The drug buprenorphine has altered the treatment of heroin dependence. Buprenorphine is characterised as a long-acting, partial mu opioid receptor agonist with effects similar to conventional opioid agonist drugs, such as morphine. It is thought to have a flat dose–response curve in that increasing the dose above about 4 mg prolongs the duration of effect but does not appear to increase the peak effect.1 Used in managing withdrawal ("detoxification"), buprenorphine provides good symptomatic relief and little rebound withdrawal on discontinuation.2,3 Many heroin users are reluctant to consider maintenance treatment initially, but after experiencing the stability produced by buprenorphine during outpatient detoxification, they often elect to remain on the drug for prolonged periods.3-5 As withdrawal from opioids is generally followed by relapse, encouraging people into maintenance treatment has potential health benefits. Buprenorphine has similar efficacy to methadone when used as a maintenance treatment for opioid dependence.6,7
Over the last decade in Australia, most patients being treated for heroin dependence have been managed by general practitioners. However, there has been little evaluation, with no direct comparisons of effectiveness between clinic-based and primary-care-based treatment. We aimed to compare the effectiveness of buprenorphine-assisted detoxification (with the option of transferring to post-withdrawal maintenance therapy) in primary care and specialist settings.
Participants were recruited between 23 May 2000 and 30 May 2001. Random allocation into experimental (primary care setting) and control (specialist setting) groups was performed by the National Health and Medical Research Council Clinical Trials Centre randomisation service.
Potential participants were either self-referred or referred from one of the treatment sites and assessed at the specialist clinic. Opioid-dependent people aged between 16 and 65 years who had stable accommodation within a 6 km radius of the clinic were eligible for recruitment. Exclusion criteria included pregnancy, lactation, dependence on benzodiazepines or alcohol, receiving methadone treatment within the previous 2 months, and unstable medical or psychiatric conditions (active psychosis, depression with significant suicide risk).
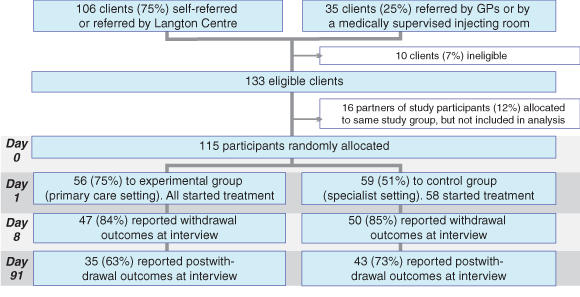
One hundred and fifteen heroin-dependent participants (defined by Diagnostic and statistical manual of mental disorders, 4th edition, criteria8) who were seeking detoxification were randomly allocated to receive treatment in either a specialist clinic (The Langton Centre) or with a GP. Six GPs working within a 5 km radius of the clinic participated, after receiving a treatment protocol and attending a training session.
Participants underwent a 5-day detoxification regimen, attending daily to receive a supervised dose of buprenorphine. The dose given was 6 mg on Day 1, 8–10 mg on Day 2, 8–12 mg on Day 3, 6–10 mg on Day 4, and 4 mg on Day 5.9 They then received no study medication for 2 days, and were re-assessed on Day 8, at which time they were given the opportunity to begin a range of treatments. Participants allocated to primary care were dispensed their detoxification medication from the doctor's surgery from Monday to Friday, and from the specialist clinic on the weekend. At each visit, practitioners were encouraged to review side effects, dose adequacy, the participant's goals and postdetoxification treatment options.
During the detoxification phase, participants did not pay for medication. Among those who subsequently elected to receive buprenorphine maintenance treatment, those allocated to the primary care setting received a daily supervised dose at a community pharmacy, paying a $25 per week dispensing fee. Those who were allocated to the specialist clinic subsequently paid no dispensing fee; all participants had given informed consent at randomisation about the possible difference in fees. Buprenorphine was provided in the form of standard sublingual tablets, and was delivered to the primary care sites by staff from the specialist clinic. It was stored, dispensed, and records were kept according to current S8 legislation.
Research interviews were conducted on Days 1, 8 and 91. Key outcome measures were completion of the detoxification program (Day 8), heroin use (Days 8 and 91), and retention in postwithdrawal treatment. Self-reported heroin use was monitored using a modified drug use section of the Opiate Treatment Index (OTI),10 and supplemented by results of urine drug testing on Days 4, 8 and 91. Urine samples for drug screening were collected weekly from participants undergoing postwithdrawal treatment. Retention in treatment programs was measured as the number of days each participant actively participated in treatment. Participants who failed to attend their first appointment (Day 1), missed 2 consecutive days during detoxification treatment, did not attend the research interview on Day 8, or missed 3 consecutive days of maintenance treatment were deemed to have discontinued.
We used cost-effectiveness analysis to compare treatment costs with treatment outcomes (see below). Any costs borne by participants or other health agencies were excluded. The primary outcome measure used in the economic analysis was abstinence from heroin use on Day 8. Results are presented as an incremental cost effectiveness ratio — the ratio of the difference in costs between the two groups over the difference in outcomes. A detailed description of the health economic method is currently being prepared in a separate report (C Doran, Health Economist, National Drug and Alcohol Research Centre, University of New South Wales, Sydney, NSW, personal communication).
Treatment costs included patient-level resources such as staff time (actual salary), diagnostics tests performed (cost derived from the Medical Benefits Schedule [MBS])11 and medications consumed (cost obtained from MIMS).12 Facility-level resources included the operating costs at the clinic, including supplies, consumables, capital and equipment. Clinical records for every second patient were reviewed, and averages of staff time and the cost of diagnostic procedures were used to estimate costs for all patients. Averages were applied based on a patient's retention in study treatment. Financial data pertaining to the clinic's operating costs were apportioned to the detoxification unit and converted into dollar value per minute of contact. The total of patient and facility resource use provides an estimate of total cost of a patient's treatment episode at the clinic.
GPs completed research forms to capture similar information to that recorded at the clinic. The cost of GP time was valued according to the MBS.11 All patients were assessed at the clinic; as some attended the clinic on weekends, some clinic-related facility costs were allocated to primary care patients.
In a previous study, 60% of clinic patients entered continuing treatment.3 Assuming that, with less access to ancillary services and having to pay for continuing treatment, only 30% of participants would enter ongoing treatment in the primary care group, power calculation indicated that to have an 80% chance of detecting this difference, 48 participants were required for each treatment-setting group (2-tailed comparison).
Statistical analyses were conducted on an intention-to-treat basis. Independent t tests, paired t tests and two-sample Kolmogorov–Smirnov tests were performed on the proportions of participants in each group who completed withdrawal, mean costs between groups, and overall changes in self-reported heroin use between baseline and Day 91. Kaplan–Meier survival analysis was performed on retention in postwithdrawal treatment. Patients who could not be interviewed for the heroin use analysis were assumed to have relapsed, and baseline data were imputed. If couples presented for treatment, only one partner was randomly allocated to a group, and both received the same treatment. Only data from the randomly allocated partner were included in analysis.
Six of seven GPs invited to participate agreed to do so. All six were experienced in treating opioid-dependent patients, but had no prior experience of using buprenorphine. One practitioner declined because his practice partners did not want more heroin users at the practice.
Box 1 shows the flow of participants through the trial, including research interview follow-up rates.
Characteristics of the two treatment groups are shown in Box 2. While median values of self-reported drug use were very similar, the distribution of responses was widely skewed. There was a significant difference in baseline self-reported heroin use between the groups, as the clinic group included several people who had undergone a period of detoxification before entering the study. A two-sample Kolmogorov–Smirnov test showed the clinic group had significantly lower baseline heroin use (P = 0.04). This baseline difference was considered in analyses of heroin use between groups at Day 91.
A summary of mean treatment costs for detoxification is provided in Box 3. An episode of buprenorphine detoxification in primary care was estimated to be $24 more expensive than treatment at the clinic. This difference in mean cost was just statistically significant (independent sample t test, t = 1.968; P = 0.05). The higher treatment costs in primary care were a result of additional staff time, particularly at the clinic on weekends, and subsequent allocation of operating costs of $107 per patient for time spent at the clinic (an average of 49 minutes).
Some form of postwithdrawal treatment was entered by 28/56 primary care participants (50%) and 36/59 clinic participants (61%) (Pearson χ2; df = 1; P = 0.23). Of these, 26/28 primary care participants (93%) and 34/36 clinic participants (94%) selected buprenorphine maintenance treatment. The remaining two participants in the primary care group selected methadone maintenance, and the two in the clinic group selected naltrexone maintenance.
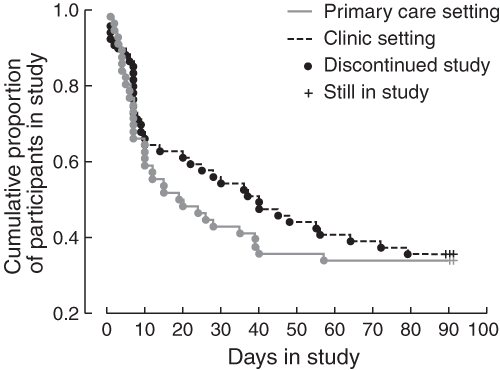
Box 4 shows an analysis of retention in treatment. Those who remained in treatment until Day 91 comprised 19/56 (34%) in primary care and 21/59 (36%) in the clinic group. A Kaplan–Meier analysis performed on the difference in retention between treatment groups did not approach statistical significance (Cox's F test; P = 0.27). A χ2 analysis of differences in retention between the six primary care doctors approached, but did not reach, significance at the 5% level (χ2 = 10.39; df = 5; P = 0.065).
There was no significant difference in self-reported heroin use between groups at Day 91 (independent-sample t test, t = 0.391; df = 76; P = 0.7). The significant difference in baseline heroin use between groups did not alter this result. As most participants in both groups had a frequency of baseline heroin use of 28 days per month, it is unlikely that even a highly significant effect of baseline heroin use on outcome would affect the analysis.
The 78 participants who were interviewed at Day 91 (see Box 5) showed a significant decrease in heroin use between baseline and Day 91 (dependent-sample t test; P < 0.001). This difference remained significant when missing data were imputed with baseline heroin use levels (dependent-sample t test, n = 115; P < 0.0001). The 38 participants who were interviewed and remained in treatment until Day 91 reported significantly less heroin use compared with the 40 who were interviewed, but had discontinued treatment before Day 91 (independent-sample t test; P < 0.001).
Outpatient heroin detoxification regimens typically result in about 20%–30% of participants completing withdrawal, although criteria for defining "completion" vary.13 In our study, 21% of participants completed 8 days of ambulatory detoxification without the use of additional opioids, and 30% submitted urine tests free of morphine at Day 8.
Precipitated withdrawal after the first dose of buprenorphine occurred in 10% of participants, but was easily managed in all but one case by elevating the second buprenorphine dose. These figures closely compare to a similar study where 7/55 participants (13%) receiving buprenorphine experienced a precipitated withdrawal.3
The results of the economic evaluation suggest that buprenorphine treatment in primary care is a cost-effective alternative to treatment in a clinic. The cost of detoxification was found to be marginally higher in primary care. However, if the use of the clinic by the primary care group decreased, particularly in the assessment phase, costs of primary care treatment may decrease.
As one aim of our study was to assess the short-term effectiveness of buprenorphine in detoxification from heroin, there was a gap of 2 days before people could choose to enter postwithdrawal treatment. This may have reduced the number of participants who entered postwithdrawal treatment.
Successful withdrawal from heroin, without follow-up treatment, is followed by a high likelihood of relapse to heroin use within 12 months.14 Therefore, there is little rationale for investing in detoxification services without also trying to retain people in postwithdrawal treatment. In our study, 64/115 participants (56%) entered postwithdrawal treatment, mostly buprenorphine maintenance, and 40/115 (35%) were still in treatment at Day 91. Assuming that participants who were not followed up had returned to baseline levels of heroin use, at Day 91 of follow-up there was a highly significant reduction in heroin use, with participants using heroin on about half the number of days.
None of the outcomes differed between the treatment settings. It appears that buprenorphine treatment can be initiated in primary care settings, and participants are retained in this treatment as well as in a clinic setting. Retention in the primary care group was particularly impressive in view of the fact that only the primary care participants paid for postwithdrawal treatment.
There are several limitations to the generalisability of our findings. The GPs in this study were all experienced in using methadone to treat heroin dependence, and were provided with training and protocols for using buprenorphine. Inexperienced and untrained practitioners would probably not attain comparable results. Indeed, there appears to be reluctance on the part of many GPs to become involved at all in the treatment of drug users.15
Our findings suggest that buprenorphine treatment of heroin dependence, involving general practitioners and retail pharmacists to supply the drug and supervise the dosing, can be initiated safely, effectively and with similar cost-effectiveness as treatment in specialist clinics.
2: Participant characteristics
|
Specialist clinic (n = 59) |
Primary care (n = 56) |
Total (n = 115) |
||||||||
Male |
37 (63%) |
33 (59%) |
70 (61%) |
||||||||
Aboriginal |
2 (3%) |
4 (7%) |
6 (5%) |
||||||||
Route of heroin administration |
|
|
|
||||||||
Inject |
47 (80%) |
42 (75%) |
89 (77%) |
||||||||
Non-injecting only |
12 (20%) |
12 (21%) |
24 (21%) |
||||||||
Highest education completed |
|
|
|
||||||||
Years 7–10 |
15 (25%) |
15 (27%) |
30 (26%) |
||||||||
Years 11–12 |
26 (44%) |
16 (29%) |
42 (37%) |
||||||||
Tertiary |
18 (31%) |
25 (45%) |
43 (37%) |
||||||||
Employment status |
|
|
|
||||||||
Employed or student |
34 (58%) |
30 (54%) |
64 (56%) |
||||||||
Unemployed or pensioner |
25 (42%) |
26 (46%) |
51 (44%) |
||||||||
Heroin use by partner |
|
|
|
||||||||
No partner |
30 (51%) |
18 (32%) |
48 (42%) |
||||||||
Dependent user |
15 (25%) |
18 (32%) |
33 (29%) |
||||||||
Non-dependent user |
3 (5%) |
3 (5%) |
6 (5%) |
||||||||
No heroin use |
11 (19%) |
17 (30%) |
28 (24%) |
||||||||
Prior treatment experience |
|
|
|
||||||||
Any treatment for heroin use* |
51 (86%) |
46 (82%) |
97 (84%) |
||||||||
Supervised detoxification† |
47 (80%) |
45 (80%) |
92 (80%) |
||||||||
Methadone maintenance |
32 (54%) |
23 (41%) |
55 (48%) |
||||||||
Age in years (median [range]) |
|
|
|
||||||||
At baseline |
30 (18–48) |
29 (17–46) |
29 (17–48) |
||||||||
First heroin use |
21 (11–41) |
20 (12–38) |
20 (11–41) |
||||||||
First regular heroin use |
23 (12–41) |
24 (13–39) |
24 (12–41) |
||||||||
Frequency of heroin use |
|
|
|
||||||||
Median days used in past 28 (range) |
28 (10–28)‡ |
28 (19–28) |
28 (10–28) |
||||||||
Median years heroin-dependent (range) |
3 (0.5–32) |
4 (0.5–22) |
3 (0.5–32) |
||||||||
* Includes outpatient and inpatient detoxification, rapid opiate detoxification, residential treatment, maintenance treatment. † Includes outpatient and inpatient detoxification, rapid opiate detoxification. ‡ Difference between specialist and primary care settings; P = 0.04, two-sample Kolmogorov–Smirnov test of significance. |
|||||||||||
3: Mean cost of treatment for detoxification
Category |
Specialist clinic (n = 59) |
Primary care (n = 56) |
|||||||||
Diagnostic tests |
$37 |
$39 |
|||||||||
Medication costs |
$18 |
$19 |
|||||||||
Staff costs |
|
|
|||||||||
Clinic |
$102 |
$69 |
|||||||||
General practitioner |
— |
$122 |
|||||||||
Overheads and administration |
$176 |
$107 |
|||||||||
Total mean cost (± standard deviation) |
$332 (± $70) |
$356 (± $74) |
|||||||||
5: Heroin use at baseline and at Day 91
|
Mean number of days participants used heroin in the month before interview (± standard deviation) |
||||||||||
|
Specialist clinic |
Primary care |
Total |
||||||||
Baseline |
(n = 59) 27.09 (± 2.31) |
(n = 56) 25.75 (± 3.86) |
(n = 115) 26.40 (± 3.26) |
||||||||
Day 91 |
|
|
|
||||||||
In treatment, interviewed |
(n = 20) 4.75 (± 5.27) |
(n = 18) 3.44 (± 5.09) |
(n = 38) 13.00 (± 5.16)* |
||||||||
Out of treatment, interviewed |
(n = 23) 10.57 (± 10.41) |
(n = 17) 13.88 (± 11.84) |
(n = 40) 11.98 (± 11.02)† |
||||||||
Full sample (missing data imputed) |
(n = 56) 15.39 (± 12.22) |
(n = 59) 12.76 (± 11.25) |
(n = 115) 14.04 (± 11.75)‡ |
||||||||
* Comparison of 38 participants at baseline and Day 91; significant at P < 0.001 (dependent-sample t test). † Comparison between participants in treatment and out of treatment at day 91; significant at P < 0.001 (independent-sample t test). ‡ Comparison of 115 participants at baseline and Day 91; significant at P < 0.001 (dependent-sample t test). |
|||||||||||
- Amy E Gibson1
- Christopher M Doran2
- James R Bell3
- Anni Ryan4
- Nicholas Lintzeris5
- 1 National Drug and Alcohol Research Centre, University of New South Wales, Sydney, NSW.
- 2 The Langton Centre, Sydney Hospital and Sydney Eye Hospital, Sydney, NSW.
- 3 National Addiction Centre, Institute of Psychiatry, Kings College, London, UK.
We thank the primary care medical practitioners and pharmacists (David Baker, Raymond Seidler, John Faros, Paul Sztolcman, Chris Blainey, Simon de Burgh, Nick Serafim, Robert Austic, Paul Sharman, Paul Kelly, Ross Fenton, Daniel Ng) and the clinicians of The Langton Centre, Sydney; NSW Health for funding this research, NHMRC Clinical Trials Centre for random allocation of participants; PALMS Toxicology for diagnostic testing; the National Drug and Alcohol Research Centre, and the National Evaluation of Pharmacotherapies for Opioid Dependence project for research support and data monitoring.
Reckitt Benckiser provided funding for J Bell and N Lintzeris to attend a conference.
- 1. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effect at high doses. Clin Pharmacol Ther 1994; 55: 569-580.
- 2. Cheskin LJ, Fudala PJ, Johnson RE. A controlled comparison of buprenorphine and clonidine for acute detoxification from opioids. Drug Alcohol Depend 1994; 36: 115-121.
- 3. Lintzeris N, Bell J, Bammer G, et al. A randomised controlled trial of buprenorphine in short-term ambulatory heroin withdrawal. Addiction 2002; 97: 1395-1404.
- 4. Vignau J. Preliminary assessment of a 10-day rapid detoxification program using high dosage buprenorphine. Eur Addict Res 1998; 4 Suppl 1: 29-31.
- 5. Lintzeris N, Clark N, Muhleisen P, et al. National clinical guidelines on the use of buprenorphine in the management of heroin dependence. Canberra: National Drug Strategy, 2001.
- 6. Barnett PG, Rodger JH, Bloch DA. A meta-analysis comparing buprenorphine to methadone for treatment of opiate dependence. Addiction 2001; 96: 683-690.
- 7. West SL, O'Neal KK, Graham CW. A meta-analysis comparing the effectiveness of buprenorphine and methadone. J Subst Abuse Treat 2000; 12: 405-414.
- 8. American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th ed. Washington DC: APA, 2000.
- 9. Lintzeris N. Buprenorphine dosing regimens in the management of outpatient heroin withdrawal. Drug Alcohol Rev 2002; 21: 39-45.
- 10. Darke S, Hall W, Wodak A, et al. Development and validation of a multi-dimensional instrument for assessing outcome of treatment among opioid users: The Opiate Treatment Index. Br J Addiction 1992; 87: 733-742.
- 11. Medicare benefits schedule book. Canberra: Commonwealth Department of Health and Family Services, 2001.
- 12. MIMS Australia. 25th ed. Sydney: MediMedia Australia, 2001.
- 13. Gowing LR, Ali RL, White JM. The management of opioid withdrawal: an overview of the research literature. DASC Monograph No. 9, Research Series. Adelaide: Drug and Alcohol Services Council, 2000.
- 14. Mattick RP, Hall W. Are detoxification programmes effective? Lancet 1996; 347: 97-100.
- 15. Abouyanni G, Stevens LJ, Harris MF, et al. GP attitudes to managing drug- and alcohol-dependent patients: a reluctant role. Drug Alcohol Rev 2000; 19: 165-170.





Abstract
Objective: To compare outcomes, costs and incremental cost-effectiveness of heroin detoxification performed in a specialist clinic and in general practice.
Design and setting: Randomised controlled trial set in a specialist outpatient drug treatment centre and six office-based general practices in inner city Sydney, Australia.
Participants: 115 people seeking treatment for heroin dependence, of whom 97 (84%) were reinterviewed at Day 8, and 78 (68%) at Day 91.
Interventions: Participants were randomly allocated to primary care or a specialist clinic, and received buprenorphine for 5 days for detoxification, then were offered either maintenance therapy with methadone or buprenorphine, relapse prevention with naltrexone, or counselling alone.
Main outcome measures: Completion of detoxification, engagement in post-detoxification treatment, and heroin use assessed at Days 8 and 91. Costs relevant to providing treatment, including staff time, medication use and diagnostic procedures, with abstinence from heroin use on Day 8 as the primary outcome measure.
Results: There were no significant differences in the proportions completing detoxification (40/56 [71%] primary care v 46/59 [78%] clinic), participating in postwithdrawal treatment (28/56 [50%] primary care v 36/59 [61%] clinic), reporting no opiate use during the withdrawal period (13/56 [23%] primary care v 13/59 [22%] clinic), and in duration of postwithdrawal treatment by survival analysis. Most participants in both groups entered postwithdrawal buprenorphine maintenance. On an intention-to-treat basis, self-reported heroin use in the month before the Day 91 interview was significantly lower than at baseline (27 days/month at baseline, 14 days/month at Day 91; P < 0.001) and did not differ between groups. Buprenorphine detoxification in primary care was estimated to be $24 more expensive per patient than treatment at the clinic. The incremental cost-effectiveness ratio reveals that, in this context, it costs $20 to achieve a 1% improvement in outcome in primary care.
Conclusions: Buprenorphine-assisted detoxification from heroin in specialist clinic and primary care settings had similar efficacy and cost-effectiveness. Buprenorphine treatment can be initiated safely in primary care settings by trained GPs.