Fragility fractures of all kinds — vertebral, hip, forearm and others — are a public health problem in both sexes. In Australia, the burden of the increased morbidity and mortality costs $7 billion annually, or a dollar a day for every man, woman and child in the country.1 This burden will increase in absolute terms because of the ageing of the population. However, despite the morbidity, mortality and increased risk of further fractures, most people with fractures related to osteoporosis are not treated for the underlying condition.
Thirty per cent to 50% of women and 15%–30% of men will suffer a fracture related to osteoporosis in their lifetime.2 Fractures increase morbidity and mortality and impose a financial burden on the community.3-4 Hip and spinal fractures are often the focus of attention, but the burden of other fractures in terms of time off work, doctor visits and investigations also contributes to the high human and financial cost of fractures.4
About 20%–25% of hip fractures occur in men.5 The overall mortality is about 20% in the first 12 months after hip fracture and is higher in men than women. Spinal fractures cause morbidity and increase mortality in both sexes (Box 1).
Bone is remodelled throughout adult life by discrete remodelling units of osteoclasts (cells that resorb a volume of bone) and osteoblasts (cells that lay down new bone matrix). In adulthood and old age, the volume of bone resorbed is greater than the volume replaced in each remodelling unit. In women, the rate of remodelling and the negative bone balance increases after menopause, accelerating bone loss, trabecular thinning and loss of connectedness, cortical thinning and porosity (Box 2A). This persistently increased rate of bone removal erodes a bone of diminishing mass, causing more architectural disruption and increasing bone fragility exponentially.7 Loads on bone become relatively greater as they are distributed over a smaller area (Box 2, B and C). Men do not have a midlife increase in remodelling, so that structural integrity of trabecular bone is maintained longer. In both women and men, the imperative to intervene pharmacologically increases with age, as fracture risk increases exponentially.
Antiresorptive agents, the bisphosphonates alendronate and risedronate and the selective oestrogen-receptor modulator (SERM) raloxifene, reduce the intensity of remodelling.10 Slower remodelling permits more complete secondary mineralisation of bone tissue, restoring bone material stiffness. These drugs reduce the progression of trabecular and cortical thinning, as well as loss of trabecular connectivity, and so maintain or increase bone strength, even though they cannot reverse structural damage.
The anabolic agent parathyroid hormone (PTH) produces some reconstruction of the skeleton, with the deposition of new bone tissue on the periosteal, endocortical and trabecular surfaces thickening the cortex and trabeculae, albeit with some increase in cortical porosity adjacent to the endosteum.11 The overall effect is to increase bone strength and reduce fracture risk.
Strontium ranelate is a new agent studied with equal rigour, as have been the above agents. It maintains the rate of remodelling, but may reduce resorption while maintaining bone formation, dissociating the two processes.12 Whether it is a bone-forming agent like PTH remains to be established.
Vertebral fractures: The most rigorously studied drugs reported to reduce rates of vertebral fracture in women with osteoporosis include alendronate, risedronate, raloxifene, parathyroid hormone10 and strontium ranelate.12 Head-to-head comparator studies have not been done. Hence, it is not known whether any one of these drugs is more efficacious than another, and this cannot be inferred from meta-analyses.13 These drugs reduce the risk of symptomatic (clinical) and asymptomatic (morphometric) single vertebral fractures by about 40%–50% and multiple vertebral fractures by about 80%–90%. The benefits have been reported within the first 6–18 months of treatment.10
Hormone replacement therapy (HRT) and the bisphosphonate etidronate have also been reported to reduce vertebral fracture, but these studies are less rigorous. The level of evidence for the efficacy of calcitonin and vitamin D metabolites in reducing risk of vertebral fractures is insufficient for inferences to be made with confidence.
Hip and other non-vertebral fractures: Alendronate and risedronate have been reported to reduce the risk of hip and other non-vertebral fractures. HRT has also been reported to reduce the risk of hip and other non-vertebral fractures in the Women’s Health Initiative, a study not specifically designed to examine anti-fracture efficacy.14 Recent data also support the notion that strontium ranelate also reduces the risk of hip and other non-vertebral fractures (unpublished data). Parathyroid hormone has been reported to reduce the risk of non-vertebral fractures, but not hip fractures. Raloxifene has not been reported to reduce the risk of hip or other non-vertebral fractures, except in a post-hoc subanalysis. Calcium and vitamin D reduce the risk of hip fractures in nursing home residents, among whom suboptimal nutrition (low calcium and protein intake) and vitamin D insufficiency are common.10 Hip protectors have been reported to reduce the risk of hip fractures, but compliance with their use is poor.15
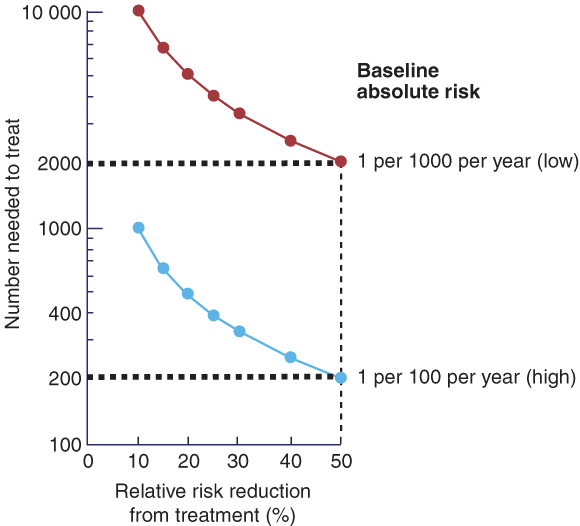
If drugs were 100% efficacious, 100% safe, and cost-free, and patients were 100% compliant, the answer would be to treat everyone and early. As this is not the case, the most important factor determining whom and when to treat is an individual’s absolute risk of fracture. If the risk is 2 per 1000 women per year, and a drug halves fracture risk, then one event is prevented, one woman will sustain a fracture despite treatment, and 998 who were not going to have a fracture anyhow had treatment. Thus, one fracture is prevented, but 999 women per year are treated without benefit. If the absolute risk is higher, say 2 per 100 women per year, and the drug still halves the risk, again one fracture is prevented, but only 99 women are treated (Box 3). Over 3 to 5 years of treatment, this equates to a number needed to treat (NNT) to prevent a fracture of 20–25 women, a figure commonly accepted for treatments designed to reduce adverse outcomes in illnesses such as hypertension and hypercholesterolaemia. Hence, cost effectiveness in terms of the NNT to avert one event is mainly driven by the baseline absolute fracture risk.
Thus, knowledge of an individual’s absolute risk is central to making treatment decisions. The imperative to intervene increases with advancing age, lower bone mineral density (BMD) and previous fracture, as each of these contributes independently to fracture risk. About 85% of fractures occur in women over 60 years of age. An important signal for the need to treat is a prior vertebral or non-vertebral fracture. The risk of further fractures increases threefold to fivefold as the number or severity of prevalent vertebral deformities increases. In a person with osteoporosis, an incident fracture (with or without a prevalent fracture at baseline) increases the absolute risk of a further incident fracture to 30%–40% within 3 years.16 Thus, the evidence for anti-fracture efficacy is strongest in patients with a baseline vertebral or non-vertebral fracture. Withholding treatment from these patients does not comply with good medical practice.
It is optimal to treat fewer older people (over 60 years) at high risk rather than many younger people at low risk. This ensures that those likely to respond to treatment receive it, and those at low absolute risk, and thus unlikely to benefit, remain untreated. Treatment of the high-risk individual also makes good sense in view of the evidence that treatments reduce the risk of fracture within 6–18 months. Furthermore, most women stop treatment within 2 years, after which bone loss recurs, so women treated in their 50s may have little or no persisting risk reduction, and their susceptibility to fracture returns as they age and their risk of falls increases.
Women with osteoporosis and fractures: TREAT
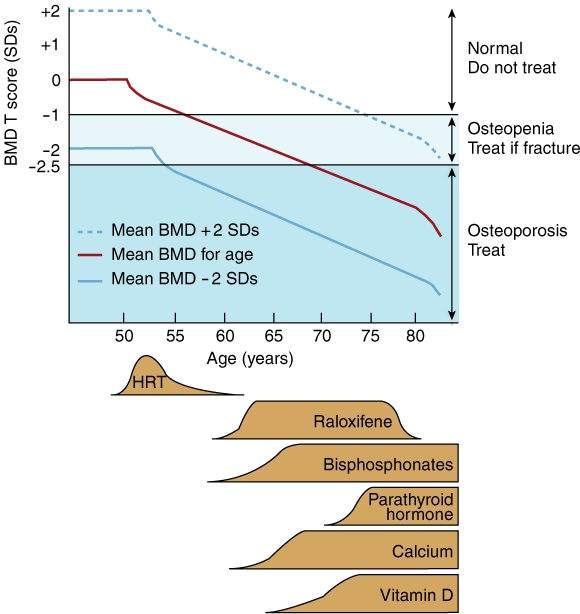
Osteoporosis is defined as a BMD T score < − 2.5 SD (where T score is the number of standard deviations from the mean BMD for young adults). Women with osteoporosis and fractures are at high risk of further fractures. The evidence for anti-fracture efficacy is compelling for alendronate, risedronate, raloxifene, parathyroid hormone and strontium ranelate (PTH is available in Australia but not reimbursed through the Pharmaceutical Benefits Scheme, while strontium ranelate is not yet available). Withholding treatment is inappropriate.
Women with osteopenia and a fracture: TREAT
Osteopenia is defined as a BMD T score between − 1 and − 2.5 SD. Women with osteopenia and a fracture are at increased risk for further fractures, even though they have modest deficits in BMD. Evidence of anti-fracture efficacy is available for alendronate, risedronate and raloxifene. Although the level of evidence is not as high as in women with osteoporosis, withholding treatment in these women is also inappropriate.
Women with osteoporosis but no fractures: TREAT
Women with osteoporosis (by BMD) are at increased risk of fracture. Evidence for anti-fracture efficacy rests with alendronate, raloxifene and possibly risedronate. For the last, the evidence is based on pooling of studies designed to examine BMD, as the main large studies enrolled only women with prevalent baseline fractures. The imperative to intervene increases with more advanced age, lower BMD and the presence of risk factors, such as illnesses causing bone loss. At present, these agents are not available on the Pharmaceutical Benefits Scheme for this group, despite good evidence to support their efficacy.
Women with osteopenia but no fractures: DEFER
Women with osteopenia (by BMD) are at increased risk of fracture, but the absolute risk is low. Therefore, treatment can be deferred. Consider follow-up in 1–3 years depending on risk factors for bone loss and other indications. Anti-fracture efficacy is difficult to demonstrate because of the low absolute risk of fracture. A recent post-hoc analysis of the MORE (Multiple Outcomes of Raloxifene Evaluation) trial provides evidence that raloxifene reduces the risk of vertebral fractures in women with osteopenia.17 There is evidence to support the use of alendronate, risedronate, raloxifene and HRT in preventing further bone loss.
A critical analysis of the main drug trials is beyond the scope of this review. Comparator trials are unavailable, so whether one drug is more efficacious than another is not known. Claims of superiority based on mean differences in relative fracture risk reduction cannot be sustained.
If the aim is to reduce vertebral fractures, then any one of the agents alendronate, risedronate or raloxifene, or parathyroid hormone, is suitable. However, use of parathyroid hormone is likely to be limited to severe osteoporosis, probably followed by an antiresorptive drug.11
If the aim is to reduce non-vertebral fractures, then a bisphosphonate should be used. The choice of drug may also be influenced by the mode of administration (eg, weekly for some bisphosphonates as opposed to daily) or by the need for extraskeletal effects, such as treatment of postmenopausal symptoms using HRT or a reduction in breast cancer risk using raloxifene. There is no evidence that combining bisphosphonates with either raloxifene or HRT reduces fractures more than either drug alone, although combined therapy may produce greater increases in BMD.
An approach to treatment of the individual is summarised in Box 4, and case reports in Box 5.
Women under 60 years of age: The absolute risk for fracture is low, and treatment is not usually necessary. HRT is a reasonable first-line approach in the early postmenopausal years in women with symptoms of menopause, provided that the risks of this treatment are understood. If the patient is asymptomatic and has a BMD T score > − 1 SD, no treatment is needed. If the BMD T score is between − 1 and − 2.5 SD, treatment depends on age. The imperative to treat increases with increasing age, lower BMD and presence of risk factors for bone loss (Box 4). If the BMD T score is already below − 2.5 SD, treatment should be considered, with a greater imperative to do so in older women and those with more severe osteoporosis, because of their higher absolute risk.
If a fracture has occurred, treatment should be given using a bisphosphonate or raloxifene. Women taking (or needing) HRT for managing symptoms of menopause should continue (or initiate) HRT, provided they are aware of the balance of risks and benefits. In the absence of oestrogen-deficiency symptoms, HRT is not recommended for fracture prevention. However, some women, stable on HRT and aware of the overall risks and benefits, may choose to continue with it. Otherwise, in women at high risk of fracture and with no menopausal symptoms, HRT should be replaced by a bisphosphonate or raloxifene. If HRT has been taken for many years, it should be ceased slowly to minimise symptoms. This is particularly important if the replacement drug is raloxifene, which should be introduced after a delay to ensure that hot flushes do not emerge (as these may be mistakenly ascribed to and worsened by raloxifene). Although breast cancer risk reduction is reported using raloxifene, prevention of breast cancer is not an approved indication for raloxifene.
Women over 60 years of age: The bisphosphonates or raloxifene are first-line approaches in women at risk of vertebral fractures because of osteoporosis. If the aim is the prevention of hip fractures, for example in women over 70–75 years with low femoral-neck BMD, a bisphosphonate should be used.
Lifestyle approaches, such as increasing calcium intake and weight-bearing exercise, as well as avoidance of excess alcohol and tobacco use, are recommended, even though the evidence for anti-fracture efficacy of each, or a combination of these approaches, is lacking. Vitamin D deficiency is common in elderly people who are housebound or institutionalised, and vitamin D replacement should be considered in these individuals.18 Hip protectors should be considered in elderly people at risk of falls.
Compelling evidence of the anti-fracture efficacy of drugs in men is lacking. Bisphosphonates and parathyroid hormone are likely to be efficacious. Androgen therapy is indicated in men with proven hypogonadism, but not in eugonadal men.
1: Cumulative survival probability after fracture
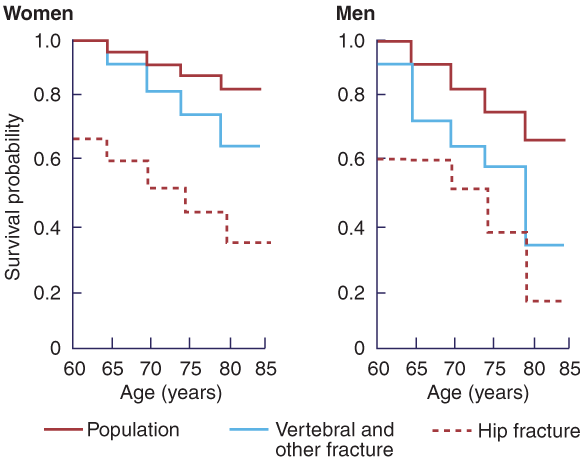
Survival is reduced after any type of fracture, vertebral or non-vertebral, more so in men than women. (Figure reprinted from Center et al4 with permission from Elsevier.)
2: Bone in osteoporosis
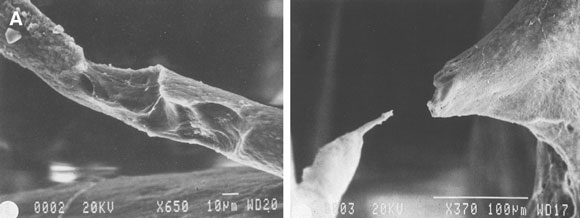
A. Imbalance between bone formation and resorption leads to trabecular thinning (left) and eventual loss of trabecular connectivity (right) (reprinted from Mosekilde,6 with permission from Elsevier).
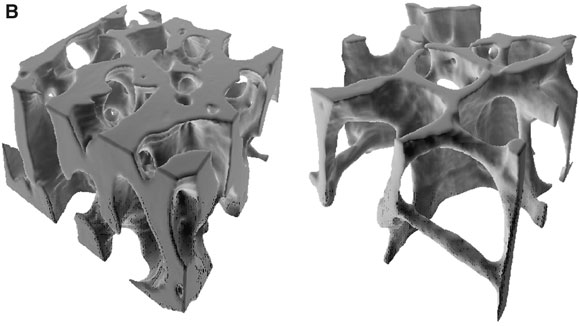
B. Loss of trabeculae and thinning (right) reduce cross-sectional area so that loads on bone are relatively greater (reprinted from Keaveny et al 8 with permission from Annual Review of Biomedical Engineering [www.annual.reviews.org]).
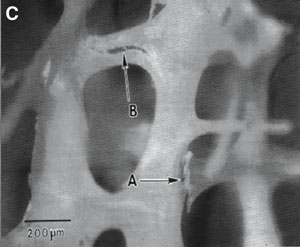 |
C. Microcracks in the trabeculae (arrows) (reprinted from Fyhrie and Schaffler, 9 with permission from Elsevier). |
4: Approach to treatment of the individual
The imperative to treat increases with:
Increasing age
Declining BMD
Prior fracture
Family history of osteoporosis
Risk factors for bone loss (eg, hyperparathyroidism, corticosteroid therapy, immobilisation, chronic illness)
High levels of bone remodelling markers
Treatment summary
If BMD is > − 1 SD, do not treat; if BMD is − 1 to − 2.5 SD, treat if fracture is present; and, if BMD is < − 2.5 SD, treat whether or not a fracture is present.
Consider hormone replacement therapy around menopause if the patient has postmenopausal symptoms.
Raloxifene or one of the bisphosphonates is appropriate in the later postmenopausal years.
Use bisphosphonates in women aged over 75 years when the main concern is preventing hip fracture.
Parathyroid hormone should be considered in patients with severe osteoporosis and fractures.
Give calcium supplements in people aged over 65 years, and vitamin D if deficiency is present or likely.
BMD = bone mineral density. * T score = number of standard deviations (SDs) from mean BMD for a young adult population.
5: Case reports — to treat or not to treat?
The perimenopausal years
Presentation: A 49-year-old woman presented because of concern about her risk of fractures, which had prompted her to have densitometry. The bone mineral density (BMD) T score (standard deviations from the young adult mean) was – 2.4 SD at the spine (defined as osteopenia) and − 2.7 SD at the femoral neck (osteoporosis).
History and examination: She was asymptomatic. Her menstrual cycles had ceased at the age of 45 years. Her mother had had a hip fracture at the age of 79 years and had since died. Examination detected no abnormalities.
Should this patient be investigated? Yes. It is important to exclude specific causes of bone loss (eg, primary hyperparathyroidism, malabsorption, thyroid disease, and vitamin D deficiency).
Should x-rays be done? Yes. Spine fractures may be asymptomatic and, if present, would be a clear indication for treatment to reduce the risk of further fractures.
Without evidence of spinal fractures, should she be treated for osteopenia of the spine or osteoporosis of the femoral neck? No, the aim of treatment is to prevent fractures, not bone loss. About 85% of all fractures occur in women over 60 years of age, so the risk of spinal fractures in the next 10 years is modest, and the risk of hip fractures is remote.
If spinal x-rays revealed several spinal fractures, should treatment be given? Yes. Prevalent fractures increase the risk of further fractures.
What drug should be used? Alendronate, risedronate or raloxifene are the best-investigated drugs for preventing further fractures.
A woman with back pain and height loss
Presentation: A 65-year-old woman presented with an episode of back pain. She mentioned that she was now 8 cm shorter than she had been in her youth.
History: The patient underwent menopause at age 50 years. Despite some hot flushes and a family history of osteoporosis, she avoided hormone replacement therapy (HRT) because of concern about its adverse effects, but did use “natural” therapy and increased her calcium intake. At 60 years, she fractured her right wrist in a hard fall and mentioned that her children thought she had become shorter. She was encouraged to increase her calcium intake and physical activity.
Investigation: On x-ray, she had three thoracic vertebral wedge and crush fractures.
Was the initial approach at age 60 years of increasing calcium intake and exercise sufficient? No. Any fracture without major trauma, especially in women with possible height loss and a family history of osteoporosis, warranted a spinal x-ray, which may have shown wedge fractures, and bone densitometry, which likely would have confirmed osteoporosis. Intervention at age 60 years could have halved her risk of further spinal and other fractures and helped maintain her quality of life.
Is it now too late to intervene? No. Agents such as alendronate, risedronate, and raloxifene can still improve bone density and halve her risk of future fractures. As her fracture risk is higher now than at age 60 years, the “absolute” benefit is even greater.
An elderly man with a fracture
Presentation: An 80-year-old man presented with a fracture of his right hip after a fall. He had lived in a nursing home for the past 8 years and had recently become less steady and had had a few falls.
Is the hip fracture typical for his age or does it require further investigation or treatment? Hip fractures are typical osteoporotic fractures and occur in men as well as in women. He requires investigation, including spinal x-rays and bone densitometry. Given that many, if not most, institutionalised or housebound elderly people in Australia are vitamin D deficient, serum 25-hydroxyvitamin D level should be checked, and vitamin D supplemented if deficient.
Is he too old to benefit from drug treatment? No. He is likely to benefit with reduced risk of further fractures from the use of a bisphosphonate, such as alendronate or risedronate. Moreover, given his high absolute risk he has a high chance of “absolute” benefit.
- Ego Seeman1
- John A Eisman2
- 1 Endocrine Unit, Austin and Repatriation Medical Centre, Melbourne, VIC.
- 2 Garvan Institute of Medical Research, St Vincent's Hospital, Sydney, NSW.
E S: Medical advisory boards of Aventis, Eli Lilly and Merck Sharp and Dohme. J A E: Paid consultancy for Aventis, Eli Lilly, Merck Sharp and Dohme, NPS (USA), Organon, Roche, and Servier.
- 1. Sambrook PN, Phillips SR, Seeman E, Ebeling PR. Preventing osteoporosis: outcomes of the Australian Fracture Prevention Summit. Med J Aust 2002; 176 (8 Suppl): S1-S16.<eMJA full text>
- 2. Randell A, Sambrook PN, Nguyen TV, et al. Direct clinical and welfare costs of osteoporotic fractures in elderly men and women. Osteoporos Int 1995; 5: 427-432.
- 3. Sanders KM, Nicholson GC, Ugoni AM, et al. Health burden of hip and other fractures in Australia beyond 2000 Projections based on the Geelong Osteoporosis Study. Med J Aust 1999; 170: 467-470. <eMJA full text>
- 4. Center JR, Nguyen TV, Schneider D, et al. Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet 1999; 353: 878-882.
- 5. Seeman E. The dilemma of osteoporosis in men. Am J Med 1995; 98: 75S-87S.
- 6. Mosekilde L. Consequences of the remodelling process for vertebral trabecular bone structure: a scanning electron microscopy study (uncoupling of loaded structures). Bone Miner 1990; 10: 13-35.
- 7. Seeman E. Pathogenesis of bone fragility in women and men. Lancet 2002; 359: 1841-1850.
- 8. Keaveny TM, Morgan EF, Niebur GL, Yeh OC. Biomechanics of trabecular bone. Annu Rev Biomed Eng 2001; 3: 307-333.
- 9. Fyhrie DP, Schaffler MB. Failure mechanisms in human cancellous bone. Bone 1994; 15: 105-109.
- 10. Delmas PD. Treatment of osteoporosis. Lancet 2002; 359: 2018-2026.
- 11. Seeman E, Delmas PD. Reconstructing the skeleton with intermittent parathyroid hormone. Trends Endocrinol Metab 2001; 12: 281-283.
- 12. Meunier PJ, Roux C, Seeman E, et al. The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med 2004; 350: 459-468.
- 13. Cranney AS, Guyatt G, Griffith L, et al. Summary of meta-analyses of therapies for postmenopausal osteoporosis. Endocr Rev 2002; 23: 570-578.
- 14. Rossouw JE, Anderson GL, Prentice RL, et al. Writing Group for the Women's Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women's Health Initiative randomized controlled trial. JAMA 2002; 288: 321-333.
- 15. Parker MJ, Gillespie LD, Gillespie WJ. Hip protectors for preventing hip fractures in the elderly. Cochrane Database Syst Rev 2003; (3): CD001255.
- 16. Lindsay R, Silverman SL, Cooper C, et al. Risk of new vertebral fracture in the year following a fracture. JAMA 2001; 285: 320-323.
- 17. Kanis JA, Johnell O, Black DM, et al. Effect of raloxifene on the risk of new vertebral fracture in postmenopausal women with osteopenia or osteoporosis: a reanalysis of the Multiple Outcomes of Raloxifene Evaluation trial. Bone 2003; 33: 293-300.
- 18. Nguyen TV, Center JR, Eisman J. Osteoporosis in elderly men and women: effects of dietary calcium, physical activity and body mass index. J Bone Miner Res 2000; 15: 322-331.





Abstract
All women and men with a history of fragility fractures should be considered for treatment of osteoporosis to reduce their risk of future fracture.
There is high-level evidence for the anti-fracture efficacy of treatment in women with osteoporosis, particularly if there is prevalent fracture; the evidence is less compelling for women with osteopenia, with or without a fracture, and for men.
The rigorously investigated drugs reported to reduce vertebral fractures are the bisphosphonates alendronate and risedronate, the selective oestrogen-receptor modulator raloxifene, the anabolic agent parathyroid hormone and, most recently, strontium ranelate.
Only the two bisphosphonates and hormone replacement therapy (HRT) have been reported to reduce hip fractures in community-dwelling women, and calcium plus vitamin D and hip protectors have been reported to reduce these fractures in elderly people in institutions.
HRT is not recommended in women for fracture risk reduction alone.
Evidence for the anti-fracture efficacy of calcitonin, fluoride, anabolic steroids and active vitamin D metabolites is insufficient to justify their use; lifestyle changes, while not shown to reduce fracture risk, may have a role in maintaining bone strength throughout life.