During 1998, the most recent year for which there are comparable international outcome data,1,2 in-vitro fertilisation (IVF), used chiefly as a clinical treatment for infertility, accounted for 1.5% of babies conceived in Australia.3 Australian national data compiled by the National Perinatal Statistics Unit (NPSU)3 do not link pregnancies from embryos transferred after a period of cryostorage to egg retrieval and fresh embryo transfer cycles, so it is not possible to determine from these data a woman's total chance for a live birth from one retrieval. Nor can the effect of demographic variables such as age be assessed using NPSU data. In particular, while it is known that the chance of success falls with the woman's age, the timing and rate of this decline in Australian women are not known.
I have analysed the outcomes of IVF egg retrievals performed at one Australian clinic during 1998, encompassing all embryo transfers from these retrievals up to 30 June 2001, with an analysis of outcomes through to 30 June 2002. The chief end-point for analysis is a live birth. My results augment nationally available data on IVF practices and outcomes in Australia and enable socially important conclusions to be drawn.
My study analysed 648 egg retrievals carried out in 1998 at Sydney IVF. The median age of the women was 36 years (range, 22–48 years). All women in the study were infertile and the couples had been investigated with repeated semen analysis, tests for ovulation, ultrasound and/or hysteroscopy for uterine and endometrial normality, and laparoscopy to establish tubal anatomy and to diagnose or exclude endometriosis.4,5
Ovarian follicular recruitment was enhanced with the subcutaneous administration of pure follicle-stimulating hormone (FSH) according to standard protocols.1,6 Aspiration of mature ovarian follicles for the retrieval of oocytes was conducted with the patient in a sedated but conversant state.
The preparation of sperm and oocytes for fertilisation in vitro, either naturally or using intracytoplasmic sperm injection (ICSI), and the culture of fertilised eggs is described elsewhere7,8 and differed from common contemporary practices. In brief, small-chamber, bench-top, "mini" incubators (K-MINC-1000, Cook IVF, Eight Mile Plains, QLD) were used, together with proprietary Sydney IVF culture medium (Cook IVF), in which cleavage occurs in medium supported by pyruvate, but with zero or low glucose concentrations and with a continuous flow of 6% carbon dioxide and 5% oxygen in nitrogen.8 Embryos were considered suitable for storing if continuing cleavage was demonstrable on Day 3 or 4 of culture and if blastomere fragmentation was slight. Embryos were stored at the 2- to 8-cell stage and were thawed later using a standard propanediol/sucrose method.7
Embryos were transferred to the uterus through the cervix without sedation using a standard vaginal speculum and a specially designed embryo transfer catheter set (K-JETS-6019, Cook IVF).
Excluding cycles in which all embryos suitable for transfer were electively stored, the modal and median number of embryos transferred was 2 (range, 0–4): in women under 35 years, a mean of 1.90 (range, 0–4) embryos were transferred, and in those over 35 years the mean was 2.08 (range, 0–4). If it was judged that the ovarian hyperstimulation syndrome (OHSS)9 was a special risk, all embryos were stored (n = 56 retrievals). This tactic enabled us to avoid withdrawing FSH before full follicular maturity, risking either immature eggs or early follicular atresia, and no women managed this way required hospital admission.
Cryostored and thawed embryos were transferred in cycles in which the ovaries were suppressed and the endometrium developed with a sequence, starting at menstruation, of exogenous orally administered oestrogen (ethinyl oestradiol, Progynon C, Schering, Berlin, imported under the Therapeutic Goods Administration's Authorised Prescriber Program; 30 μg per day for 7 days, then 50 μg per day until Day 14), and vaginally administered progesterone pessaries (USP, Sigma, St Louis, Mo, USA; 200 mg/d, with 20 μg ethinyl oestradiol). This sequence is consistent with standard protocols, while also inhibiting spontaneous ovulation.
Non-parametric statistics were used for ranked, non-normally distributed data, χ2 statistics for categorically distributed data, and conventional parametric statistics (including t tests) for normally distributed comparison of groups; in each case, probability levels of falsely rejecting null hypotheses are stated as calculated. For non-parametric data, 95% confidence intervals are given; confidence intervals for proportions were calculated or obtained from tables.10
A total of 565 women (median age, 36 years; range, 22–48 years) had 648 egg retrievals at Sydney IVF during the period. Their age was about normally distributed around an average of 35.8 years (Box 1), about 1.5 years older than the national average of women having IVF according to NPSU data3 (34.2 years) (df > 120; t = 8.2; P < 0.001). During this time there were 54 treatments that were initiated but were cancelled before egg retrieval. Forty of the latter are included in the study population, as egg retrieval was subsequently accomplished with a changed stimulatory regimen, whereas in 14 of 579 women (an effective permanent cancellation rate of 2.4%) no egg retrieval could be performed.
Pregnancy outcomes are given in Box 2. For the cohort less than 35 years, of the 132 live births 45 (34.1%) were from embryos that had been cryostored; for the 35–39-years cohort, cryostored embryos accounted for 25 of 76 live births (32.9%), and for the over-40 cohort for 4 of 16 live births (25.0%).
The implantation rate of embryos — the chance of each transferred embryo forming an ultrasound-visible gestational sac — also fell with age. Under 35 years it averaged 24.7% per embryo, from 35 to 39 years it averaged 13.4%, from 40 to 44 years it averaged 7.0%, and over 44 years there were no implantations.
The miscarriage rate was highly age-dependent (P < 0.001, multiple logistic regression), and in each age group the rate appeared to be lower among fresh embryos than cryostored embryo transfers: < 35 years, 8/99 (8.1%) and 8/54 (14.8%), respectively; 35–39 years, 11/71 (15.5%) and 4/22 (18.2%), respectively; 40–44 years, 9/22 (40.9%) and 3/6 (50.0%), respectively, but the apparent trend is not significant (odds ratio = 1.6 that an absence of cryostorage is an independently important advantageous variable; 95% CI, 0.8–3.3; P = 0.2, logistic regression). The miscarriage rate for combined fresh and frozen embryos (Box 2) was normal for age and low for an infertile population.12
Since the first IVF birth was reported in Australia in 1980,13 there have been more than 43 500 babies born after IVF and related assisted-conception techniques in Australia and New Zealand.14 During this time, major changes in clinical practice have taken place, especially with transvaginal ultrasound-based egg retrieval; the development of intracytoplasmic sperm injection into eggs (ICSI); a steady but slow general improvement in pregnancy rates;14 and a decline after 1992 in an alternative form of assisted conception — gamete intrafallopian transfer (GIFT). In GIFT, gametes are obtained as for IVF, but then transferred immediately to the fallopian tubes, where fertilisation is meant to follow.15 Internationally, GIFT had declined in use by 1998,1,2 and today IVF is widely and effectively used for infertility caused by a wide range of pathological causes, including unexplained infertility and infertility of multiple aetiology.16
In 1998, the viable birth rate per embryo transfer after IVF (including cycles in which ICSI was employed) was 17.4% in Australia3 (or 16.8% excluding the present series), 22.1% in the United Kingdom,1 26% in Sweden,1 27% in Norway,1 and 31% in the United States (where high numbers of embryos are commonly transferred at once).2
Pregnancy rates per retrieval or transfer do not correspond exactly with the practical question of what chance an individual couple has of having a baby as a result of their decision to embark on an assisted-conception treatment cycle. In Australia, Kovacs and colleagues17 linked fresh and cryostored embryo transfer cycles in estimating the chance of a clinical pregnancy from a single round of egg retrieval and IVF treatment, conducted between 1993 and 1997. However, they did not perform analyses according to the age of the women. They found the chance of such a clinical pregnancy from one round of IVF treatment ranged up to 20.7%. From my study, the comparable statistic for 1998 is 42.3%. The overall chance of a baby was 34.9%, and there was a strong effect on fertility of increasing age (Box 1).
The inferences to be drawn from analysing the effects of maternal age on the chance of a live birth are notable. Firstly, there is a suggestion that live birth rates per egg retrieval among women in their early 20s could be particularly high. Although few women present with infertility before 26 years of age, data from egg donors also suggest higher live birth rates from women in their early 20s compared with later in their 20s.18,19 Live birth rates then plateau at just over 50% up to and including the age of 33 years.
After 33 years, there is an almost unremitting, linear decline in fertility to reach zero at 45 years, an age that is five to six years short of the median age at menopause in Australia.20 It has long been recognised that fertility fails well before the menopause. Modern demographers have relied chiefly on new interpretation of historical marriage and birth records to estimate normal human fertility.21 Records dating from the mid-19th century, for example, show relative fertility rates holding to about 1.0 until age 35 years, with rates of 0.90 from 35 to 39 years, 0.62 from 40 to 44 years, and 0.14 after 45 years.21 My data suggest that the decline in live births after IVF could be about two years in advance of this natural fall, which means that IVF per se should not be relied on to overcome female age-related infecundity. Interestingly, in egg donation programs in the United States (programs which aim to circumvent this decline for recipients), 33 years has been recommended as a cut-off age for the commercial recruitment of donors.19
The cause of the decline in the chance of live births with female age has been attributed to diminished oocyte cytoplasmic competence,22 part of the spectrum of which is an increase in meiotic spindle disturbances that manifest with aneuploidy.23 Most failing eggs in older women, however, do not carry an abnormal number of chromosomes,24 and the nature of the ooplasmic deficiency behind this physiological cause for miscarriages and sterility remains elusive.25
Of special social importance is the fact that, in 1998, the peak age of utilisation of IVF (39 years) was six years after the age at which the decline in fertility commenced. It is important for society to recognise that it might be preferable for childbearing to take place by the age of 33 years or women will increasingly encounter unexpected difficulties — difficulties that might or might not be overcome by IVF. Investigation and management of infertility should commence at an earlier age than it presently does.
In summary, live birth rates with modern IVF technology now exceed 50% per egg retrieval for women under the age of 34 years. The chance of a pregnancy that produces a live birth then falls steadily to become rare after 42 years of age. The community should be aware that, despite IVF, fertility ceases for an increasing proportion of women from their mid-30s.
1: Egg retrievals for in-vitro fertilisation (IVF), age of the women, and percentage of live births (95% CIs) from single IVF attempts
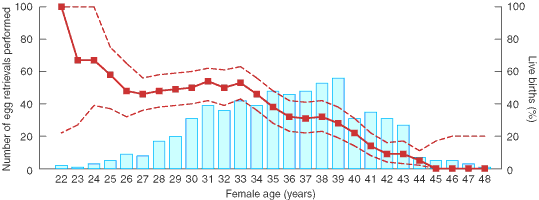
Histogram (bar graph and left axis) of female age at egg retrieval, with (superimposed) the percentage of single IVF attempts followed by at least one live birth (line graph and right axis), with interrupted line graphs indicating 95% CIs. Data smoothed by averaging over three years. Median age (36 years) is about one year older than the national average for IVF. Peak age of utilisation of IVF (39 years) is six years after the start of a linear decline in the chance of achieving a live birth.
2: Pregnancy outcome according to maternal age at egg retrieval; combined data for fresh and frozen embryo transfers
Age group (years) |
All clinical pregnancies |
Ectopic pregnancies |
Clinical miscarriage |
Immature deliveries and stillbirths |
Singleton live births |
Twin live births |
Births from cryostored embryos |
||||
< 35 |
156/252 |
3/156* |
16/153† |
5 |
102 |
30‡/132§ |
45/132 |
||||
(n = 252) |
(61.9%) |
(1.9%) |
(10.5%) |
|
|
(22.7%) |
(34.1%) |
||||
35–39 |
94/251 |
1/94* |
15/93† |
2 |
61 |
15/76§ |
25/76 |
||||
(n = 251) |
(37.5%) |
(1.1%) |
(16.1%)† |
|
|
(19.7%) |
(32.9%) |
||||
40–44 |
28/131 |
0/28 |
12/28† |
0 |
15 |
1/16§ |
4/16 |
||||
(n = 131) |
(21.4%) |
|
(42.9%) |
|
|
(6.3%) |
(25.0%) |
||||
Total¶ |
278/634 |
4/278* |
43††/274† |
7**†† |
178†† |
46††/224§ |
74/224 |
||||
(n = 634)†† |
(43.9%) |
(1.4%) |
(15.7%)† |
|
|
(20.5%) |
(33.0%) |
||||
* Ectopic pregnancy rate denominator is total pregnancies. † Miscarriage rate denominator is uterine pregnancies. ‡ Includes a triplet pregnancy electively reduced to twins by a feticidal operation. § Twin pregnancy rate denominator is live births. ¶ Excludes 14 women aged 45–48, none of whom conceived. ** Two singleton pregnancies (age of women, < 35 years) lost at 22 weeks; remaining five were twin pregnancies lost between 21 and 32 weeks. †† The overall clinical pregnancy rate per IVF treatment is calculated by dividing the miscarriages, immature births, stillbirths and live births (n = 274) into the total number of treatments (648), and is 42.3%. |
|||||||||||
Received 23 January 2002, accepted 21 January 2003
- Robert P S Jansen1
- Sydney IVF, Sydney, NSW.
I thank Jim Catt PhD and Michael Henman MSc(Med) for collecting clinical and laboratory data, Sally Muir SRN MSc(Med) and her staff for managing the clinical programs, and especially Didi Bower SRN for collating clinical cycle data. I also thank my colleagues at Sydney IVF, Drs Warwick Birrell CREI, Mark Bowman CREI, Nalini Gayer CREI, Farouk Mahmood FRANZCOG, Robert Lyneham CREI, Jeffrey Persson CREI, David Shelley-Jones FRANZCOG, Paul Sutherland FRANZCOG, and Iris Wang CREI, for helping with additional clinical and outcomes information; the partners and ultrasonographers of Sydney Ultrasound for Women for skilled ultrasound assessments before and during treatments, and in pregnancy; and Dr Andrew Speirs, Melbourne IVF, for help with logistic regression analysis of data and other statistical advice.
I am a Director of Sydney IVF Limited and own shares in the company. Sydney IVF embryo culture medium is licensed to William Cook (Aust.) Pty Ltd.
- 1. Hurst T, Shafir E, Lancaster P. Assisted conception Australia and New Zealand 1998 and 1999. AIHW cat. no. PER 16. Sydney: Australian Institute of Health and Welfare National Perinatal Statistics Unit (Assisted Conception Series no. 5), 2001.
- 2. Nygren KG, Andersen AN. European IVF monitoring programme. Assisted reproductive technology in Europe, 1998. Results generated from European registers by ESHRE. Hum Reprod 2001; 16: 2459-2471.
- 3. Society for Assisted Reproductive Technology and the American Society for Reproductive Medicine. Assisted reproductive technology in the United States: 1998 results generated from the American Society for Assisted Reproductive Medicine/Society for Assisted Reproductive Technology Registry. Fertil Steril 2002; 77: 18-31.
- 4. Jansen R. The clinical impact of in vitro fertilization. I. Results and limitations of conventional reproductive medicine. Med J Aust 1987; 146: 342-353.
- 5. Jansen R. Getting pregnant. A compassionate resource for overcoming infertility. Sydney: Allen & Unwin, 1997.
- 6. Schipper I, Hop WCJ, Fauser BCJM. The follicle-stimulating hormone (FSH) threshold/window concept examined by different interventions with exogenous FSH during the follicular phase of the normal menstrual cycle: duration, rather than magnitude, of FSH increase affects follicle development. J Clin Endocrinol Metab 2001; 83: 1292-1298.
- 7. Lalic I, Catt JW, Henman MJ, Muir S. Does short duration of sperm-oocyte incubation increase viability of embryos in human in vitro fertilization? Reprod Techn 2000; 10: 62-69.
- 8. Catt JW, Henman M. Toxic effects of oxygen on early human embryo development. Hum Reprod 2000; 15 (Suppl 2): 199-206.
- 9. Schenker JG, Polishuk WZ. Ovarian hyperstimulation syndrome. Obstet Gynecol 1975; 46: 23-28.
- 10. Gardner MJ, Altman DG. Statistics with confidence. London: The British Medical Journal, 1989.
- 11. National Health and Medical Research Council. Ethical guidelines on assisted reproductive technology. Canberra: AGPS, 1996.
- 12. Jansen RPS. Spontaneous abortion incidence in the treatment of infertility. Am J Obstet Gynecol 1982; 143: 451-473.
- 13. Lopata A, Johnston IW, Hoult IJ, Speirs AL. Pregnancy following intrauterine implantation of an embryo obtained by in vitro fertilization of a preovulatory egg. Fertil Steril 1980; 33: 117-120.
- 14. Hurst T, Lancaster P. Assisted conception Australia and New Zealand 1999 and 2000. AIHW cat. no. PER 18. Sydney: Australian Institute of Health and Welfare National Perinatal Statistics Unit (Assisted Conception Series no. 6), 2001.
- 15. Jansen RPS, Anderson JC, Birrell WRS, et al. Outpatient gamete intrafallopian transfer: 710 cases. Med J Aust 1990; 153: 182-188.
- 16. Jansen RPS. Relative infertility: modeling clinical paradoxes. Fertil Steril 1993; 59: 1041-1045.
- 17. Kovacs GT, MacLachlan V, Brehny S. What is the probability of conception for couples entering an IVF program? Aust N Z J Obstet Gynaecol 2001; 41: 207-209.
- 18. Cohen MA, Lindheim SR, Sauer MV. Donor age is paramount to success in oocyte donation. Hum Reprod 1999; 14: 2755-2758.
- 19. Faber BM, Mercan R, Hamacher P, et al. The impact of an egg donor's age and her prior fertility on recipient pregnancy outcome. Fertil Steril 1997; 68: 370-372.
- 20. Walsh RJ. The age of the menopause of Australian women. Med J Aust 1978; 2: 181.
- 21. Menken J, Trussell J, Larsen U. Age and infertility. Science 1986; 233: 1389-1394.
- 22. Jansen RPS. Older ovaries: ageing and reproduction. Med J Aust 1995; 162: 623-624.
- 23. Battaglia DE, Goodwin P, Klein NA, Soules MR. Influence of maternal age on meiotic spindle assembly in oocytes from naturally cycling women. Hum Reprod 1996; 11: 2217-2222.
- 24. Lim AST, Tsakok MFH. Age-related decline in fertility: a link to degenerative oocytes. Fertil Steril 1997; 68: 265-271.
- 25. Jansen RPS. Germline passage of mitochondria: quantitative considerations and possible embryological sequelae. Hum Reprod 2001; 15 (Suppl 5): 112-128.





Abstract
Objective: To determine the chance of at least one live birth from one round of in-vitro fertilisation (IVF) treatment and the effect of the woman's age on that likelihood.
Design: Retrospective analysis of outcomes from IVF treatment that did not involve donated gametes, but which included embryos cryopreserved in the retrieval cycle.
Setting and patients: All IVF patients (median age, 36 years; range, 22–48 years) who attended a private IVF clinic in Sydney for an egg retrieval between 1 January 1998 and 31 December 1998, and had embryo placements (fresh and cryostored) performed up to 30 June 2001.
Main outcome measure: Independently audited live births surviving the neonatal period.
Results: 565 women had 648 egg retrievals during the period. The age of peak utilisation of IVF was 39 years. For women aged 34 years or less, the chance of a live birth from one round of egg retrieval and IVF treatment was 52.4% (95% CI, 47%–59%). For women aged 35–44 years, there was a linear decline in the live birth rate, and no babies were born from retrievals at age 45 years and over. There was an age-dependent rise in the frequency of miscarriages, from 10.5% (95% CI, 5%–18%) for women under 35 years, to 16.1% (95% CI, 9%–25%) for those 35–39 years, and 42.9% [95% CI, 24%–63%] for those over 40 years (P < 0.001). A third of the first births resulted from embryo transfers performed after a period of cryostorage.
Conclusion: As fertility with IVF falls from the age of 34 years, and the age of peak IVF utilisation is 39 years, many Australian women are seeking IVF at an age when the likelihood of a live birth is reduced.