Prostate cancer (PCA) is the most common non-skin cancer in men (over 11 000 cases in Australia in 2001), and, with a lifetime risk of 1 in 10 men, it is the second leading cause of death from cancer in men.1 Despite these statistics, there have been very few studies of the psychosocial impact on the patient and his family of PCA and its treatment, and limited research on psychosocial interventions to promote adaptive coping.2-4
Clearly, the negative psychosocial effects of this cancer represent a major health care issue. As with breast cancer, research on men’s reactions to the diagnosis and treatment of this intensely personal condition — without involving partners — risks overlooking a key relational dimension.5 As the side effects of mastectomy (disfigurement and scarring) and hormone therapy and chemotherapy (hair loss) can challenge a woman’s self-image as mother, wife and sexual partner, similarly the side effects of prostatectomy (impotence and urinary incontinence), hormone therapy (feminisation and loss of libido) and external beam radiotherapy (bowel disturbance) can shake a man’s view of himself in his various roles.
A review of the limited number of reports on the psychosocial impact of PCA on patient and partner demonstrates a pressing need for integrated research to establish the psychological needs of couples as they grapple with decisions about treatment and their consequences.6 A prospective observational study incorporating different stages of the patient and partner’s journey is needed to guide the development of a focused, coherent psychosocial intervention, which can be tested and, if effective, woven into routine clinical care to improve psychosocial adjustment. Here, we report the findings of such a study and recommend the development of a program of counselling.
Our multisite study was designed to elicit data from two groups of couples facing PCA: (i) couples where the patient has just learnt he has localised (potentially curable) PCA; and (ii) couples where the patient has been told that he has metastatic (incurable) PCA. Consecutive clinic attendees who met study criteria (Box 1) were recruited between March 2002 and March 2005 by oncologists and urologists working in public hospitals and private practices in metropolitan Melbourne. (The patients were also recruits for a larger, patient only, 18-month longitudinal study.) Interviews took place before the launch of definitive treatments, such as prostatectomy or radiotherapy, or soon after.
The anxiety and depression modules of the Composite International Diagnostic Interview (CIDI)7 were administered to patients and partners. The CIDI, which derives psychiatric diagnoses according to the Diagnostic and statistical manual of mental disorders 4th edition (DSM-IV),8 has confirmed reliability and validity.7 The CIDI computer software takes account of symptoms arising from physical illness (including cancer and its treatments) to ensure they are not incorrectly ascribed to depression or anxiety diagnoses.7 The CIDI has recency codes that separate past psychiatric disorders from current disorders. For our study, only DSM-IV diagnoses over the 12 months before the interview are reported (12-month prevalence rates).
The Brief Symptom Inventory (BSI)9 is a 53-item self-report psychological symptom scale. It yields a global rating of psychological status called the Global Severity Index (GSI), comprising a grand total of scores divided by the number of valid items. The BSI also provides subscale scores for depression and for anxiety. In each case, a higher score denotes greater psychological distress. The BSI’s ability to discriminate clinically distressed from non-distressed samples has been well established, using structured psychiatric interviews as benchmarks.9 Extensively used in psycho-oncology research, the BSI is sensitive to changes in psychological adjustment and to psychopathology.10 Specific threshold scores of psychiatric “caseness” (where an individual’s levels of symptoms are deemed to correspond to a diagnosable psychiatric disorder) have been established for both men and women.11
The Dyadic Adjustment Scale (DAS)12 is a six-item self-report scale widely used in the context of families experiencing cancer.13 The DAS is designed to measure the quality and level of satisfaction of romantic relationships in cohabiting couples.14 The DAS yields the DAS Full Scale score, comprising the sum of the recoded item scores; a higher score represents greater marital satisfaction (maximum 30).
The remaining 103 couples (73% of identified couples) were recruited. The mean age of the patients was 66 years (SD, 9.5; range, 43–92), and that of their partners was 62 years (SD, 9.3; range, 41–83) (Box 2). The patients’ ages were in the expected range.15
Forty-seven patients (46%) were diagnosed with localised PCA and 56 (54%) with metastatic disease. Twenty-four of the 103 patients (23%) received no active treatment during the study period, and 49 (48%) had hormone treatment. Of the 47 patients with localised PCA, 13 (28%) had a radical prostatectomy and five (11%) had some form of radiotherapy. Of the 56 patients with metastatic PCA, eight (14%) received chemotherapy. The range of treatments was generally consistent with Victorian Cancer Registry data.16 Seventy-one of the patients (69%) were recruited from public hospital clinics, the remainder from private practice.
Patients and partners not participating at Time 2 were significantly more likely to have been facing advanced PCA at Time 1 (if a patient died, his partner was not eligible to continue, but if he survived but withdrew she remained eligible) (Box 3). The age of patients (but not partners) was significantly higher among non-participants at Time 2. There was no significant difference between participants and non-participants at Time 2 in GSI or DAS scores (data not shown).
The rates of DSM-IV anxiety and depression for 90 patients and 103 partners at Time 1 are given in Box 4. The partners had rates of major depression more than twice the normal rate for the Australian (Victorian) community;17 patient rates were also higher than the community rate but proportionally not as elevated. A similar pattern emerged for rates of generalised anxiety disorder and other anxiety disorders in patients and partners.
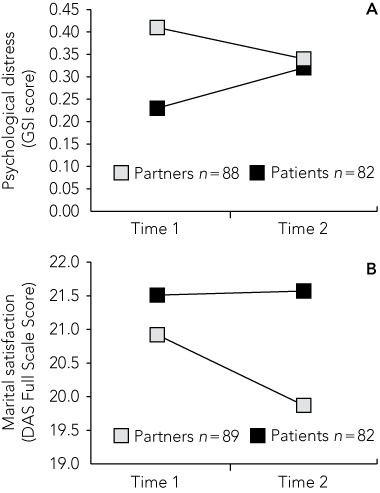
Overall mean psychological distress (GSI) in partners at Time 1 was almost twice as high as in patients (Box 5). Similarly, depressive symptoms and anxiety symptoms at Time 1 (BSI Depression and Anxiety Subscales) were substantially higher in partners compared with patients.
Patients experienced an increase in distress at Time 2 compared with Time 1 (F1,81, 7.26; P < 0.05; n = 82), whereas partners reported a reduced level of distress at Time 2 compared with Time 1 (F1,87, 3.99; P < 0.05; n = 88) (Box 6A). At Time 2, distress in partners did not differ significantly from that of patients, unlike the situation 6 months earlier.
Patients’ and partners’ marital satisfaction at Time 1 was not significantly different. At Time 2, however, partners’ satisfaction was lower than that of patients (t, 2.35; df, 70; P < 0.05; n = 71). Moreover, partners’ satisfaction fell from Time 1 to Time 2 (F1,88, 5.71; P < 0.05, n = 89), whereas that of patients did not change significantly (Box 6B).
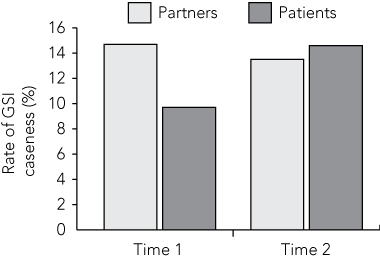
At Time 1, 15% of partners (n = 102) exceeded the female GSI threshold for psychiatric caseness;11 at Time 2, 14% of partners (n = 89) met criteria for psychiatric caseness. Ten per cent of patients (n = 103) met criteria for psychiatric caseness at Time 1, but this went up to 15% of patients (n = 82) at Time 2 (Box 7).
To our knowledge, this is the first prospective, observational study of couples facing PCA using a sample of this size and gathering information from both patients and partners concurrently.6 Our findings of a change in the psychological status of both partners and patients over a period of 6 months, and of different patterns of change in the two groups, are intriguing. The partners were more distressed at the time of diagnosis, and less so 6 months later; the converse occurred in the patients. With respect to marital satisfaction, there were again different patterns, with patients’ satisfaction levels remaining constant over 6 months but partners’ satisfaction declining.
The diminution in distress in partners supports findings from a previous study that the anxiety of partners of patients with advanced PCA decreased over a period of 6 months.18 That study is the only prospective one, other than our own, that includes partners. The worsening marital satisfaction we identified in partners agrees with the findings of a cross-sectional study of 164 partners, a mean of 4.3 years after initiation of PCA treatment; most reported dissatisfaction with their relationship.19
The elevated rates of diagnosable (DSM-IV) depression and anxiety in the partners we studied, compared with both a community sample of women and the patients diagnosed with cancer, also agree with previous research involving cross-sectional measurement of distress in patients with PCA and their partners.20,21 Contradictory findings in one study22 — lower distress in partners than patients — may be explained by sampling (patients were in remission with no metastatic disease and no hormone therapy and were interviewed many months after completing successful therapy). In the former two studies,20,21 a broader range of patient stages and treatment modalities were sampled.
Comparisons of mean BSI scores between patients and partners (or raw rates of DSM-IV diagnoses) need to be made with caution in the light of the widely established epidemiological finding that women report anxiety and depression symptoms at higher rates than men.23 However, rates of BSI caseness should not be affected by this phenomenon, since separate caseness thresholds have been derived from normative samples for men and women.11
Studies that obtain their sample from cancer registries, such as that of Valdimarsdottir et al,24 may enjoy reduced selection bias, but the inevitable delays in registration into databases preclude researcher access to patients (and partners) at the time they are dealing psychologically with the diagnosis and its implications. Studies that seek recruits through advertisements, attendees at information sessions, or self-help group memberships, such as that of Ko et al,25 face considerable potential selection bias as participants are self-selected. Recruitment of consecutive clinic attendees is a favoured form of sampling in psycho-oncology research.5,6,10
Our findings suggest the value of a couple-focused counselling program, similar to that devised and applied for couples facing breast cancer.26 A preventive intervention of this type could help the growing number of middle-aged and older men affected by PCA and their partners to, as much as possible, enjoy productive lives together. Such an intervention could buttress the most salient form of social support both patient and partner will need — each other’s — in the months and years that follow the diagnosis of this common cancer.
1 Eligibility criteria for patients and partners
Diagnosis of prostate cancer
Localised prostate cancer — T1–T3, N0, M0 (pretreatment or early treatment)
OR
Metastatic prostate cancer — N+, M+
Does not currently have other cancer
Does not have intellectual disability, dementia, or psychotic disorder
Can speak/understand English
Female partner of man eligible for the men’s study (marital status unimportant)
Does not have currently active cancer or other severe debilitating illness
Does not have intellectual disability, dementia, or psychotic disorder
Can speak/understand English
3 Comparison of stage of prostate cancer (PCA) at Time 1 in partners and patients completing and not completing Time 2 assessments
4 Rates of DSM-IV psychiatric disorders diagnosed in prostate cancer patients and their partners* at Time 1, compared with 12-month prevalence rates for adults†
Community prevalence of psychiatric diagnosis for adult males† |
|||||||||||||||
Community prevalence of psychiatric diagnosis for adult females† |
|||||||||||||||
|
* Diagnosed using the anxiety and depression modules of the Composite International Diagnostic Interview.7 † Rates for adults in the Victorian community (Australian Bureau of Statistics, 1997).17 DSM-IV = Diagnostic and statistical manual of mental disorders, 4th edition. | |||||||||||||||
5 Comparison of mean Brief Symptom Inventory (BSI) scores for patients and their partners at the time of diagnosis of localised/metastatic prostate cancer (Time 1)
Received 16 June 2006, accepted 31 August 2006
- Jeremy W Couper1
- Sidney Bloch1
- Anthony Love2
- Gillian Duchesne3
- Michelle Macvean4
- David W Kissane5
- 1 Department of Psychiatry, University of Melbourne, Melbourne, VIC.
- 2 School of Psychological Science, La Trobe University, Melbourne, VIC.
- 3 Peter MacCallum Cancer Centre, Melbourne, VIC.
- 4 The Cancer Council Victoria, Melbourne, VIC.
- 5 Department of Psychiatry and Behavioral Sciences, Memorial Sloan-Kettering Cancer Centre, New York, NY, USA.
Our study was supported by grants from the National Health and Medical Research Council and the Bethlehem Griffiths Research Foundation. These funding bodies had no role in study design, data collection, analysis, interpretation or writing of this article. We would like to thank all of the patients and their partners who kindly participated in this study and the clinicians who recruited them, namely: David Angus, Adam Broad, Alex Cato, Michael Chamberlain, Martin Cherk, David Clarke, Laurence Cleeve, Owen Cole, Niall Corcoran, Anthony Costello, Mike Dally, Ian Davis, Anthony Dowling, Gillian Duchesne, Farshad Foroudi, Mark Frydenberg, Jeremy Goad, Bronwyn Matheson, Jeremy Millar, Robin Murray, Justin Peters, Mark Rosenthal, Andrew See, Jeremy Shapiro, Ross Snow, Jo Stewart, and Keen Hun Tai.
None identified.
- 1. Australian Institute of Health and Welfare, and Australasian Association of Cancer Registries. Cancer in Australia 2001. Canberra: AIHW, 2004. (AIHW Catalogue No. CAN 23; Cancer Series No. 28.)
- 2. Manne S, Babb J, Pinover W, et al. Psychoeducational group intervention for wives of men with prostate cancer. Psychooncology 2004; 13: 37-46.
- 3. Davison BJ, Goldenberg SL, Gleave ME, Degner LF. Provision of individualized information to men and their partners to facilitate treatment decision making in prostate cancer. Oncol Nurs Forum 2003; 30: 107-114.
- 4. Giarelli E, McCorkle R, Monturo C. Caring for a spouse after prostate surgery: the preparedness needs of wives. J Fam Nurs 2003; 9: 453-485.
- 5. Baider L, Kaplan De-Nour A. Cancer and couples — its impact on the healthy partner: methodological considerations. In: Baider L, Cooper CL, Kaplan De-Nour A, editors. Cancer and the family. Chichester: Wiley, 2000: 41-51.
- 6. Couper J, Bloch S, Love A, et al. Psychosocial adjustment of female partners of men with prostate cancer: a review of the literature. Psychoncology 2006; Mar 6: [epub ahead of print].
- 7. World Health Organization. Composite International Diagnostic Interview. CIDI-Auto Version 2.1. Sydney: Wild and Woolley, 1997.
- 8. American Psychiatric Association. Diagnostic and statistical manual of mental disorders, fourth edition (DSM-IV). Washington, DC: APA, 1994.
- 9. Derogatis LR, Melisaratos N. The Brief Symptom Inventory: an introductory report. Psychol Med 1983; 13: 595-605.
- 10. Kissane DW, Bloch S. Family focused grief therapy. Buckingham: Open University Press, 2002.
- 11. Derogatis LR, Spencer PM. Administration and procedures. BSI Manual – I. Baltimore: Clinical Psychometric Research, 1982.
- 12. Sharpley CF, Cross DG. A psychometric evaluation of the Spanier Dyadic Adjustment Scale. J Marriage Fam 1982; 44: 739-741.
- 13. Ptacek JT, Pierce GR, Ptacek JJ, Nogel C. Stress and coping processes in men with prostate cancer: the divergent views of husbands and wives. J Soc Clin Psychol 1999; 18: 299-324.
- 14. Spanier GB. Measuring dyadic adjustment: new scales for assessing quality of marriage and similar dyads. J Marriage Fam 1976; Feb: 15-28.
- 15. Giles G. Canstat: Cancer in Victoria 2003. Melbourne: The Cancer Council Victoria — Cancer Epidemiology Centre, 2004.
- 16. Frydenberg M, Giles GG, Mameghan H. Prostate cancer in Victoria in 1993: patterns of reported management. Med J Aust 2000; 172: 270-274.
- 17. Australian Bureau of Statistics. Mental health and wellbeing profile of adults Victoria, 1997. Canberra: ABS, 1997. (Catalogue No. 4326.2.40.001.)
- 18. Kornblith AB, Herndon JE 2nd, Zuckerman E, et al. The impact of docetaxel, estramustine, and low dose hydrocortisone on the quality of life of men with hormone refractory prostate cancer and their partners: a feasibility study. Ann Oncol 2001; 12: 633-641.
- 19. Neese LE, Schover LR, Klein EA, et al. Finding help for sexual problems after prostate cancer treatment: a phone survey of men’s and women’s perspectives. Psychooncology 2003; 12: 463-473.
- 20. Cliff AM, Macdonagh RP. Psychosocial morbidity in prostate cancer: II. A comparison of patients and partners. BJU Int 2000; 86; 834-839.
- 21. Kornblith AB, Herr HW, Ofman US, et al. Quality of life of patients with prostate cancer and their spouses: the value of a database in clinical care. Cancer 1994; 73: 2791-2802.
- 22. Baider L, Ever-Hadani P, Goldzweig G, et al. Is perceived family support a relevant variable in psychological distress? A sample of prostate and breast cancer couples. J Psychosom Res 2003; 55: 453-460.
- 23. Weissman MM, Klerman GL. Sex differences and the epidemiology of depression. Arch Gen Psychiatry 1977; 34: 98-111.
- 24. Valdimarsdottir U, Helgason AR, Furst C-J, et al. The unrecognised cost of cancer patients’ unrelieved symptoms: a nationwide follow-up of their surviving partners. Br J Cancer 2002; 86: 1540-1545.
- 25. Ko CM, Malcarne VL, Varni JW, et al. Problem-solving and distress in prostate cancer patients and their spousal caregivers. Support Care Cancer 2005; 13: 367-374.
- 26. Scott JL, Halford WK, Ward BG. United we stand? The effects of a couple-coping intervention on adjustment to early stage breast or gynaecological cancer. J Consult Clin Psychol 2004; 72: 1122-1135.





Abstract
Objective: To assess the psychosocial impact of the diagnosis of either localised or metastatic prostate cancer (PCA) on patients and their female partners.
Design: Observational, prospective study at Time 1 and 6 months later at Time 2 of two groups of couples facing PCA. Time 1 was when patients were first diagnosed with histologically confirmed localised (potentially curable) PCA or metastatic (incurable) PCA.
Main outcome measures: Depression and anxiety disorders according to the Diagnostic and statistical manual of mental disorders 4th edition (DSM-IV); psychological distress; marital satisfaction.
Results: At Time 1, partners had rates of DSM-IV major depression and generalised anxiety disorder twice those of women in the Australian community, and considerably higher than the patients’ rates. At Time 2, psychological distress in partners had lessened but that in patients had increased. On the other hand, at Time 2, partners’ marital satisfaction had deteriorated.
Conclusions: To be fully effective, interventions aimed at reducing the psychosocial morbidity of PCA must involve both patient and partner, rather than the patient alone.