Assessment of acute bronchodilator reversibility is a commonly used tool in lung-function laboratories. The response to inhaled bronchodilator is used to help discriminate asthma from smoking-related chronic obstructive pulmonary disease (COPD) — a larger bronchodilator response is believed to indicate asthma. This has major implications for therapeutic management, especially with respect to whether inhaled corticosteroids are indicated.
Although spirometry is used to define the degree of reversibility in airflow limitation, there is little concordance between national1,2 and international3-7 guidelines as to the definition of significant bronchodilator reversibility. Furthermore, recent national1,2 and long-standing international8 guidelines, designed to optimise management of COPD and asthma, stress the importance of bronchodilator-response testing, but are inconsistent in their recommendations as to how to assess bronchodilator reversibility. Perhaps paradoxically, two guidelines that do attempt to describe methods for assessing bronchodilator response acknowledge that no standardised method exists.5,9
The method used to assess reversibility is likely to be crucial, as the degree of response may be affected by many factors, including the dose and type of bronchodilator used, the method of administration, and the time elapsed between drug administration and repeat spirometry to assess bronchodilator responsiveness.
We conducted a postal survey to determine the degree of concordance in the methods used to assess and interpret the reversibility of airflow limitation in lung-function laboratories throughout Australia and New Zealand.
In October 2000, we mailed a questionnaire to the senior scientist in 60 lung-function laboratories throughout Australia and New Zealand. The laboratories were identified from the Australian and New Zealand Society of Respiratory Science (ANZSRS) mailing list.
The questionnaire, designed specifically for our study, enquired about the bronchodilator drugs used, mode of administration, dose administered, and the definition of a significant bronchodilator response. Additionally, we asked how much time was allowed to elapse between the administration of the bronchodilator and repeat testing. We also asked what the minimal acceptable time was between the patients’ most recent use of bronchodilator agent and assessment of spirometry before calling a test “prebronchodilator”.
The criteria used to define a significant bronchodilator response were judged against the National Asthma Council (NAC) guidelines (an increase of 15% in forced expiratory volume in 1 second [FEV1] postbronchodilator),1 the Thoracic Society of Australia and New Zealand COPDX guidelines (an increase in FEV1 of more than 12% and 200 mL),2 and the American Thoracic Society (ATS) guidelines (≥ 2% of baseline and an absolute change of ≥ 200 mL in FEV1 or forced vital capacity [FVC]).3
It is important to note that the NAC and COPDX guidelines were released after our survey.
Ethics approval was not sought because of the nature of the study.
Of the 60 questionnaires posted, two were returned to sender. Fifty-one of the remaining 58 questionnaires were sent to hospital-based laboratories and seven to private laboratories. Thirty-seven completed questionnaires (33 hospital-based, 4 private) were returned, giving an overall response rate of 64% (Box 1).
Most respondents (27/37) had a written protocol for assessing reversibility of airflow limitation.
Twenty-four laboratories usually or always used a metered-dose inhaler (MDI), and 21 of these reported the use of a spacer device. Six laboratories reported using either an MDI and spacer device or jet nebuliser. This choice depended on criteria such as the age of the patient (1 laboratory), the patient’s current mode of therapy (2), FEV1/FVC ratio at baseline (1), and the patient’s clinical condition at the time of testing (1). One laboratory did not comment on their criteria.
Thirteen laboratories exclusively used jet nebulisers.
No laboratory used an ultrasonic nebuliser or a dry-powder delivery system.
Box 2 summarises the range of inhalation techniques used to deliver the bronchodilator using an MDI and spacer device. Twenty-two of the 24 laboratories using MDIs reported using salbutamol as the sole bronchodilator agent, and administered between two and eight actuations of 100 μg each (Box 3).
Laboratories that administered between six and eight MDI “puffs” of bronchodilator were exclusively assessing the bronchodilator response in paediatric patients.
Box 4 shows the time interval between administering the bronchodilator and repeat spirometry in the 33 laboratories that reported using only salbutamol as their bronchodilator agent. Where a laboratory reported a range of times, the mean has been used arbitrarily. Overall, the median time interval between administration and repeat spirometry was 10 minutes (range, 4–20 min).
The criteria used to define a significant bronchodilator response varied widely. Nine laboratories used criteria consistent with the NAC guidelines,1 one was consistent with the COPDX guidelines2 and two laboratories used the ATS guidelines.3 Another five, five and four approximated the NAC, COPDX and ATS guidelines, respectively.
The remaining respondents used absolute change only in FEV1 (2 laboratories), included changes in mid-flows (2), or used percentage change in FEV1 and/or FVC (2). Four did not state which parameter they measured, and one stated that doctors decide what is significant.
Box 5 shows the intervals that laboratories reported as minimum periods after taking a bronchodilator agent for spirometry to be “prebronchodilator”. Not all respondents answered for every bronchodilator agent listed in the questionnaire, but they were invited to make additional comments.
Twelve respondents commented that they would perform spirometry both pre- and postbronchodilator regardless of when the bronchodilator was last taken, but that the time and dose would be noted.
Five respondents stated that their request form asked for bronchodilator medications to be withheld before testing. The protocol in three laboratories was to discuss the appropriateness of bronchodilator testing with a physician if the patient had taken a bronchodilator within the specified time interval, to determine whether a further dose should be administered.
Our study has confirmed that there is wide variation in the methods used to assess and interpret reversibility of airflow limitation in Australian and New Zealand lung-function laboratories. This is an important finding, considering we only contacted laboratories employing staff who were members of the ANZSRS and that most respondents (89%) were from hospital-based laboratories.
This wide variety of methods to assess bronchodilator response is a result of the lack of evidence in this area, leading to consensus-based rather than evidence-based practice. Further studies are clearly needed to determine the clinical implications of these findings. The methods were so diverse that it is highly likely that the diagnosis of COPD and asthma would be affected. We feel laboratories should report the method used to assess bronchodilator response until standardised, evidence-based methods are adopted.
Most laboratories used salbutamol as their sole bronchodilator agent. However, the mode and duration of administration varied considerably, and some laboratories used anticholinergics or a combination of drugs to assess bronchodilator response. The rationale for these different practices is unclear. Some data suggest that using higher doses of bronchodilators in combination (ie, anticholinergics and β-agonists) will detect more reversibility in COPD,10,11 but whether this is clinically relevant is doubtful, and it could lead to further ambiguity in diagnosis and management.
The length of time before repeating spirometry to assess reversibility also varied considerably. Studies in asthmatic subjects have found that a significant improvement in ventilatory function can be expected to occur within 5 minutes of 200 μg salbutamol being administered via an MDI.12-15 This suggests that evidence for a bronchodilator “response” per se does not require a prolonged period of observation. However, this response may continue and reach a peak up to 50 minutes after administration of a bronchodilator.12-14 The significance of this in asthma has not been assessed, and even less is known about the time scale of bronchodilator response in COPD.
An additional confounding factor in determining bronchodilator responsiveness is the increasing use of long-acting β2-agonists. In this study, although the recommended times for withholding such medications were similar to published times,2,8,9 there was a wide range, and almost a third of respondents stated that they would assess bronchodilator reversibility regardless of how recently a long-acting β2-agonist had been taken. This will inevitably lead to an underestimation of the underlying bronchodilator response.
The variety of definitions used for a significant bronchodilator response is also likely to have a significant effect on patient management. For example, underestimation of the bronchodilator response in COPD may lead to withholding of bronchodilators or inhaled corticosteroid therapy, although few data exist for this latter recommendation. Of greater concern, and somewhat difficult to explain, is that the NAC1 and COPDX2 guidelines have different reversibility criteria, and neither set of recommendations is consistent with international guidelines.3-7 This disparity among guidelines probably reflects the lack of evidence.
Although our survey was performed in 2000, before the publication of either the NAC (2002) or COPDX (2003) guidelines, we believe comparing our data against their recommendations remains valid. It would be interesting to repeat the survey now, although we doubt laboratory practice will have changed given the inconsistent and conflicting information given by the NAC and COPDX guidelines.
In summary, there was little concordance between laboratories in the methods used to assess and interpret reversibility of airflow limitation, although three-quarters of responding laboratories had a written protocol. Further investigations to determine the likely clinical implications of these findings, especially in the setting of smoking-related COPD, and to assess the validity of different methods of bronchodilator administration are required. Only then can the methodology be standardised. Until this happens, bronchodilator reversibility data cannot be reliably interpreted across individual centres with any confidence.
Finally, if specialised lung-function laboratories cannot agree to use a standardised method and interpretation of bronchodilator-response testing and our professional bodies cannot agree on recommendations, what hope can we hold for developing reliable recommendations for community assessments in general practice?
1: Distribution and return of questionnaires
|
Distributed |
Returned |
|||||||||
|
Hospital-based |
Private |
Hospital-based |
Private |
|||||||
New Zealand |
8 |
1 |
3 |
1 |
|||||||
Australian Capital Territory |
1 |
0 |
0 |
0 |
|||||||
New South Wales |
9 |
2 |
6 |
2 |
|||||||
Queensland |
11 |
0 |
9 |
0 |
|||||||
South Australia |
7 |
0 |
4 |
0 |
|||||||
Tasmania |
2 |
0 |
1 |
0 |
|||||||
Victoria |
11 |
3 |
9 |
1 |
|||||||
Western Australia |
2 |
1 |
1 |
0 |
|||||||
Total |
51 |
7 |
33 |
4 |
|||||||
2: Inhalation technique for metered-dose inhaler via spacer device
Each actuation |
Number of laboratories (n = 21) |
||||||||||
One vital capacity (VC) breath and then breathhold |
12 |
||||||||||
Four tidal breaths |
1 |
||||||||||
One VC breath, breathhold and then tidal breathing |
2 |
||||||||||
Vary above methods to suit patients’ abilities |
6 |
||||||||||
3: Number of metered-dose inhaler actuations administered to assess bronchodilator response
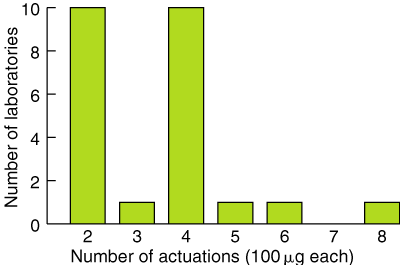
Results are for the 22 laboratories that used an MDI and salbutamol as the sole bronchodilator agent. Two laboratories gave more than one answer.
4: Time between bronchodilator administration and repeat spirometry
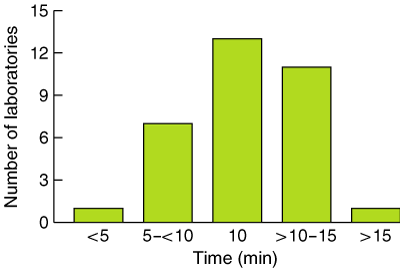
Results are for 33 laboratories that used salbutamol as the sole bronchodilator agent.
5: Reported minimum time elapsed after taking a bronchodilator agent for spirometry to be “prebronchodilator”
Drug |
Median (hours) |
Range (hours) |
Response rate |
||||||||
Salbutamol |
4 |
1–8 |
86% |
||||||||
Terbutaline |
4 |
1–12 |
76% |
||||||||
Ipratropium bromide |
4 |
1–8 |
81% |
||||||||
Fenoterol |
4 |
1–24 |
51% |
||||||||
Salmeterol |
12 |
1–24 |
76% |
||||||||
Eformeterol |
12 |
1–24 |
70% |
||||||||
Theophylline |
12 |
0.5–24 |
62% |
||||||||
Received 6 June 2003, accepted 8 March 2004
- Brigitte M Borg1
- David W Reid2
- E Haydn Walters3
- David P Johns4
- 1 Department of Allergy, Immunology and Respiratory Medicine, The Alfred Hospital and Monash University Medical School, Melbourne, VIC.
- 2 Discipline of Medicine, University of Tasmania, Hobart, TAS.
We are grateful to Eleonora Side for her assistance with the questionnaire and to laboratory staff who completed the questionnaire.
None identified.
- 1. National Asthma Council. Asthma management handbook 2002. Melbourne: National Asthma Council Australia Ltd, 2002.
- 2. Thoracic Society of Australia and New Zealand and The Australian Lung Foundation. The COPDX Plan: Australian and New Zealand guidelines for the management of chronic obstructive pulmonary disease 2003. Med J Aust 2003; 178 (6 Suppl): S1-S39. <MJA full text>
- 3. American Thoracic Society. Lung function testing: selection of reference values and interpretive strategies. Am Rev Respir Dis 1991; 144: 1202-1218.
- 4. European Respiratory Society. Standardized lung function testing. Eur Respir J 1993; 6 (Suppl 16).
- 5. British Thoracic Society and Association of Respiratory Technicians and Physiologists. Guidelines for the measurement of respiratory function. Respir Med 1994; 88: 165-194.
- 6. American College of Chest Physicians. Committee report. Criteria for the assessment of reversibility in airways obstruction: report of the committee on emphysema. Chest 1974; 65: 552-553.
- 7. Morris AH, Kanner RE, Crapo RO, Gardner RM. Clinical pulmonary function testing: a manual of uniform laboratory procedures. 2nd ed. Salt Lake City, Utah: Intermountain Thoracic Society, 1984.
- 8. British Thoracic Society. Guidelines for the management of COPD. Thorax 1997; 52 (Suppl 5): S1-S28.
- 9. American Association of Respiratory Care. Clinical practice guideline spirometry, 1996 update. Respir Care 1996; 41: 629-636.
- 10. Dorinsky PM, Reisner C, Ferguson GT, et al. The combination of ipratropium and albuterol optimizes pulmonary function reversibility testing in patients with COPD. Chest 1999; 115: 966-971.
- 11. Levin DC, Little KS, Laughlin KR, et al. Addition of anticholinergic solution prolongs bronchodilator effect of β agonist in patients with chronic obstructive pulmonary disease. Am J Med 1996; 100: 40S-48S.
- 12. Riding WD, Dinda P, Chatterjee SS. The bronchodilator and cardiac effects of five pressure-packed aerosols in asthma. Br J Dis Chest 1970; 64: 37-45.
- 13. Kamburoff PL, Prime FJ. Oral and inhaled salbutamol as a bronchodilator. Br J Dis Chest 1970; 64: 46-54.
- 14. Blom MW, Sommers DeK. Comparative pharmacology of salmeterol and formeterol and their interaction with salbutamol in healthy volunteers. Clin Drug Invest 1997; 14: 400-404.
- 15. Seberova E, Andersson A. Oxis (formeterol given by Turbuhaler) showed as rapid an onset of action as salbutamol given by a pMDI. Respir Med 2000; 94: 607-611.





Abstract
Objectives: To determine the variation in the methods used to assess and interpret the reversibility of airflow limitation in lung-function laboratories throughout Australia and New Zealand.
Design: A postal survey performed in 2000, requesting details of methods used to assess and interpret bronchodilator reversibility.
Setting and participants: 60 lung-function laboratories identified from the Australian and New Zealand Society of Respiratory Science mailing list.
Main outcome measures: Bronchodilator agent, dose, mode of administration, time to repeat spirometry and definition of a significant response.
Results: 37 laboratories responded (response rate, 64%). Thirty-three laboratories used salbutamol as their routine bronchodilator agent. Twenty-four laboratories used a metered-dose inhaler (MDI) with (21) or without (3) a spacer device as the preferred mode of bronchodilator administration. There was wide variation in the bronchodilator dose administered (median, 400 μg; range, 200–800 μg salbutamol for MDIs) and the time to repeat spirometry following bronchodilator administration (median, 10 min; range, 4–20 min). Ten laboratories used criteria consistent with either the National Asthma Council or Thoracic Society of Australia and New Zealand COPDX guidelines to define a significant bronchodilator response, and two used American Thoracic Society criteria. The remaining 25 respondents listed a variety of other criteria.
Conclusion: The methods used to assess and interpret acute bronchodilator reversibility in lung-function laboratories in Australia and New Zealand vary considerably. This may have a significant effect on the diagnosis and management of patients. Laboratories should report the method used to assess bronchodilator response.