Clinical record
A 53-year-old woman with a 10-year history of coeliac disease controlled by a gluten-free diet presented with a 6-month history of dramatic weight loss and pyrexia. Physical examination showed wasting and peripheral oedema. Plasma albumin level was 23 g/L (normal, 34–48 g/L). Computed tomography of head, neck, chest, abdomen and pelvis, gallium and positron emission tomography scans, liver biopsy and bone marrow biopsy were normal. The serum lactate dehydrogenase (LDH) level was 466 U/L (normal, 110–230 U/L), raising the possibility of occult malignancy, especially enteropathy-associated T-cell lymphoma (EATL). Endoscopic duodenal biopsy revealed mild increase in intraepithelial lymphocytes, crypt hyperplasia and villous abnormality in keeping with coeliac disease. Molecular clonality studies of the lymphoid cells in the duodenal biopsy yielded a monoclonal band of T-cell receptor (TCR) γ gene rearrangement in two separate series of biopsies taken 6 weeks apart.
Refractory sprue was diagnosed, and the patient received sequential trials of prednisolone, cyclosporin A and infliximab without benefit. High dose cyclophosphamide (2 g/m2) was administered 3 months later for presumed EATL. This resulted in immediate resolution of the fever and improved weight gain.
The patient subsequently complained of paraesthesiae in her feet. Neurological examination was normal. Nerve conduction studies suggested an early axonal sensory peripheral neuropathy. Vitamin B12, folate, vitamin A-tocopherol, red blood cell transaminase and transketolase studies, vasculitic screen, glycated haemoglobin, syphilis serology and thyroid function tests were normal. Paraprotein was not detected in serum electrophoresis. Magnetic resonance imaging (MRI) of the brain showed non-specific patchy periventricular areas of high signal not suggestive of the suspected lymphoma. Her systemic symptoms of fever, sweats and weight loss responded to two further doses of cyclophosphamide (1 g/m2), despite worsening neurological symptoms.
After 3 months, the patient relapsed with recurrence of pyrexia, and the serum LDH level progressively rose to 838 U/L. Neuropathy progressed to the point where she could not walk. Reflexes were lost, with a glove-and-stocking pattern of sensorimotor disturbance. A repeated nerve conduction study confirmed severe axonal sensorimotor peripheral neuropathy affecting both upper and lower limbs. Cerebrospinal fluid was normal, and there were no malignant cells. Repeated investigations, including brain MRI and upper gastrointestinal endoscopy, did not reveal any further evidence of malignancy.
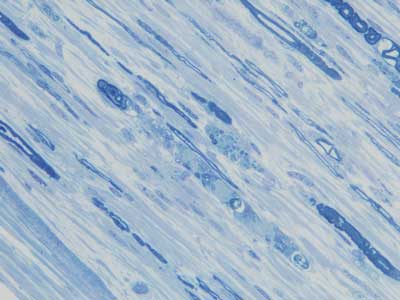
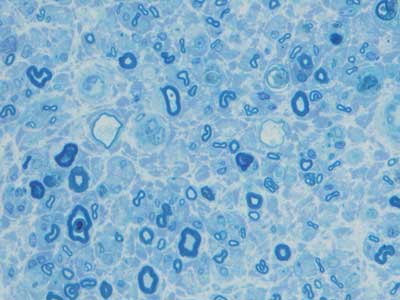
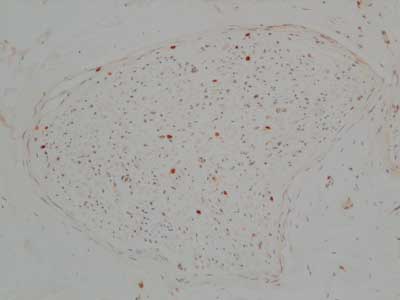
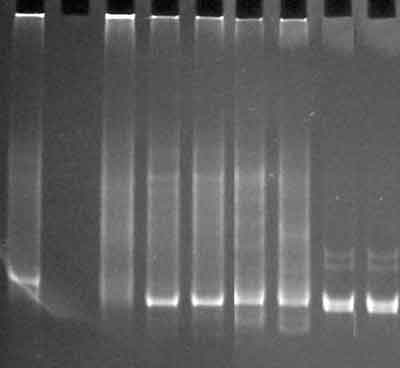
A right sural nerve biopsy showed severe loss of myelinated nerve fibres, and many of the surviving fibres showed varying stages of axonal degeneration (Box 1) associated with a sparse, patchy endoneurial mononuclear cell infiltrate in some fascicles and prominent endoneurial fibrosis on trichrome stain. The CD45RB immunostain showed increased numbers of immunopositive endoneurial lymphoid cells that showed immunopositivity with the T-cell markers CD3 (Box 2) and CD5. The CD68 immunostain showed increased endoneurial macrophages, and ultrastructural assessment confirmed macrophage phagocytosis of degenerate myelinated fibres. CD20 immunostain was negative. The Congo red stain for amyloid was negative. A TCRγ clonality study of the nerve biopsy showed a monoclonal band identical to the previous band detected in the duodenal biopsies, confirming infiltration of the nerve by T-cell lymphoma (Box 3).
Subsequent treatment with cyclophosphamide, doxorubicin and dexamethasone was complicated by Enterococcus faecalis septicaemia and florid disseminated intravascular coagulation, leading to the patient’s death.
Coeliac disease is associated with an increased risk of malignancy.1 Most of these tumours are T-cell lymphomas, in contrast to the common sporadic B-cell lymphomas that usually occur in the gastrointestinal tract.
Enteropathy-associated T-cell lymphoma (EATL) often presents as refractory sprue. The risk of EATL usually decreases to a low level after adhering to a gluten-free diet for 5 years. Common symptoms of presentation include diarrhoea, abdominal pain and weight loss. Patients may also present with acute bleeding, perforation or obstruction.2 Our patient presented with weight loss, pyrexia and, subsequently, neuropathy. Clinical deterioration despite compliance with a gluten-free diet should raise suspicion of EATL. Conversely, as coeliac disease may be undiagnosed at the time of presentation of the intestinal lymphoma, patients with T-cell lymphoma or a gut primary localisation should be tested for coeliac disease. Endoscopy with biopsy is usually the test of choice. Laparotomy is performed in some instances, especially when patients present with an acute abdomen.
Our case illustrates a number of interesting features. First, a definitive diagnosis of EATL may be elusive, and persistence in obtaining histological confirmation is warranted. The elevated serum lactate dehydrogenase (LDH) level raised the possibility of an underlying malignancy. Second, in patients with refractory sprue, a clonal TCRγ rearrangement often precedes the onset of overt T-cell lymphoma, which constitutes an early or cryptic form of EATL.3-5 The difficulty is distinguishing antigen-dependent T cells from neoplastic T cells. Management decisions may have to be based on DNA clonality studies alone. Finally, although central nervous system involvement has been reported,6,7 to our knowledge this is the first reported case of peripheral nerve involvement in EATL. Clinical evidence of peripheral neuropathy is observed in 0.1% to 8% of patients with malignant lymphomas,8 and neurotoxic chemotherapeutic agents are frequently presumed to be the cause. Peripheral nerve involvement in T-cell lymphomas is very uncommon.8-10 It is also interesting that the infiltrative neuropathy progressed relentlessly despite an initial good response of systemic symptoms to cyclophosphamide.
Both refractory sprue and EATL carry a very poor prognosis, with a 5-year survival rate of 11%. This is related to the unresponsive malabsorption in cryptic EATL, which is often diagnosed late. Associated malabsorption and malnutrition make tolerance of chemotherapy difficult, and treatment-related complications such as gastrointestinal bleeding and perforation also worsen prognosis.2 Favourable outcomes with multi-drug therapy generally occur in patients who have minimal gastrointestinal symptoms before the diagnosis of lymphoma and can tolerate therapy.
Chemotherapy should be considered for all patients and should constitute drugs active in intermediate- and high-grade lymphoma. A variety of regimens, including CHOP (cyclophosphamide, doxorubicin, vincristine and prednisolone), have been used. The optimal cytotoxic regimen is unclear, as data are limited to uncontrolled retrospective case series. The role of salvage treatments and high-dose chemotherapy at relapse also remains to be investigated.2 Earlier diagnosis and development of more effective treatments are required to improve the outcome for these patients.
- 1. Askling J, Linet M, Gridley G, et al. Cancer incidence in a population-based cohort of individuals hospitalized with celiac disease or dermatitis herpetiformis. Gastroenterology 2002; 123: 1428-1435.
- 2. Gale J, Simmonds PD, Mead GM, et al. Enteropathy-type intestinal T-cell lymphoma: clinical features and treatment of 31 patients in a single center. J Clin Oncol 2000; 18: 795-803.
- 3. Cellier C, Delabesse E, Helmer C, et al. Refractory sprue, coeliac disease, and enteropathy-associated T-cell lymphoma. Lancet 2000; 356: 203-208.
- 4. Carbonnel F, Grollet-Bioul L, Brouet JC, et al. Are complicated forms of celiac disease cryptic T-cell lymphomas? Blood 1998; 92: 3879-3886.
- 5. Ashton-Key M, Diss TC, Pan L, et al. Molecular analysis of T-cell clonality in ulcerative jejunitis and enteropathy-associated T-cell lymphoma. Am J Pathol 1997; 151: 493-498.
- 6. Tutt ANJ, Brada M, Sampson SA. Enteropathy associated T cell lymphoma presenting as an isolated CNS lymphoma three years after diagnosis of coeliac disease: T cell receptor polymerase chain reaction studies failed to show the original enteropathy to be a clonal disorder. Gut 1997; 40: 801-803.
- 7. Shams PN, Waldman A, Dogan A, et al. Ataxia in the setting of complicated enteropathy: double jeopardy. J Neurol Neurosurg Psychiatry 2002; 72: 527-529.
- 8. Gherardi R, Gaulard P, Prost C, et al. T-cell lymphoma revealed by a peripheral neuropathy. Cancer 1986; 58: 2710-2716.
- 9. Peris K, Fargnoli M, Berardelli A, et al. Peripheral nervous system involvement in a patient with large T-cell lymphoma arising from a pre-existing mycosis fungoides. Br J Dermatol 1998; 139: 299-301.
- 10. Atiq OT, DeAngelis LM, Rosenblum M, Portlock CS. Cutaneous T-cell lymphoma presenting with diffuse lymphomatous infiltration of the peripheral nerves: response to combination chemotherapy. Am J Clin Oncol 1992; 15: 212-215.





We wish to thank Drs H Nicholson and I Pollard for referring the patient.
None identified.