Patient 1 A 77-year-old woman was referred to us for an urgent surgical opinion regarding an abdominal mass. Fifteen months previously, she had been referred to a major metropolitan hospital with symptomatic iron deficiency anaemia, and weight loss of 5 kg in 6 months. Gastroscopy, colonoscopy and abdominal CT scan were unremarkable. There were no clinical features to suggest malabsorption, nutritional deficiency or inflammatory bowel disease. A diagnosis of angiodysplasia was considered, although there was no direct evidence of this. The patient received a 5-unit blood transfusion and iron supplements. During the 7 months after initial presentation, she was seen in medical outpatient clinics six times. Over the next 8 months, she was admitted six times, transfused 13 units of red cells, and given two iron infusions. A labelled red cell scan showed no gastrointestinal bleeding. An abdominal ultrasound was unremarkable. Results of investigations for coeliac disease and pernicious anaemia were normal. A bone marrow biopsy was consistent with iron deficiency anaemia. On one occasion, the admitting registrar considered a small bowel follow-through, but this was not performed. Paroxysmal nocturnal haemoglobinuria was excluded. During the last of these admissions, a firm abdominal mass was palpated. A computed tomography (CT) scan revealed an 8 cm heterogeneously enhancing soft tissue mass in the proximal jejunum. Lymphoma was considered the most likely diagnosis, and the patient was referred to our department. Push enteroscopy revealed an ulcerated adenocarcinoma in the proximal jejunum (A). A 10 cm, poorly differentiated jejunal adenocarcinoma was resected. Eleven months after surgery, the patient has shown no further symptoms of anaemia, and no evidence of recurrence. |
Patient 2 A 68-year-old man was admitted to our department for laparotomy for a small bowel tumour. The patient had a past history of an open cholecystectomy. Twenty-eight months before admission, he had been referred to a gastroenterologist with symptoms of gastro-oesophageal reflux disease; he underwent gastroscopy, which showed reflux oesophagitis, and was treated with proton pump inhibitors. Eleven months later, he was referred to a gastroenterologist again with persistent symptoms. He was seen several times in the next 9 months, and was then admitted to a general surgical service of a metropolitan hospital with abdominal pain. A provisional diagnosis of small bowel obstruction was made, and the patient was discharged the following day once his pain had settled. Twice during the next 5 months, he presented to the emergency department with abdominal pain, nausea and vomiting. His symptoms settled with analgesia and fluids. Persistent retrosternal pain had not been helped by a variety of H2 antagonists, proton pump inhibitors or prokinetic agents. He was admitted to our surgical service after presenting on two successive days with severe epigastric pain. Upper gastrointestinal endoscopy showed severe reflux oesophagitis. The first and second parts of the duodenum were normal. Abdominal CT showed a circumferential mass in the proximal jejunum with dilation of proximal bowel (B). Small bowel follow-through then showed an annular, stenosing lesion in the region of the duodenojejunal flexure. At laparotomy, a localised adenocarcinoma was resected (C). The patient had adjuvant chemotherapy and 12 months after surgery was asymptomatic, taking no medication, and had no evidence of recurrence. |
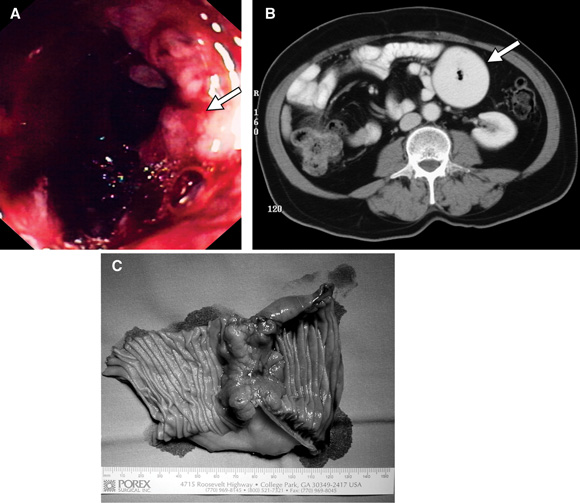
A: Small bowel carcinoma detected on push enteroscopy (Patient 1). B: Abdominal CT scan showing a large dilated small bowel loop in the left upper quadrant (Patient 2). C: Operative specimen showing a localised, constricting cancer (Patient 2).
Small bowel carcinoma is rare compared with gastric and colorectal cancer. Fifty-eight cases were reported in Victoria in 2001.1 Although the small bowel comprises 75% of the gastrointestinal tract length, less than 2% of gastrointestinal malignancies arise there.2-6 Adenocarcinoma accounts for 40% of small bowel malignancies; others include carcinoids, lymphomas and gastrointestinal stromal tumours, as well as metastases from melanoma, breast, lung and renal cancer.3,5,6
Small bowel tumours are rarely considered as a differential diagnosis, and their discovery is usually greeted with surprise. They are much more common in patients with coeliac disease (risk of lymphoma) and in hereditary bowel cancer syndromes (hereditary non-polyposis colorectal cancer, familial adenomatous polyposis, and Peutz–Jegher syndrome).2,4,6 Thus, small bowel tumours must be considered in patients with a family history of bowel cancer. A low index of suspicion for small bowel tumours is a result of their relative rarity, but delayed diagnosis may reduce the chance of successful treatment.
Reasons for diagnostic delay include the non-specific presentation, the lack of awareness of the diagnosis and the inaccessibility of the small bowel to investigation. In one analysis of 77 consecutive patients with primary small bowel malignancy over 22 years, average delays were 2 months before presentation to primary care physicians, 8 months from presentation until appropriate investigation, and 12 months from presentation until definitive diagnosis.7
Our first patient represents a common scenario. A patient with iron deficiency anaemia had investigations to exclude gastroduodenal, colonic, and extra-intestinal sources of blood loss. The inaccessibility of the small bowel to investigation resulted in a 15-month delay to diagnosis of a small bowel adenocarcinoma. An abdominal computed tomography (CT) scan performed early in the investigation of anaemia failed to show any disease, and the patient’s symptoms were attributed to intestinal angiodysplasia, a diagnosis that is difficult to confirm. The failure to perform small bowel follow-through, despite its low sensitivity, may have contributed to the delay in diagnosis.
Our second patient highlights the non-specific way in which small bowel adenocarcinoma can present. The patient presented with symptoms of reflux oesophagitis refractory to treatment. The abdominal CT scan was the pivotal investigation that alerted clinicians to a lesion in the jejunum.
CT is frequently used to assess the abdominal cavity. The accuracy of abdominal CT in detecting primary small bowel tumours is poor — reported as 57% in one study of 85 patients.5 It is a useful staging procedure, but, given the poor sensitivity for small bowel pathology, abdominal CT should be used in concert with other imaging modalities.
Barium contrast studies are the standard test for intraluminal or mucosal abnormalities beyond the duodenojejunal flexure, but have limited sensitivity.3-5 Small bowel enteroclysis is more sensitive than small bowel follow-through,8 and involves delivery of barium to the small bowel via an intestinal tube. Push enteroscopy, using a paediatric colonoscope, is an alternative, but does not visualise the entire small bowel. Video capsule endoscopy has shown promise in the diagnosis of small bowel disorders.9,10 It is not yet widely available, and further trials are required before its role is defined. It is contraindicated when small bowel strictures are suspected, so should be preceded by small bowel follow-through in these cases.9,10 Early studies show that it is more accurate than small bowel follow-through,9,10 so, when there is a strong clinical suspicion of small bowel pathology, patients should be referred to a specialist centre. Occasionally, exploratory surgery is appropriate, particularly when anaemia and abdominal pain coexist and the results of all other investigations have been negative.
- Ronil V Chandra1
- Julie A Miller2
- Ian T Jones3
- Brett Manley4
- G Bruce Mann5
- Department of Surgery, Royal Melbourne Hospital, University of Melbourne, Parkville, Vic.
- 1. Giles G, Thursfield V, editors. Canstat. Cancer in Victoria 2001. Melbourne: Cancer Council Victoria, 2003. Available at: www.cancervic.org.au/cancer1/facts/pdfs/canstats/canstat_39_cancer_vic_2001.pdf (accessed Jan 2004).
- 2. Neugut AI, Jacobson JS, Suh S, et al. The epidemiology of cancer of the small bowel. Cancer Epidemiol Biomarkers Prev 1998; 7: 243-251.
- 3. Neugut AI, Marvin MR, Rella VA, Chabot JA. An overview of adenocarcinoma of the small intestine. Oncology 1997; 11: 529-536.
- 4. Hutchins RR, Bani Hani A, Kojodjojo P, et al. Adenocarcinoma of the small bowel. Aust N Z J Surg 2001; 71: 428-437.
- 5. Minardi AJ Jr, Zibari GB, Aultman DF, et al. Small-bowel tumors. J Am Coll Surg 1998; 186: 664-668.
- 6. O’Riordan BG, Vilor M, Herrera L. Small bowel tumors: an overview. Dig Dis 1996; 14: 245-257.
- 7. Maglinte DDT, O’Connor K, Bessette J, et al. The role of the physician in the late diagnosis of primary malignant tumors of the small intestine. Am J Gastroenterol 1991; 86: 304-308.
- 8. Bessette JR, Maglinte DDT, Kelvin FM, Chernish SM. Primary malignant tumors of the small bowel: a comparison of the small-bowel enema and conventional follow-through examination. AJR Am J Roentgenol 1989; 153: 741-744.
- 9. Costamagna G, Shah SK, Riccioni ME, et al. A prospective trial comparing small bowel radiographs and video capsule endoscopy for suspected small bowel disease. Gastroenterology 2002; 123: 999-1005.
- 10. Chong AK, Taylor AC, Miller AM, Desmond PV. Initial experience with capsule endoscopy at a major referral hospital. Med J Aust 2003; 178: 537-540. <MJA full text>




