Parkin DM, Whelan SL, Ferlay J, et al. Cancer incidence in five continents. Vol VIII. Lyon, France: International Agency for Research on Cancer, 2002. (IARC Scientific Publications No. 155.)
- 16. Patnick J. Cervical cancer screening in England. Eur J Cancer 2000; 36: 2205-2208.
- 17. Howe A, Owen-Smith V, Richardson J. Health authority cervical screening recall policies and time since last smear: a retrospective cohort analysis in the north west England. J Med Screen 2003; 10: 184-188.
- 18. Farnsworth A, Mitchell HS. Prevention of cervical cancer. Med J Aust 2003; 178: 653-654. <MJA full text>
- 19. Mitchell H. Outcome after a cervical cytology report of low-grade squamous abnormality in Australia. Cancer 2005; 105: 185-193.
- 20. Soutter WP, Fletcher A. Invasive cancer of the cervix in women with mild dyskaryosis followed up cytologically. BMJ 1994; 308: 1421-1423.
- 21. Rana DN, Marshall J, Desai M, et al. Five-year follow-up of women with borderline and mildly dyskaryotic cervical smears. Cytopathology 2004; 15: 263-270.
- 22. Moss S, Gray A, Legood R, et al. Effect of testing for human papillomavirus as a triage during screening for cervical cancer: observational before and after study. BMJ 2006; 332: 83-85.
- 23. Australian Government Department of Health and Ageing. Annual report 2003-04. http://www.health.gov.au/internet/wcms/publishing.nsf/content/annual-report2003.htm (accessed Nov 2006).
- 24. Medicare Australia. PIP news update. 2005. http://www.medicareaustralia.gov.au/resources/incentives_allowances/pip_news/pip_news_may_2005.pdf (accessed Feb 2006).
- 25. Kim HJ, Fay MP, Feuer EJ, et al. Permutation tests for joinpoint regression with applications to cancer rates. Stat Med 2000; 19: 335-351.
- 26. Taylor R, Morrell S, Mamoon H, et al. Decline in cervical cancer incidence and mortality in New South Wales in relation to control activities (Australia). Cancer Causes Control 2006; 17: 299-306.
- 27. Coory MD, Fagan PS, Muller JM, Dunn NAM. Participation in cervical cancer screening by women in rural and remote Aboriginal and Torres Strait Islander communities in Queensland. Med J Aust 2002; 177: 544-547. <MJA full text>
- 28. Sasieni P, Adams J, Cuzick J. Benefit of cervical screening at different ages: evidence from the UK audit of screening histories. Br J Cancer 2003; 89: 88-93.
- 29. Smith JS, Green J, Berrington DG, et al. Cervical cancer and use of hormonal contraceptives: a systematic review. Lancet 2003; 361: 1159-1167.
- 30. International Collaboration of Epidemiological Studies of Cervical Cancer. Cervical carcinoma and reproductive factors: collaborative reanalysis of individual data on 16,563 women with cervical carcinoma and 33,542 women without cervical carcinoma from 25 epidemiological studies. Int J Cancer 2006; 119: 1108-1124.
- 31. International Collaboration of Epidemiological Studies of Cervical Cancer. Carcinoma of the cervix and tobacco smoking: collaborative reanalysis of individual data on 13,541 women with carcinoma of the cervix and 23,017 women without carcinoma of the cervix from 23 epidemiological studies. Int J Cancer 2006; 118: 1481-1495.
- 32. Morrell S, Mamoon H, O’Callaghan J, et al. Early cervical cancer rescreening. J Med Screen 2002; 9: 26-32.
- 33. International Agency for Research on Cancer. Cervix cancer screening. IARC handbooks of cancer prevention. Volume 10. Lyon, France: IARC Press, 2005.





Abstract
Objective: To compare cervical screening policy, screening uptake, and changes in cervical cancer incidence and mortality between Australia and the United Kingdom.
Design: Analysis of screening registry data and national cancer statistics.
Setting: In Australia, organised cervical screening was initiated in 1991 for sexually active women aged 18–69 years, with a recommended 2-yearly interval. In the UK, organised screening began in 1988 for women aged 20–64 years, with a recommended 3-yearly interval in most regions.
Results: Estimated lifetime screening participation rates in 2001 were similar in the two countries, at 88% in Australia and 90% in the UK. For women who were screened and had a negative result, the median time to the next screen was 27 months in Australia and 38 months in the UK. At 39 months, equivalent proportions (74%) had been re-screened in the two countries, and by 60 months the re-screened proportions were 81% in Australia and 94% in the UK. From 1991–1993 to 1998–2000, the incidence of cervical cancer in women aged 20–69 years fell by 33% in Australia and 33% in the UK, and mortality from cervical cancer fell by 36% in both countries.
Conclusions: After the introduction of organised screening, similar reductions in cervical cancer incidence and mortality were achieved in Australia and the UK. Therefore, the 2-yearly screening policy in Australia and the predominantly 3-yearly screening policy in the UK appear to have been of broadly similar effectiveness.