Australia was declared malaria-free in 1983. However, about 250 000 Australians visit malarious countries each year,1 and annual notifications of imported malaria in Australia have varied between 466 and 951 since 1991.2 Most cases are caused by Plasmodium vivax, but a third are caused by the potentially more serious Plasmodium falciparum.3,4 There is an expanding malaria-receptive zone in northern Australia, with Anopheles farauti the vector,5 and several outbreaks of vivax malaria have occurred in northern Queensland following importation, the latest in 2002.6 Drug-resistant Plasmodium strains are found increasingly in tropical countries,7 and their introduction into northern Australia could have serious consequences.
Malaria is notifiable in each Australian state and territory, and cases are reported to the National Notifiable Diseases Surveillance Scheme (NNDSS). National data were last reported in 1998,4 but most published analyses have been confined to simple demographic variables.3,4
In Western Australia (WA), an enhanced surveillance questionnaire that provides supplementary details of the circumstances of the infection in individual patients has been used for more than a decade.8 We used these data to analyse trends in notifications of imported malaria in WA from 1990 to 2001, and to describe associated risk factors and chemoprophylaxis used.
A de-identified dataset containing all malaria notifications from January 1990 to December 2001 was extracted from the WA Notifiable Infectious Diseases Database. In addition, enhanced surveillance questionnaires were sent to each attending medical practitioner to obtain details of chemoprophylaxis, itinerary, diagnosis and outcome. These supplementary data were available for 318 cases (66%), but, as the questionnaire was revised in 1992, 1993 and 1996, information was not uniform.
The appropriateness of chemoprophylaxis was assessed by comparing the drug(s) prescribed for the countries visited with the corresponding contemporary regimen recommended by the World Health Organization (WHO) in its annual guidelines published from 19909 to 2001.10 Between 1990 and 1993, when no specific regimens were recommended by WHO, chemotherapeutic appropriateness was assessed from regional parasite drug resistance patterns (Box 1).9,11,12
Data were analysed using SPSS for Windows13 and are presented as percentages and means (95% CI), or, for non-normally distributed variables, as medians (interquartile range). Percentages for specific variables were calculated using the number of cases for which the variable was known as denominator. Two-sample comparisons were assessed using Student’s t test or the Mann–Whitney U test. Differences between two proportions were analysed by Fisher’s exact test. Temporal trends in annual notifications of falciparum and vivax malaria were assessed using linear regression analysis.
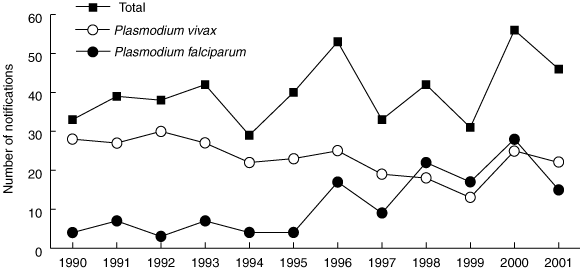
A total of 482 cases were notified, ranging from 29 to 56 per year (Box 2). Mean age was 31 years (95% CI, 11–55 years), and 80% were male. Comparison of the 6-year periods 1990–1995 (n = 221) and 1996–2001 (n = 261) showed an increase in notifications of people born in the Asia–Pacific region (from 35 [19%] to 66 [27%]; P = 0.05), a fall in those born in Australia, New Zealand or Europe (from 135 [73%] to 125 [52%]; P < 0.001), and an increase in immigrants in detention (5 [2%] v 64 [25%]; P < 0.001) and defence force personnel (5 [4%] v 18 [9%]; P = 0.048). Patients in detention were younger than those living in urban or rural areas (mean [95% CI], 25 [10–40] v 33 [10–58] and 31 [12–49] years, respectively; P ≤ 0.001).
Most infections were with P. vivax (Box 2). However, there was a significant increase in the number of P. falciparum cases between 1990 and 2001 (P < 0.001) and a decrease in P. vivax cases (P < 0.001; Box 3). When categorised by 6-year period, the number and percentage of P. falciparum cases rose from 29 (14%) to 108 (44%; P < 0.001), while P. vivax notifications fell from 157 (77%) to 122 (50%; P < 0.001).
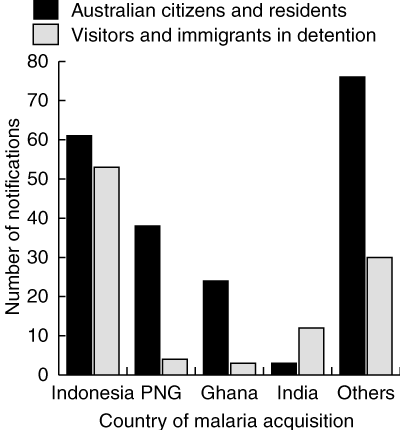
Malaria was acquired mostly in the AsiaPacific region (Box 2). However, cases from Africa increased significantly between 1990–1995 and 1996–2001, from 34 (21%) to 62 (30%; P = 0.04). As a single infecting species, P. falciparum was more frequent than P. vivax in patients infected in Africa (59 [64%] v 23 [25%]; P < 0.001), in contrast to those infected in the Asia-Pacific region (53 [21%] v 190 [75%]; P < 0.001). The most common country of malaria acquisition was the Indonesian archipelago, and most notified visitors and immigrants in detention were from Indonesia (Box 4).
Of the 283 patients (59%) with information on symptoms, 63% became unwell overseas, and 35% in Australia, while 3% were asymptomatic when diagnosed. The time from symptom onset to diagnosis ranged from 0 to 46 weeks (median, 3 days) and did not differ significantly between vivax and falciparum malaria or between 6-year periods. Immigrants in detention had the shortest time to diagnosis, with a median (interquartile range) of 0 (0–5) days versus 2 (1–11) days for rural cases (P = 0.08) and 4 (1–11) days for metropolitan cases (P = 0.009).
Details of chemoprophylaxis are shown in Box 2. Almost half the patients reported taking no antimalarial prophylaxis. Australians were more likely than non-Australian residents to have started chemoprophylaxis (63% v 23%; P < 0.001) and to have taken it regularly (39% v 14%; P < 0.001). Use of prophylaxis was similar in people on holiday (67%) and those on business (58%), and ranged from 3% in the 30 residents from malarious countries to 100% in the 12 military personnel.
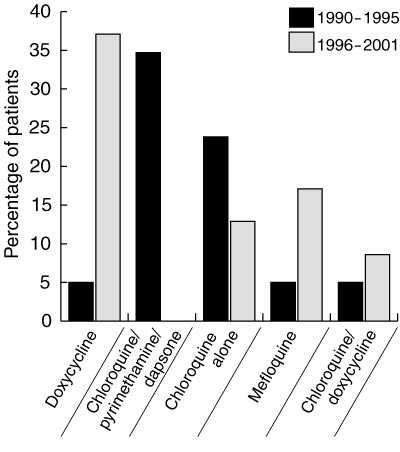
Use of doxycycline and mefloquine increased markedly between 1990–1995 and 1996–2001 (Box 5). Chloroquine alone was less popular in 1996–2001, while use of chloroquine/pyrimethamine/dapsone ceased.
In 131 patients (27%), the value of antimalarial chemoprophylaxis could be assessed by the region or country visited and WHO recommendations or reports of local parasite resistance. Of these 131, 25% were taking appropriate chemoprophylaxis, 47% were prescribed regimens that were not recommended or likely to be ineffective, and 28% used regimens that were only partially consistent with recommendations. Twenty-three individuals (18%) reported full compliance with the most appropriate chemoprophylaxis, yet still acquired malaria.
Of the 198 cases (41%) with outcome data, 84% were assessed as cured by the notifying doctor, with the remainder either referred for further assessment or awaiting test results. Hospitalisation data were available for 99 patients in whom Plasmodium species was recorded. Almost all of these were before 1996, when details of hospitalisations stopped being requested. Patients infected with P. falciparum were significantly more likely to be hospitalised than those with P. vivax (86% v 53%; P = 0.04). One patient died of severe falciparum malaria, in 1995. This patient acquired malaria in Kenya and was the first person to receive an artemisinin derivative in Australia.14
These data reveal a rise in cases of falciparum malaria reported in WA since 1990. This increase reflects, in part, the influx of immigrants in detention in WA who arrived via Indonesia in the latter part of the study period,15 as well as deployment of Australian military personnel to East Timor. P. falciparum infections comprise up to a half of all blood-slide-positive cases in Indonesia,16 and two-thirds of cases of malaria in Australian troops returning from East Timor.17 However, our data also show that increasing numbers of Australians acquire falciparum malaria in Africa, a trend also reported recently in the United Kingdom18 and the United States.19 Consistent with the increase in drug-resistant P. falciparum in most tropical regions,7 some individuals contracted malaria despite completing a course of appropriate chemoprophylaxis, emphasising the need for vector avoidance as well as use of chemoprophylaxis.
We had no information on mosquito avoidance strategies. Where chemoprophylaxis use was known, the regimen was consistent with WHO recommendations in only a quarter of cases. This finding parallels data from US citizens diagnosed with imported malaria in 2002, a group in which only 20.9% took medication recommended for the area(s) visited.19
These observations raise questions about the availability of up-to-date information for medical practitioners in the developed world, where malaria is an uncommon, imported disease. Nevertheless, mefloquine use increased over the period of our study, while chloroquine use declined, consistent with WHO guidelines (Box 1) and national trends in antimalarial drug prescribing.20 This suggests that awareness of chloroquine resistance in P. falciparum and perhaps P. vivax21 is growing. However, the greater rise in use of doxycycline compared with mefloquine is at odds with WHO policy up to 2001,10 with doxycycline recommended for areas with mefloquine-resistant P. falciparum or in mefloquine-intolerant individuals. It remains to be established whether newer approaches, including daily atovaquone–proguanil and stand-by treatment10 (including artemisinin-based therapy22) will reduce the incidence of imported malaria.
As highlighted in previous brief reports,23,24 the percentage of notifications in immigrants in detention increased over the later years of the study. This percentage may fall with the reduction in the number of “boat people” arriving in Australia since 2002.15 Perhaps because of close surveillance, routine screening and increased awareness of the possibility of tropical infections in this group, they were diagnosed more rapidly than others.
Excluding immigrants in detention, one in three of our notified patients were resident in rural WA when diagnosed. Diagnosis did not appear delayed in rural patients, suggesting that a high index of clinical suspicion and greater familiarity with the patient can offset a lack of local laboratory facilities. Given the potentially serious nature of the infection, our data provide some evidence that most patients with falciparum malaria in WA are appropriately hospitalised for treatment. However, many rural hospitals and clinics may not stock malaria treatments (including intravenous quinine, the only parenteral therapy available in Australia for severe cases). The data suggest that this situation should be reviewed and, as potential transmission to local mosquito vectors is possible in northern WA, the gametocidal drug primaquine should also be available in centres north of latitude 19°S.5
Our study has limitations. WA laboratories are not required to report notifiable infectious diseases, and informal laboratory reporting began systematically only in 2000. The consequent reliance on medical practitioners to notify cases is likely to have led to an underestimation of cases, particularly given that the public health consequences of malaria apply only in the far north of WA. Four patients (0.01%) had more than one episode of malaria during the 12-year study period, but this is most unlikely to have influenced the results. The database did not include details of the antimalarial drugs used for treatment, including use of prima-quine, which is given after initial treatment to eliminate liver forms of vivax and ovale malaria (radical cure). Missing data and the lack of uniformity of the enhanced surveillance data over time created some difficulties of interpretation. Nevertheless, our data represent one of the most comprehensive assessments of imported malaria in Australia to date.3,4
Malaria remains an important parasitic disease in Australia. Significant numbers of Australian travellers take inadequate chemoprophylaxis, while visitors, immigrants in detention and defence force personnel are additional sources of imported malaria. Early diagnosis and effective treatment, especially in malaria-receptive areas of northern Australia, are vital and can be enhanced by increasing practitioner awareness of local experience with malaria. A national malaria register with more detailed information than that held by the NNDSS would be important in this regard. Such a register was created in 1969 but stopped collecting data in 1995. Its reactivation should be supported.
1 WHO recommendations on chemoprophylaxis for falciparum malaria in countries where most notified cases were acquired, at start9 and end10 of study period
|
1990* |
2001 |
|||||||||||||
Indonesia |
High-grade chloroquine resistance; reports of sulfadoxine–pyrimethamine resistance |
Chloroquine plus proguanil; mefloquine in Irian Jaya |
|||||||||||||
Papua New Guinea |
High-grade chloroquine resistance; reports of sulfadoxine–pyrimethamine resistance |
Mefloquine |
|||||||||||||
Ghana |
Chloroquine resistance reported |
Mefloquine |
|||||||||||||
India |
High-grade chloroquine resistance |
Chloroquine plus proguanil |
|||||||||||||
WHO = World Health Organization. * In 1990, WHO made only statements about reported parasite resistance patterns. Current WHO recommendations are available at www.who.int/ith. |
|||||||||||||||
2 Characteristics of 482 patients notified with malaria in Western Australia, 1990–2001
Characteristic |
Number of patients |
||||||||||||||
Country of birth (n = 427; 89%) |
|||||||||||||||
Australia/New Zealand |
222 (52%) |
||||||||||||||
Asia |
101 (24%) |
||||||||||||||
Europe |
38 (9%) |
||||||||||||||
Middle East |
35 (8%) |
||||||||||||||
Other |
31 (7%) |
||||||||||||||
Place of residence in WA (n = 472; 98%) |
|||||||||||||||
Perth metropolitan area |
271 (57%) |
||||||||||||||
Country area |
146 (31%) |
||||||||||||||
Immigration detention centre* |
55 (12%) |
||||||||||||||
Occupation (n = 335; 70%) |
|
||||||||||||||
White/blue collar worker |
129 (39%) |
||||||||||||||
Visitor to Australia |
95 (28%) |
||||||||||||||
Student |
34 (10%) |
||||||||||||||
Other |
77 (23%) |
||||||||||||||
Past history of malaria (n = 172; 36%) |
|||||||||||||||
Plasmodium vivax |
124 (72%) |
||||||||||||||
Other |
48 (28%) |
||||||||||||||
Use of chemoprophylaxis (n = 243; 50%) |
|||||||||||||||
Regular |
75 (31%) |
||||||||||||||
Irregular/incomplete |
48 (20%) |
||||||||||||||
None |
120 (49%) |
||||||||||||||
Chemoprophylaxis (n = 150; 31%) |
|||||||||||||||
Doxycycline |
30 (20%) |
||||||||||||||
Chloroquine/pyrimethamine/ dapsone |
28 (19%) |
||||||||||||||
Chloroquine alone |
28 (19%) |
||||||||||||||
Mefloquine |
16 (11%) |
||||||||||||||
Chloroquine/doxycycline |
10 (7%) |
||||||||||||||
Other |
38 (25%) |
||||||||||||||
Region of acquisition (n = 370; 77%) |
|||||||||||||||
Asia–Pacific |
259 (70%) |
||||||||||||||
Indonesia |
130 (35%) |
||||||||||||||
Papua New Guinea |
59 (16%) |
||||||||||||||
India |
18 (5%) |
||||||||||||||
Other |
52 (14%) |
||||||||||||||
Africa |
96 (26%) |
||||||||||||||
Ghana |
34 (9%) |
||||||||||||||
Other |
62 (17%) |
||||||||||||||
Other region |
15 (4%) |
||||||||||||||
Plasmodial species (n = 482; 100%) |
|||||||||||||||
Plasmodium vivax |
279 (58%) |
||||||||||||||
Plasmodium falciparum |
137 (28%) |
||||||||||||||
Plasmodium malariae |
7 (2%) |
||||||||||||||
Plasmodium ovale |
3 (1%) |
||||||||||||||
Mixed |
24 (5%) |
||||||||||||||
Unspecified |
32 (7%) |
||||||||||||||
* Curtin Immigration and Processing Centre in Derby or Broome Prison. |
|||||||||||||||
- Donnetta M Charles1
- Julie Hart2
- Wendy A Davis3
- Timothy M E Davis4
- Eleanor Sullivan5
- Gary K Dowse6
- 1 School of Medicine and Pharmacology, University of Western Australia, Fremantle, WA.
- 2 Communicable Disease Control Directorate, Western Australian Department of Health, Perth, WA.
We thank the Western Australian Department of Health for access to the malaria database.
None identified.
- 1. Prociv P. Deaths of Australians overseas. Med J Aust 1995; 163: 27-30.
- 2. Australian Government Department of Health and Ageing. Australia's notifiable diseases status: annual report of the National Notifiable Diseases Surveillance System. Available at: www.cda.gov.au./pubs/annlrpt/nndssar.htm (accessed Oct 2004).
- 3. Longbottom H. Epidemiology of malaria in Australia 1991-1995. Comm Dis Intell 1996; 20: 84-97.
- 4. Harvey B. Trends in malaria in Australia, 1991-1997. Commun Dis Intell 1998; 22: 247-248.
- 5. Bryan JH, Foley DH, Sutherst RW. Malaria transmission and climate change in Australia. Med J Aust 1996; 164: 345-347. <eMJA full text>
- 6. Hanna JN, Ritchie SA, Eisen DP, et al. An outbreak of Plasmodium vivax malaria in Far North Queensland, 2002. Med J Aust 2004; 180: 24-28. <MJA full text>
- 7. Wongsrichanalai C, Pickard AL, Wernsdorfer WH, Meshnick SR. Epidemiology of drug-resistant malaria. Lancet Infect Dis 2002; 2: 209-218.
- 8. Ashwell MJS, Rouse IL. A review of notified cases of malaria in Western Australia, 1988-1992. Epidemiology Branch, Health Information Centre, Department of Health. Health Topics 1996; 3: 1-8.
- 9. World Health Organization. International travel and health. Vaccination requirements and health advice — situation as on 1 January 1990. Geneva: WHO, 1990: 61-68.
- 10. World Health Organization. International travel and health. Vaccination requirements and health advice — situation as on 1 January 2001. Geneva: WHO, 2001: 67-84.
- 11. World Health Organization. International travel and health. Vaccination requirements and health advice — situation as on 1 January 1991. Geneva: WHO, 1991: 61-74.
- 12. World Health Organization. International travel and health. Vaccination requirements and health advice — situation as on 1 January 1992. Geneva: WHO, 1992: 63-73.
- 13. Statistical Package for the Social Sciences Inc. SPSS for Windows. Version 10.0. Chicago, Ill: SPSS Inc, 1999.
- 14. Davis TME, Breheny FX, Kendall PA, et al. Severe falciparum malaria with hyperparasitaemia treated with intravenous artesunate. Med J Aust 1997; 166: 416-418.
- 15. Department of Immigration and Multicultural and Indigenous Affairs. Managing the border: Immigration compliance. Canberra: Commonwealth of Australia, 2004: 27-28.
- 16. World Health Organization Regional Office for South-East Asia. Indonesia malaria profile. Available at: w3.whosea.org/malaria/pdf/ino.pdf (accessed Jan 2003).
- 17. Kitchener SJ, Auliff AM, Rieckmann KH. Malaria in the Australian Defence Force during and after participation in the International Force in East Timor (INTERFET). Med J Aust 2000; 173: 583-585. <MJA full text>
- 18. Health Protection Agency. Illness in England, Wales, and Northern Ireland associated with foreign travel: a baseline report to 2002. Available at: www.hpa.org.uk/infections/topics_az/travel/publications.htm (accessed Oct 2004).
- 19. Shah S, Filler S, Causer LM, et al. Malaria surveillance — United States, 2002. MMWR Surveill Summ 2004; 53 (SS01): 21-34. Available at: www.cdc.gov/mmwr/preview/mmwrhtml/ss5301a2.htm (accessed Oct 2004).
- 20. Leggat PA, Speare R. Trends in antimalarial drugs prescribed in Australia 1992 to 1998. J Travel Med 2003; 10: 189-191.
- 21. Sumawinata IW, Bernadeta, Leksana B, et al. Very high risk of therapeutic failure with chloroquine for uncomplicated Plasmodium falciparum and P. vivax malaria in Indonesian Papua. Am J Trop Med Hyg 2003; 68: 416-420.
- 22. Bradley DJ, Bannister B. Health Protection Agency Advisory Committee on Malaria Prevention for UK Travellers. Guidelines for malaria prevention in travellers from the United Kingdom for 2003. Commun Dis Public Health 2003; 6: 180-199.
- 23. Dowse G, Thomas R. Malaria in illegal entrants — Western Australia. Commun Dis Intell 2000; 24: 394.
- 24. Skull SA, Tallis G. Epidemiology of malaria in Victoria 1999-2000: East Timor emerges as a new source of disease. Commun Dis Intell 2001; 25: 149-151.





Abstract
Objective: To assess changes in and factors associated with recent malaria notifications in Western Australia (WA).
Design: Retrospective analysis of the WA Notifiable Infectious Diseases Database and enhanced surveillance questionnaires completed by attending medical practitioners.
Patients: Cases of malaria notified between January 1990 and December 2001.
Main outcome measures: Annual notifications by demographic variables (including age, sex, occupation and place of residence), region/country of acquisition, chemoprophylaxis used, Plasmodium species and outcome.
Results: 482 patients were notified (mean age, 31 years; 80% male); 57% lived in Perth, 31% in country areas and 12% in an immigration detention centre. Comparison between the 6-year periods 1990–1995 and 1996–2001 showed that Plasmodium falciparum cases increased from 29 (14%) to 108 (44%; P < 0.001), while Plasmodium vivax cases decreased from 157 (77%) to 122 (50%; P < 0.001); immigrants in detention, defence force personnel and cases from Africa were increasingly represented (P < 0.05 in each case). Only 31% of patients took regular chemoprophylaxis and, among these, the regimen was appropriate in only a quarter. There was a median period of 3 days between symptom onset and diagnosis. One patient died.
Conclusions: There has been an increase in P. falciparum cases in WA since 1990. This reflects the influx of immigrants in detention, deployment of military personnel to East Timor and increasing numbers of cases from Africa. A significant number of Australian travellers who developed malaria had not taken chemoprophylaxis either regularly or at all, and, of those who had, the regimen was inadequate in most.