Asthma is a National Health Priority Area in Australia, but asthma in older people has received relatively little attention. Here, we briefly describe Australian data on the burden of asthma in older people.
We first discuss some of the problems that arise in identifying asthma in older people, and then present data on prevalence, mortality and hospital utilisation. These data are based on information recently published by the Australian Centre for Asthma Monitoring.1 Finally, we discuss what needs to be done to improve the quality of data on asthma in older Australians.
It has long been recognised that asthma is a chronic illness characterised by widespread variable airflow obstruction and the respiratory symptoms (shortness of breath, wheeze, chest tightness and cough) that accompany this.2 In addition, characteristic pathological changes in the airway are well recognised.3. While this understanding of the nature of asthma enables clinicians to correctly identify many people with the disease, it is not universally applicable. There are several theoretical and practical reasons for this:
The symptoms are not unique to asthma but are shared by other diseases, particularly in the elderly and the young;
The pathological features of the disease can rarely be elicited in clinical practice;
The lung function abnormalities characteristic of asthma are measured on a continuous scale, and the distinction between reversible airflow obstruction diagnostic of asthma and that which is normal or characteristic of other disease states is arbitrary;
As asthma is variable over time, any or all of the features may not be present at any particular time;
Certain diseases that share some of the features of asthma may be classified as a type of asthma or as a separate disease entity (eg, chronic asthma with chronic airflow limitation, and allergic bronchopulmonary aspergillosis).
The symptoms of asthma in older people, as in younger age groups, are dyspnoea, wheeze, chest tightness and cough. However, these symptoms are not specific to asthma. In particular, dyspnoea is common in older people. While it may be the sole presenting symptom of asthma, it may also be attributable to various non-disease states (eg, poor physical fitness, obesity, or excessive expectations as to exercise capacity) or disease states (eg, cardiac failure or chronic obstructive pulmonary disease [COPD]).4,5 However, angina, interstitial lung disease, pulmonary vascular disease, neuromuscular or chest-wall disease, and anaemia may all cause dyspnoea and chest tightness and, under some circumstances, can be confused with asthma in older patients. Indeed, several causes of dyspnoea may coexist in the same patient. While most of these diagnoses can be distinguished by investigation (in particular, spirometry), this is not commonly performed in practice or in general health surveys.
Information on the prevalence of asthma is derived from community-based surveys in which participants are asked about a reported diagnosis of asthma or symptoms of asthma. For the reasons described above, prevalence data that are based on reported diagnoses of asthma may be problematic. However, for similar reasons, it is also difficult to draw inferences about the prevalence of asthma from questionnaire-based measures of the prevalence of symptoms. While the use of spirometry and measures of airway hyper-responsiveness would improve the validity of the available data, no nationwide surveys have used such measurements.
In 2001, the prevalence of current asthma among people aged over 55 years was 7.3% for men and 10.7% for women (Box 1). These data are derived from the National Health Survey6 and refer to individuals who report that they have had asthma diagnosed by a doctor, and that they still have it. The prevalence of asthma is similar among all adult age groups, but is lower in adults than in children. It is not yet clear whether this is attributable to remission of asthma with age or to the higher incidence of asthma in more recent birth cohorts.
Surveys show that women have a higher prevalence of asthma than men, and this is evident in all adult age groups. Without objective measures, it is not possible to ascertain the contribution of diagnostic bias (ie, a preference for labelling women with asthma as opposed to COPD) to this difference.
The National Health Survey6 and the National Indigenous Health Survey 20017 show a higher prevalence of asthma among Indigenous than non-Indigenous Australian adults. The difference is greatest in older adults (21% v 9% in people aged 55 years and over).
From 1997 to 2001, there were, on average, 98 asthma-related deaths per year among men and 182 per year among women aged 65 and over.1 Box 2 shows that asthma-related death rates in people aged 65 and over far exceed rates in younger age groups. In fact, most deaths attributed to asthma occur in this older age group. Within this group, certain subgroups may be at greater risk of death from asthma. Although asthma is actually less common among older people of non-English-speaking backgrounds, their mortality rate from asthma is relatively higher (134 v 70 per 100 000 people with asthma).1
Interpreting these data requires caution. Many deaths from asthma occur outside hospital, and few cause-of-death diagnoses are confirmed by autopsy. The cause of death reflects the diagnostic label that was applied before death. Hence, the problems inherent in establishing the diagnosis of asthma in older people and the potential for misclassifying other causes of dyspnoea as “asthma” in older people may confound data on mortality from asthma.8-10
Data on seasonal variation are less subject to misclassification bias. Among older people, there is a seasonal increase in mortality from asthma in the winter months of July and August. The lowest death rates from asthma in this age group are recorded in February to April. The seasonal variation observed in older people differs from that recorded in younger people (Box 3).
The largest users of health care services for asthma, including hospital care, are children. However, longer hospital stays mean that the number of bed-days used for asthma is relatively high among older people (Box 4). Similarly, although children have the highest rates of visits to general practitioners for asthma, rates in older people are higher than rates in younger adults (Box 5).
Indigenous older people have particularly high rates of hospitalisation for asthma compared with non-Indigenous people (819 v 166 per 100 000 people aged 55 years and over).1
Improving the quality of data on asthma requires improvements to existing survey measures and improvements to the accuracy of diagnosis (which forms the basis of the routine data sources). Spirometry has a central role in evaluating dyspnoea in older people and in identifying individuals with obstructive lung disease. It also contributes to the diagnosis of asthma in this population. There is a significant association between lower-than-predicted lung function and mortality from both respiratory and vascular diseases.11,12 Hence, to improve the quality of information about asthma and other causes of dyspnoea in older people, it is important to incorporate measures of spirometric function into health surveys of representative samples of the general population. It is equally important that doctors caring for older patients are encouraged to measure spirometric function as part of the diagnostic work-up of patients with dyspnoea.
Understanding the effects of ageing on asthma requires separation of age effects from cohort effects. This cannot be achieved in individual cross-sectional studies, but requires analysis of cross-sectional studies, repeated at intervals, with the use of similar methods, as well as follow-up of cohorts. Within Australia, there are several cohort studies involving children and young adults with asthma, but few of these have progressed to include older people.13 On the other hand, several cohorts of older people are either established or proposed to be established in the near future to study various health problems in this age range. These cohorts will provide a valuable opportunity to learn more about the effect of ageing on asthma.
1 Prevalence (95% CIs) of current asthma, by age group and sex in Australia, 2001*
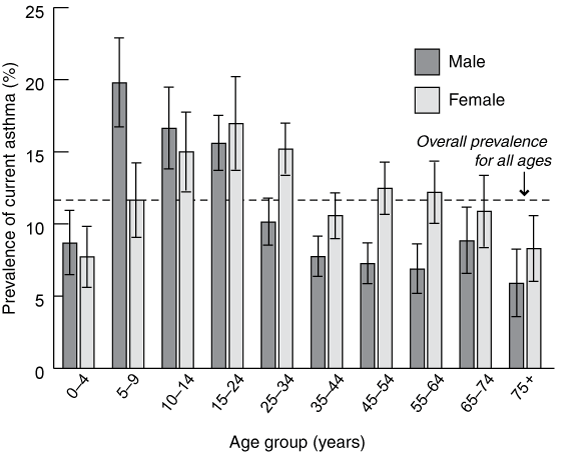
* Data from the National Health Survey, 2001, Australian Bureau of Statistics. Reproduced with permission from the Australian Centre for Asthma Monitoring, 2003.1
2 Death rates (95% CIs) for asthma by age group and sex in Australia, 1997 to 2001*
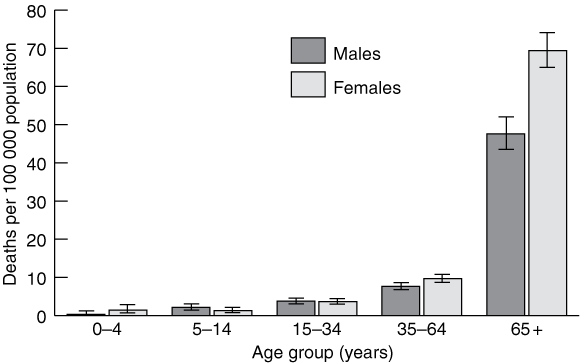
* Data from the Australian Institute of Health and Welfare National Mortality database. Reproduced with permission from the Australian Centre for Asthma Monitoring, 2003.1
3 Seasonal variation in asthma mortality*
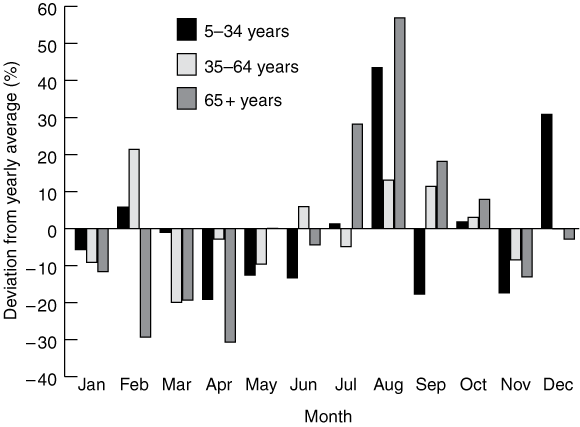
* Shown as average monthly deviation from the annual average number of deaths attributed to asthma, by age group, Australia, 1979 to 2001. Data from the Australian Institute of Health and Welfare National Mortality database. Reproduced with permission from the Australian Centre for Asthma Monitoring, 2003.1
4 Hospital care for asthma*
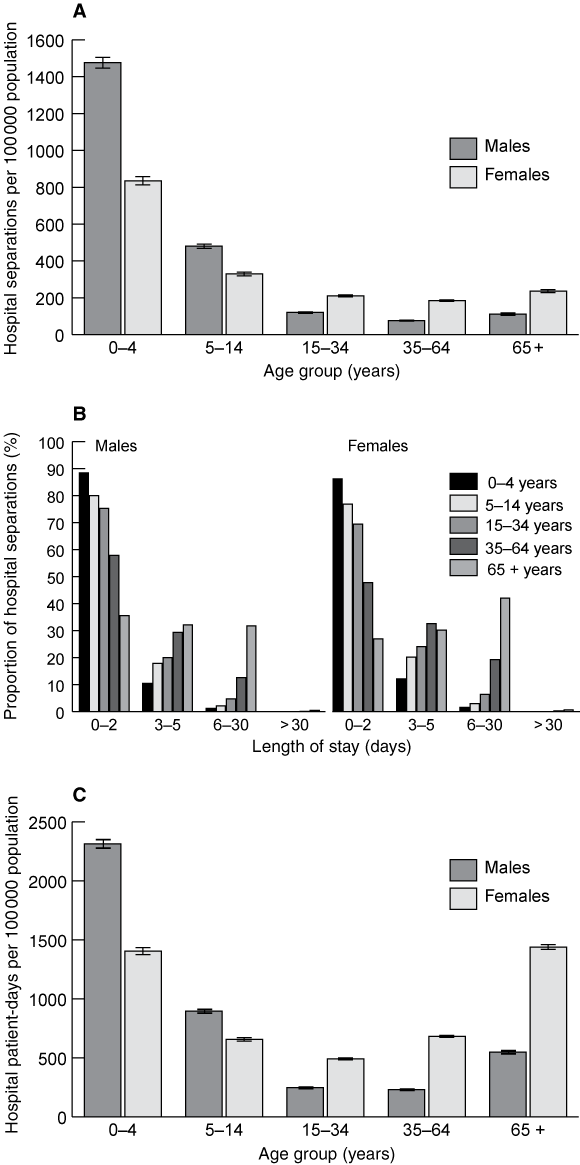
(A) Hospital separation rate (95% CIs) for asthma, by age group and sex, Australia, 2000–01.
(B) Distribution of length of hospital stay, by age group and sex, Australia, 2000–01.
(C) Hospital patient-days for asthma (95% CIs) by age group and sex, Australia, 2000–01.
* Data from the Australian Institute of Health and Welfare National Hospital Morbidity dataset. Reproduced with permission from the Australian Centre for Asthma Monitoring, 2003.1
- Guy B Marks1
- Leanne M Poulos BMedSc(Hons)2
- Australian Centre for Asthma Monitoring,* Woolcock Institute of Medical Research, Sydney, NSW.
The Woolcock Institute of Medical Research receives grants from GlaxoSmithKline, AstraZeneca and Boehringer Ingelheim, and G B M’s research group receives an allocation of this funding to support research studies in asthma and related diseases. The Woolcock Institute has also received payments from GlaxoSmithKline and AstraZeneca for seminars given by G B M and for advisory boards in which G B M has participated. Some of G B M’s research is funded by the CRC for Asthma, which includes GlaxoSmithKline and AstraZeneca as partners. G B M has undertaken contract research (data analysis) on behalf of GlaxoSmithKline and has also provided reports for environmental impact statements for SinclairKnightMertz and PPK Environment and Infrastructure, acting on behalf of government proponents of infrastructure.
- 1. Australian Centre for Asthma Monitoring. Asthma in Australia 2003. Canberra: Australian Institute of Health and Welfare, 2003. (AIHW Cat. No. ACM1.)
- 2. National Asthma Education and Prevention Program. Expert panel report 2. Guidelines for the diagnosis and management of asthma. Bethesda, MD: National Institutes of Health; National Heart, Lung, and Blood Institute, 1997.
- 3. James A, Pare P, Hogg J. The mechanics of airway narrowing in asthma. Am Rev Respir Dis 1989; 139: 242-246.
- 4. Kennedy S, Burrows B, Vedal S, et al. Methacholine responsiveness among working populations. Relationship to smoking and airway caliber. Am Rev Respir Dis 1990; 142: 1377-1383.
- 5. Peat J, Woolcock A, Cullen K. Rate of decline of lung function in subjects with asthma. Eur J Respir Dis 1987; 70: 171-179.
- 6. Australian Bureau of Statistics. National health survey: summary of results. Canberra: ABS, 2002. (ABS Cat. No. 4364.0.)
- 7. Australian Bureau of Statistics. National health survey: Aboriginal and Torres Strait Islander results, Australia. Canberra: ABS, 2002. (ABS Cat. No. 4715.0.)
- 8. Guite H, Burney P. Accuracy of recording of deaths from asthma in the UK: the false negative rate. Thorax 1996; 51: 924-928.
- 9. Jones K, Berrill W, Bromly C, Hendrick D. A confidential enquiry into certified asthma deaths in the north of England, 1994–96: influence of co-morbidity and diagnostic inaccuracy. Respir Med 1999; 93: 923-927.
- 10. Sears M, Rea H, de Boer G, et al. Accuracy of certification of deaths due to asthma. A national study. Am J Epidemiol 1986; 124: 1004-1011.
- 11. Ebi-Kryston KL. Respiratory symptoms and pulmonary function as predictors of 10-year mortality from respiratory disease, cardiovascular disease, and all causes in the Whitehall Study. J Clin Epidemiol 1988; 41: 251-260.
- 12. Strachan DP. Ventilatory function as a predictor of fatal stroke. BMJ 1991; 302: 84-87.
- 13. Phelan P, Robertson C, Olinsky A. The Melbourne Asthma Study: 1964–1999. J Allergy Clin Immunol 2002; 109: 189-194.





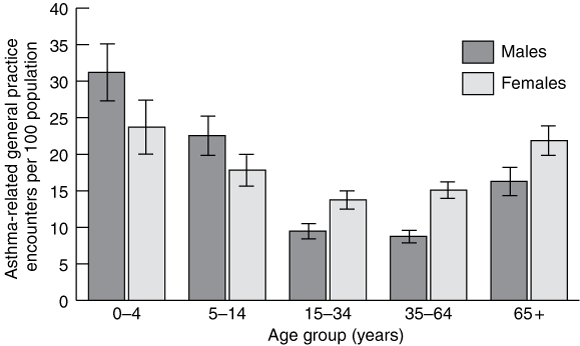
Abstract
What we need to know
What is the prevalence of impaired lung function among older people in Australia?
How can we differentiate between asthma and other causes of breathlessness in population-based health surveys of older people?
What are the effects of ageing on people with asthma?
What we need to do
Improve existing survey measures and accuracy of diagnosis.
Incorporate measures of spirometric function into health surveys of representative samples of the general population.
Use spirometry in the diagnostic work-up of older patients with dyspnoea.