Hepatitis A was a major public health problem in north Queensland during the 1990s.1 Two prolonged, extensive community-wide epidemics occurred during the decade (Box 1) and led to many occupational exposures to the hepatitis A virus, and several nosocomial outbreaks in this area (which has a southern boundary approximately defined by a line between Mackay and Mt Isa).2-4
Outbreaks of hepatitis A associated with child daycare centres were also prominent during that decade. For example, 18 such outbreaks were recognised over the 2 years 1998–1999, with infections in a total of 33 children, 62 of their household contacts and 10 staff members.5 In response to these outbreaks, normal human immunoglobulin was administered to 928 children and 105 staff members.5
Of even greater concern was the disproportionate number of notifications of hepatitis A in Indigenous people in north Queensland, particularly young Indigenous children. The average annual notification rate in the region in 1996–1997 in Indigenous people was about 69 cases per 100 000 persons overall, and 134 per 100 000 in Indigenous children aged under 5 years (compared with 5 per 100 000 in non-Indigenous children of the same age).1 Furthermore, three Indigenous children aged under 5 years died of fulminant hepatitis A between 1993 and 1998.6
In response, the paediatric formulation of an inactivated hepatitis A vaccine has been made freely available to Indigenous children in north Queensland from February 1999. Two doses of the vaccine, at 18 months and 2 years of age, were recommended, with catch-up vaccination (two doses, six months apart) recommended up to the sixth birthday.6 The vaccine was also provided free to non-Indigenous children of these ages who resided in rural and remote Indigenous communities in north Queensland. The introduction of the vaccine was rolled out progressively throughout the region over the year, so that by the end of 1999 most clinics had incorporated it into routine childhood immunisation activities.
This report examines the impact of this vaccination on notifications of hepatitis A throughout north Queensland in the first four years (2000–2003) after the introduction of the program.
Hepatitis A is a notifiable disease in Queensland; laboratories throughout the state are required to notify public health units when hepatitis A IgM antibodies are detected in a serum sample. “Enhanced” surveillance of the disease has been undertaken in north Queensland since 1996. This involves public health personnel interviewing each patient or his/her guardian and, using a standardised questionnaire, ascertaining each patient’s Indigenous status, potential exposures to known risk factors for the infection, and the potential for further transmission from each case. This information not only informs immediate public health responses, but is also collated and reviewed as necessary.1,5
Cases were included in this report regardless of where the infection was acquired, provided that each case was considered to have been infectious, and therefore requiring further public health measures, while in north Queensland.
Hepatitis A incidence and vaccine uptake were calculated using population denominators from the Australian Bureau of Statistics 2001 census.7 In 2001, there were about 596 500 people in north Queensland, including about 43 500 children aged under 5 years. Nearly 6900 of the latter (16%) were Indigenous. Nearly 1380 Indigenous children were born in north Queensland in 2000.
Ethical approval was not necessary for this study, as the vaccine was registered for use in Australia, and as surveillance of communicable diseases of public health importance is required by law.
In the 4 years 1996–1999, 787 cases of hepatitis A were notified in north Queensland, 237 (30%) of which were in Indigenous people (Box 1). The average annual notification rates in Indigenous and non-Indigenous people during this period were 110 and 25 cases per 100 000 persons, respectively, and 149 per 100 000 for Indigenous children aged under 5 years (Box 2).
However, in the first 4 years after the introduction of the (2000–2003) vaccination program, 66 cases of hepatitis A were notified in north Queensland. Only nine of these (14%) were in Indigenous people (Box 3). The average annual notification rates in Indigenous and non-Indigenous people in this period were 4 and 2.5 cases per 100 000 persons, respectively, and 3.5 per 100 000 for Indigenous children aged under 5 years.
Of the 66 cases in the period 2000–2003, 34 (52%) occurred in 2000; thereafter, the average was about 10 cases per year. Twenty-two of the cases (33%) were not locally acquired: 14 were acquired overseas (eight in Papua New Guinea), seven interstate (five in the Northern Territory) and one elsewhere in Queensland. Only one child daycare centre outbreak (involving five cases) occurred during the 4-year period — in the year 2000.
Of the nine Indigenous cases in the period 2000–2003, eight occurred in 2000. Only two of these were in children; despite being age-eligible for the free hepatitis A vaccine, neither had been vaccinated. The last locally acquired Indigenous case was in June 2000, with no further Indigenous cases occurring over the next 3 years. However, in August 2003, hepatitis A was diagnosed in a 49-year-old urban Indigenous woman who had acquired the infection while undertaking Indigenous studies in the Northern Territory.
There was a rapid and marked decline in notifications of hepatitis A after the roll-out of the vaccination program. The total number of cases in 2000–2003 was a 12-fold reduction from the 1996–1999 total. This decline was particularly marked in Indigenous people (Box 3), which is consistent with the apparent very high vaccine uptake in Indigenous children. This decline, together with the lack of cases in vaccinated children, attests to the very high efficacy of hepatitis A vaccines, previously determined by randomised clinical trials.8,9
The last case of locally acquired hepatitis A in an Indigenous person occurred 18 months after the beginning of the program. This suggests that the vaccine uptake and effectiveness were sufficient to enable the apparent eradication of hepatitis A virus (HAV) from Indigenous communities in north Queensland within a very short period of time. A similar rapid and virtually complete disappearance of hepatitis A after the introduction of a vaccination program has recently been reported in American Indian and Alaska Native communities in the United States.10
However, there was also a very marked decline in the notification rate of hepatitis A in non-Indigenous people in north Queensland (Box 3). This is extremely unlikely to have been a direct vaccine effect, as only 51 non-Indigenous children born in 2000 were recorded on VIVAS as having received hepatitis A vaccine. Yet, it is quite possibly an indirect effect of the vaccination program.
Young children with hepatitis A are a particularly important source of HAV for the further transmission of the virus.11,12 As they usually have no symptoms, they are not recognised as potentially infectious, and, as they are not fully toilet-trained, other people are exposed to their faeces. For these reasons child daycare centres provide an ideal environment for HAV transmission; day-care centre outbreaks are also of concern, as HAV can spread rapidly from daycare centres into the general community.5,13
We believe that, before the implementation of the hepatitis A vaccination program in north Queensland, young Indigenous children were a particularly important source of HAV, contributing to transmission not only within extended family networks in their communities, but also within daycare centres in the main urban centres. These networks and centres became, in turn, sources of HAV transmission to the general community. Vaccinating Indigenous children broke these chains of transmission, leading to the cessation of transmission of HAV to the broader community and thereby an abrupt decline in the incidence of hepatitis A, not only in susceptible Indigenous, but also non-Indigenous, contacts.
As further evidence for the reduction in local transmission of HAV, there was an apparent increase in the proportion of cases that were acquired outside north Queensland. Before the program, about 10% of cases were acquired elsewhere,1 compared with 33% in 2000–2003. Papua New Guinea and the Northern Territory were prominent sources for these non-locally acquired cases.
There are three possible explanations as to why 104% of the estimated 2000 Indigenous birth cohort received at least one dose of hepatitis A vaccine. Firstly, there may have been underenumeration of Indigenous children in the 2001 national census. Secondly, some of the doses may have been given to non-Indigenous children whose ethnicity was incorrectly recorded on VIVAS. Thirdly, it is probable that a number of Indigenous children born outside north Queensland moved into the region and became eligible for the vaccine. Conversely, it is probable that some Indigenous children moved out of the region some time after receiving the first dose of vaccine, and were therefore no longer eligible for the second dose of the vaccine. This could explain in part why 27% of the children had not received a second dose.
Although paediatric hepatitis A vaccines are registered in Australia for use in children from 2 years of age,14 the vaccine was recommended to be given at 18 and 24 months of age. This was mainly so that it could be conveniently integrated into the existing childhood vaccination schedule, with only one extra visit to a vaccine provider required, at 24 months of age. However, there is a growing body of evidence that the paediatric hepatitis A vaccines can be safely administered to children aged under 2 years.15-17 Furthermore, the same schedule has been in use in Israel since 1999.18
Although the north Queensland program is the first occasion that the hepatitis A vaccine has been used widely in children in Australia, there are international precedents.19 In the United States, the vaccine has been recommended for American Indian and Alaska Native children since the mid-1990s,10,20 and for all children in states, counties and communities with rates at least twice the 1987–1997 national average (ie, ≥ 20 per 100 000 population) since the late 1990s.21 In Puglia, Italy, a first dose of the vaccine has been offered to all children (with the second dose in adolescence) since 1997, and it has been offered to all children in Israel since 1999.18 All these programs have been followed by a rapid and profound decline in notifications of hepatitis A.18,22,23
However, the north Queensland program is perhaps unique in that it is the only program that we are aware of that targeted a high-risk population of children within a defined community. This program provides evidence that an effective targeted program can reduce the notification rate of hepatitis A within a particular region, not only in those who were vaccinated but possibly also in those who were not.
The north Queensland program has recently been endorsed by the National Health and Medical Research Council.14 It now appears very unlikely that vaccinated children will require booster doses in the future.24 We agree that consideration of hepatitis A vaccination options for Australia is now timely.19 We believe that there is a compelling case for expanding the north Queensland program to other high-risk Indigenous children, such as those residing elsewhere in northern Australia. Fatal cases of hepatitis A in Aboriginal children have recently been reported in north Western Australia.25
1 Hepatitis A in north Queensland, 1991–2003
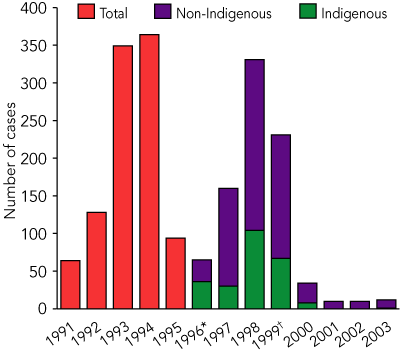
* Enhanced surveillance with systematic collection of Indigenous status began in 1996. † The vaccination program began in February 1999, and was rolled out progressively over that year.
2 Average annual notification rates of hepatitis A in north Queensland before and after implementation of the vaccination program*
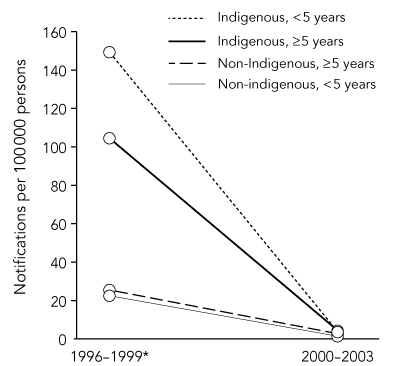
* The vaccination program was implemented from February 1999.
3 Cases of hepatitis A in north Queensland before and after implementation of the hepatitis A vaccination program for Indigenous children
|
Before program (1996–1999) |
After program (2000–2003) |
|||||||||||||
|
< 5 years |
≥ 5 years |
Total |
< 5 years |
≥ 5 years |
Total |
|||||||||
Indigenous |
41 |
196 |
237 |
1 |
8 |
9 |
|||||||||
Non-Indigenous |
33 |
517 |
550 |
2 |
55 |
57 |
|||||||||
Total |
74 |
713 |
787 |
3 |
63 |
66 |
|||||||||
- Jeffrey N Hanna1
- Susan L Hills2
- Jan L Humphreys3
- 1 Tropical Public Health Unit Network, Queensland Health, Cairns, QLD.
- 2 Tropical Public Health Unit Network, Queensland Health, Aitkenvale, QLD.
We thank all those in the Communicable Diseases Unit and the Tropical Public Health Unit Network, Queensland Health, who assisted in the planning and implementation of the program. We particularly wish to thank all the vaccine service providers in north Queensland who have supported the roll out and maintenance of the program.
Dr Hanna has received funding from vaccine companies to attend conferences and workshops.
- 1. Merritt A, Symons D, Griffiths M. The epidemiology of acute hepatitis A in North Queensland, 1996-1997. Commun Dis Intell 1999; 23: 120-124.
- 2. Hanna J, Brookes D. Some occupational exposures to hepatitis A. Commun Dis Intell 1994; 18: 3-5.
- 3. Hanna J. Hospital-acquired hepatitis A. Commun Dis Intell 1994; 18: 312-313.
- 4. Hanna JN, Loewenthal MR, Negel P, Wenck DJ. An outbreak of hepatitis A in an Intensive Care Unit. Anaesth Intensive Care 1996; 24: 440-444.
- 5. Hanna JN, Humphreys JL, Hills SL, et al. Recognising and responding to outbreaks of hepatitis A associated with child day-care centres. Aust N Z J Public Health 2001; 25: 525-528.
- 6. Hanna JN, Warnock TH, Shepherd RW, Selvey LA. Fulminant hepatitis A in Indigenous children in north Queensland. Med J Aust 2000; 172: 19-21. <MJA full text>
- 7. Australian Bureau of Statistics. 2001 Census of population and housing (1st release, Aboriginal and Torres Strait Islander profile by statistical local area). ABS: Canberra, 2002. Available at: http://datahub.govnet.qld.gov.au/ausstats/absATcensus (accessed Sep 2002).
- 8. Werzberger A, Mensch B, Kuter B, et al. A controlled trial of a formalin-inactivated hepatitis A vaccine in healthy children. N Engl J Med 1992; 327: 453-457.
- 9. Innis BL, Snitbhan R, Kunasol P, et al. Protection against hepatitis A by an inactivated vaccine. JAMA 1994; 271: 1328-1334.
- 10. Bialek SR, Thoroughman DA, Hu D, et al. Hepatitis A incidence and hepatitis A vaccination among American Indians and Alaska Natives, 1990-2001. Am J Public Health 2004; 94: 996-1001.
- 11. Smith PF, Grabau JC, Werzberger A, et al. The role of young children in a community-wide outbreak of hepatitis A. Epidemiol Infect 1997; 118: 243-252.
- 12. Staes CJ, Schlenker TL, Risk I, et al. Sources of infection among persons with acute hepatitis A and no identified risk factors during a sustained community-wide outbreak. Pediatrics 2000; 106: E54.
- 13. Venczel LV, Desai MM, Vertz D, et al. The role of child care in a community-wide outbreak of hepatitis A. Pediatrics 2001; 108: E78.
- 14. National Health and Medical Research Council. Hepatitis A. In: The Australian immunisation handbook. 8th ed. Canberra: Commonwealth of Australia, 2003: 133-145.
- 15. Piazza M, Safary A, Vegnente A, et al. Safety and immunogenicity of hepatitis A vaccine in infants: a candidate for inclusion in the childhood vaccination programme. Vaccine 1999; 17: 585-588.
- 16. Dagan R, Amir J, Mijalovsky A, et al. Immunization against hepatitis A in the first year of life: priming despite the presence of maternal antibody. Pediatr Infect Dis J 2000; 19: 1045-1052.
- 17. Abarca K, Ibanez I, Flores J, et al. Efficacy of hepatitis A vaccination in children aged 12 to 24 months [erratum published in Arch Med Res 2002; 33: 205]. Arch Med Res 2001; 32: 468-472.
- 18. Dagan R, Leventhal A, Anis ER, et al. National hepatitis A virus (HAV) immunization program aimed exclusively at toddlers in an endemic country resulting in > 90% reduction in morbidity rates in all ages. Abstracts of the 40th Annual Meeting of the Infectious Diseases Society of America; Chicago; Oct 24-27 2002: 189. Available at: www.journals.uchicago.edu/CID/2002Abstracts (accessed Feb 2004).
- 19. MacIntyre CR, Burgess MA, Hull B, McIntyre PB. Hepatitis A vaccination options for Australia. J Paediatr Child Health 2003; 39: 83-87.
- 20. Welty TK, Darling K, Dye S, et al. Guidelines for prevention and control of hepatitis A in American Indian and Alaska Native communities. S D J Med 1996; 49: 317-322.
- 21. Centers for Disease Control and Prevention. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 1999; 48 (RR-12): 1-37.
- 22. McMahon BJ, Beller M, Williams J, et al. A program to control an outbreak of hepatitis A in Alaska by using an inactivated hepatitis A vaccine. Arch Pediatr Adolesc Med 1996; 150: 733-739.
- 23. Averhoff F, Shapiro CN, Bell BP, et al. Control of hepatitis A through routine vaccination of children. JAMA 2001; 286: 2968-2973.
- 24. Van Damme P, Banatvala J, Fay O, et al. Hepatitis A booster vaccination: is there a need? Lancet 2003; 362: 1065-1071.
- 25. Adams L, Johnson G. Hepatitis A virus infection, immunisation and the Kimberley. Kimberley Public Health Bull 2003; 34 (May): 13.





Abstract
Objective: To describe the impact of a hepatitis A vaccination program for Indigenous children in north Queensland.
Design: Enhanced surveillance of all notified cases of hepatitis A in north Queensland from 1996 to 2003.
Setting: North Queensland; population, 596 500 people, including about 6900 Indigenous children aged under five years.
Interventions: Hepatitis A vaccine was provided to Indigenous children in north Queensland from February 1999; two doses were recommended (at 18 months and 2 years of age), as was catch-up vaccination up to the sixth birthday.
Results: In the 4 years 1996–1999, 787 cases of hepatitis A were notified in north Queensland, 237 (30%) of which were in Indigenous people. The average annual notification rates in Indigenous and non-Indigenous people during this period were 110 and 25 cases per 100 000 persons, respectively. In the first 4 years after introduction of the vaccination program (2000–2003), 66 cases of hepatitis A were notified. Only nine of the 66 (14%) were in Indigenous people. The average annual notification rates in Indigenous and non-Indigenous people in 2000–2003 were 4 and 2.5 cases per 100 000 persons, respectively.
Conclusion: Hepatitis A seems to have been eradicated from Indigenous communities in north Queensland very soon after the vaccination program began. The rapid decline in notifications in non-Indigenous as well as Indigenous people suggests the program quickly interrupted chains of transmission from Indigenous children to the broader community. To our knowledge this is the first evidence that a hepatitis A vaccination program targeting a high-risk population within a community can reduce disease in the broader community. Hepatitis A vaccine should be provided to other high-risk Indigenous children elsewhere in Australia.