Herpes simplex virus (HSV; Box 1) infects the skin and neurones of the dorsal root ganglia, where it causes lifelong latent infection. The virus is often reactivated, leading it to spread down neuronal axons in spinal or trigeminal nerves and either to be shed asymptomatically in saliva (HSV-1) or genital secretions (HSV-2), or to cause disease of the skin, mucosa and occasionally major organs (Box 2).
Primary HSV-1 infection occurs when a susceptible person, usually a child, comes into close contact with a person with primary or recurrent infection. Primary infection in children is often asymptomatic, but can cause stomatitis severe enough to require hospitalisation. Symptoms are more common in adolescents and adults. Most HSV-1 seroconversions occur in the first five years of life, and by adulthood 80% of individuals have HSV antibodies. However, as HSV-1 seroprevalence is falling in many developed countries, risk of primary HSV-1 infection in adults is increasing. HSV-1 may also cause genital herpes; the increasing prevalence of this infection may be due to increasing orogenital contact.1
HSV-2 infection is usually acquired sexually in early adulthood, often in people with pre-existing HSV-1 infection. Thus, clinical infection with HSV-2 is often an initial rather than a primary infection (the latter occurs when an adult acquires HSV-2 in the absence of HSV-1 antibodies). HSV-2 usually causes genital herpes, but is also a rare cause of herpes labialis.2 The major influence on HSV-2 acquisition is the number of lifetime sexual partners, and women are generally infected at an earlier age than men.3 Most transmission occurs via asymptomatic viral shedding in skin or genital secretions.2 In Australia, the seroprevalence of HSV-2 in women is 14%.1
Both HSV-1 and HSV-2 infections recur often, with genital infections more likely to recur if caused by HSV-2 than HSV-1.
In primary HSV infection of the oropharynx, the most common manifestation is gingivostomatitis. Shallow ulcers form on the buccal mucosa and under the tongue (Box 3), and may also occur on the hard palate (which helps differentiate HSV infection from herpangina caused by coxsackie virus). These ulcers may be accompanied by fever and submandibular lymphadenopathy. Autoinoculation from the primary infection may occur elsewhere on the body (eg, herpetic whitlow).
Recurrent orolabial infections ("cold sores") may be triggered by stimuli such as fever, stress, cold, menstruation and ultraviolet radiation. Lesions usually occur on the vermilion border of the lips but may develop elsewhere on the face, including inside the nose. There is often a prodromal tingling or itching at the site of recurrence. Asymptomatic oral shedding of HSV is common and can transmit the virus.4 Lesions may be widespread in people with eczema and severe in those who are immunocompromised.
Symptomatic primary genital infection is moderately severe (more so in women) and lasts up to three weeks. Common signs include fever, dysuria, widespread ulceration, inguinal lymphadenopathy, malaise and pain. It may be accompanied by radiculomyelitis with urinary retention and neuralgia, and secondary bacterial infection. Perianal infections and proctitis may occur, especially in homosexual men. Genital infection often causes significant emotional and psychosexual disturbance.
About 20% of HSV-2-seropositive patients have overt recurrences of genital herpes; lesions are more limited in area and severity than in primary infection (Box 4). About 60% of HSV-2 seropositive patients have lesions but do not recognise their herpetic nature, especially if they are small or atypical, unless appropriately trained. The remaining 20% have true asymptomatic viral shedding. Genital HSV-2 recurrences usually last longer than oral HSV-1 recurrences and are more frequent in the six to 12 months after initial infection.3,4,5
This varies in prevalence around the world, from 1 in 2500 live births in the United States to 1 in 13 000 births in Australia,6-8 reflecting the wide variation in HSV-2 seropositivity in different regions and socioeconomic groups. Most neonatal HSV infection occurs after birth through an infected birth canal, although rarely it may occur in the postpartum period from direct contact or even from congenital infection. Most cases (about 70%) are due to HSV-2, although more HSV-1 neonatal disease may follow the current rise in HSV-1 genital infections.
Primary maternal genital herpes in the last trimester, particularly around the time of labour, leads about a third of babies to be infected. Neonatal HSV may also follow symptomatic or asymptomatic recurrences and, in fact, most babies with neonatal herpes are born to mothers with asymptomatic viral shedding. The clinical picture ranges from disease localised to the skin, eyes and mouth to encephalitis and disseminated disease (Box 5).6 Neonatal herpes may recur, necessitating long-term antiviral therapy.
HSV-1 keratitis may present with conjunctivitis or a unilateral dendritic (branching) ulcer. If recurrences are frequent and severe, they may involve deeper corneal layers, leading to interstitial or disciform keratitis and possible loss of vision.
Adult herpes encephalitis is a severe focal encephalitis caused by direct viral invasion of the brain (usually by HSV-1), typically in the frontotemporal and parietal areas. It occurs in all age groups but is uncommon — about 1 in 106 population per year.9
Immunocompromised people are at risk of severe HSV infection, including progressive and extensive oral and genital herpes and disseminated herpes.10
When possible, all patients with suspected herpes simplex requiring therapy (especially genital herpes) should have laboratory confirmation of infection, although this should not delay appropriate therapy.
Virus isolation by culture is the laboratory gold standard and works best when lesions are fresh and moist. It requires fresh vesicle fluid and cells to be collected from the base of a lesion using a rayon-tipped plastic swab, which is then transported in viral transport medium.
Direct antigen or genome detection methods are more rapid than virus isolation and more sensitive when lesions have begun to crust. Genome detection by polymerase chain reaction (PCR) is highly sensitive and is the technique of choice for examining cerebrospinal fluid in suspected encephalitis.11 It is also used increasingly in genital infection, and has confirmed that asymptomatic shedding from this infection is far more frequent than detected by virus culture and also more frequent than clinical recurrences.12 HSV can also be detected in tissue specimens (eg, brain biopsy) by microscopy and immunohistochemistry.
HSV antibody testing can reveal acute infection or previous exposure. Total HSV antibody seroconversion can be used to diagnose primary infection, but is not useful if the patient has already been infected with the other HSV type. HSV type-specific antibody testing is now possible using western blot or enzyme immunoassay. It allows accurate seroepidemiological surveys and confirmation of new infection in the presence of antibodies to the other virus (eg, HSV-2 genital infection against a background of HSV-1 antibodies from childhood herpes labialis13). This can aid risk assessment of HSV seroconversion in pregnancy and determine previous exposure to either virus.13 HSV type-specific IgM can be detected in acute infection, but may take up to 10–14 days to develop in primary infection and may also appear in severe clinical recurrences. Individual episodes of recurrent genital herpes cannot be reliably diagnosed by serological testing.
Antiviral agents for HSV infection are shown in Box 6.
Oral agents available in Australia for genital herpes comprise the nucleoside analogues aciclovir, valaciclovir and famciclovir. They are effective in acute first-episode genital herpes and also in recurrent genital herpes — as both episodic therapy (intermittent use at the onset of a recurrence) or suppressive therapy (continuous use to reduce the frequency of recurrences).14,15-17
Antiviral therapy speeds healing of lesions and, when used for suppression, reduces the frequency of recurrences. It also reduces virus shedding, but whether this reduces transmission is not proven. Antivirals do not prevent the development of neural latency. Early treatment — within 48–72 hours — is necessary for maximum effect, particularly with recurrences, and patients using episodic treatment should commence therapy during the prodrome.
Aciclovir has been used widely for over 20 years and has a good safety profile.14,18 Laboratory-confirmed resistance to this drug is very rare in the immunocompetent host;19 treatment failure is more likely to result from non-compliance or problems with drug absorption (eg, in diarrhoea).
The decision to use antivirals for suppressing recurrences of genital herpes must be made on an individual basis. The severity and number of recurrences, and their effect on the patient's physical and mental wellbeing, need to be assessed. More than six recurrences annually is a reasonable threshold to consider suppressive therapy for three to six months.
Antivirals are only one component of management of genital herpes. Patients with laboratory-confirmed infection also require:
Screening for other sexually transmitted diseases;
Counselling about the disease and associated psychosexual issues, including safer sexual practices, transmission risks, evaluation of sexual partners, pregnancy and childbirth; and
Education about the symptoms of recurrent infection, as many people do not recognise minor genital symptoms (eg, fissures and non-vesicular lesions and localised tingling or itching) as being caused by HSV.
People with genital herpes need accurate information and should be involved in management decisions, and, if necessary, referred to specialists or counsellors. More information on herpes is available at the Australian Herpes Management Forum website <http://www.ahmf.com.au>.
Vaccines for genital herpes are under development or in clinical trial.
Management of pregnant women with recurrent genital herpes is complex. The main issue is preventing transmission to the neonate during birth.4,20 In first-episode infection at the time of delivery, caesarean section is required and possibly also antiviral treatment. If typical recurrent genital lesions are present at delivery, then caesarean section is probably also indicated, but, if there are no lesions at delivery in a woman with a history of genital herpes, vaginal delivery is appropriate (case history, Box 7). Collecting virus cultures in the third trimester or at delivery to predict viral shedding is probably not worthwhile.21 Antiviral therapy has been advocated for patients with first-episode genital herpes in late pregnancy.20
In managing the neonate born to a mother with first-episode or recurrent genital herpes at the onset of labour, specimens should be collected from the baby for virus culture (eg, nasopharyngeal aspirate, conjunctival and umbilical swabs, urine and stool), and the baby treated with aciclovir if cultures are positive. Parents should be advised to report early signs of infection in the baby, such as lethargy, fever, poor feeding or skin lesions. In babies born to mothers with a history of genital herpes but no symptoms at delivery, routine virus culture is not recommended, but parents should again be advised to report early signs of infection. Routine antenatal screening for HSV antibodies is not indicated.
Varicella–zoster virus (VZV) is spread by the respiratory route and disseminates to lymph nodes and then via lymphocytes back to the skin, resulting in the rash of chickenpox. Like HSV, VZV infects the neurones of the dorsal root ganglia, where it causes lifelong latency. However, VZV reactivates much less often than HSV — in association with waning T-cell immunity — usually causing only one lifetime episode of herpes zoster (shingles). Nevertheless, VZV causes more severe damage to the nerve and dorsal root ganglia than HSV, leading to pain and often neural dysfunction. Prolonged pain may result from scarring of neural tissue.22
Most people in developed countries are infected with VZV in childhood, with 90% seropositive by adulthood. Herpes zoster can develop at any age, but the highest incidence is after 60 years. Overall, it occurs in 20% of the population, with more than one recurrence in 4%.22
Chickenpox is usually a mild disease in healthy children, but more severe in adults. It is often heralded by posterior cervical lymphadenopathy and fever, after an incubation period of two weeks. The rash is centripetal, being concentrated on the body rather than the limbs, and the lesions evolve through papular, vesicular and crusting stages, with lesions at different stages of evolution evident.
Complications include haemorrhagic lesions, cerebellitis, pneumonitis, arthritis and secondary infection (usually staphylococcal or streptococcal). Complications are rare in immunocompetent children, but about 50 times more common in adults (especially pneumonitis and hepatitis; Box 8). Visceral spread to lungs and liver occurs in 30%–50% of children with T-cell deficiency, with a mortality rate as high as 15%,23 compared with 0.1%–0.4% in healthy children.
A congenital varicella syndrome occurs after maternal chickenpox during pregnancy, at rates of 2%, if infection occurs during weeks 13–20 of gestation, and 0.4% if infection occurs before 13 weeks.24 Features of fetal infection include limb hypoplasia, dermatomal skin scarring and ocular and brain abnormalities.24 Severe neonatal infection can follow maternal chickenpox around the time of delivery.
This can involve any dermatome, including the lower sacral dermatome. However, as lower sacral dermatomal zoster is much less common than genital herpes, so-called "recurrent zoster" is usually recurrent HSV infection.
Prodromal neurological symptoms of herpes zoster comprise pain rather than the typical paraesthesiae of recurrent herpes simplex. The rash of zoster is often intensely pruritic and spreads throughout the dermatome, evolving through papular, vesicular and crusting stages (Box 9). It usually lasts two to four weeks. The most troubling symptom is usually pain, which ranges from mild to severe, and from burning to lancinating (case history, Box 10). Paraesthesiae, or anaesthesia and allodynia (pain induced by touch, often from trivial stimuli), can accompany severe pain. The pain may be self-limited or persist beyond the rash for up to a year ("postherpetic neuralgia").22,25 Another relatively common complication is zoster ophthalmicus (2%–4%), which follows involvement of the first division of the fifth cranial (trigeminal) nerve. It ranges from keratitis to the more severe iritis.26
In about 1% of immunocompromised people with herpes zoster, the virus spreads to the eye, brain or liver. Prolonged pain is uncommon. People with AIDS may have recurrent or prolonged zoster or multiple dermatomal involvement, and zoster in a person at risk of HIV may be an indicator of unrecognised HIV infection.27 Herpes zoster during pregnancy is not associated with intrauterine infection.24
Primary varicella infection and herpes zoster are usually diagnosed clinically. Laboratory diagnosis is recommended when the clinical picture is atypical or complicated, and to determine immune status to VZV in high-risk people.
Like HSV, VZV is detected in lesions by culture, antigen or genome detection, or by the antibody response to infection. However, VZV is more closely associated with cells than HSV, so a swab must scrape infected cells from the base of the lesion. Direct antigen detection by immunofluorescence can be performed within two hours,28 while genome detection by PCR is highly sensitive.
Antibody testing for VZV IgM and IgG can detect either acute infection or previous exposure. Measurement of VZV IgG can determine immunity to varicella, which is useful in determining if patients need zoster immunoglobulin after varicella exposure, or varicella vaccination. VZV IgM is seen in acute chickenpox and in about 70% of individuals with herpes zoster.
Aciclovir, valaciclovir and famciclovir have efficacy in acute herpes zoster and are used to ameliorate postherpetic neuralgia. If possible, they should be given within 72 hours of rash onset. Antiviral therapy is particularly indicated if postherpetic neuralgia is more likely (eg, patients aged over 50 years and those with severe pain in the prodrome or at presentation, or neurological abnormalities in the area involved).29,30 It is also required for immunocompromised patients and for ophthalmic zoster.
Valaciclovir and famciclovir are superior to aciclovir in reducing the incidence and duration of postherpetic neuralgia. The addition of corticosteroids is of no advantage in preventing postherpetic neuralgia. The pain associated with zoster should be treated aggressively with agents such as opioids and topical lignocaine and may require early referral to a pain clinic.22,25
Zoster immunoglobulin is indicated within 96 hours of significant exposure (eg, household or classroom contact) in people who are immunocompromised, susceptible pregnant women, neonates who are premature (less than 28 weeks' gestation or weight less than 1000 g) or whose mothers are susceptible to varicella or develop chickenpox within seven days before or after delivery.31
An effective live attenuated varicella vaccine is available and is highly effective against moderately severe and severe disease.32 It is given as a single dose to children aged 12 months to 12 years, and as two doses one to two months apart to people over 13 years or those who are immunocompromised. In Australia, the vaccine is recommended for vaccination of immunocompetent children from 12 months, but is not part of the standard immunisation schedule. It is especially indicated for non-immune people in the following categories: workers in high-risk occupations, women before pregnancy, parents of young children, and contacts of immunocompromised people.31
Evidence-based recommendations
Continuous antiviral therapy suppresses both clinical recurrences of genital herpes and asymptomatic shedding of HSV12,15,17,33 (E1).
Aciclovir therapy of recurrent genital herpes shortens lesion duration by one day16,18 (E2).
In herpes zoster, valaciclovir, famciclovir or aciclovir administered within 48–72 hours of rash onset shortens the median duration of zoster-associated pain and postherpetic neuralgia25,30,31 (E1).
2: Clinical syndromes caused by herpes simplex virus types 1 and 2 (HSV-1 and HSV-2)
HSV-1 |
HSV-2 |
||||||||||
Gingivostomatitis |
+++ |
– |
|||||||||
Genital lesions |
+ |
+++ |
|||||||||
Other cutaneous lesions |
++ |
++ |
|||||||||
Keratoconjunctivitis |
+++ |
– |
|||||||||
Retinitis |
+ |
– |
|||||||||
Oesophagitis |
+++ |
+ |
|||||||||
Pneumonitis |
+++ |
+ |
|||||||||
Hepatitis |
++ |
+ |
|||||||||
Meningitis |
+ |
+++ |
|||||||||
Adult encephalitis |
+++ |
+ |
|||||||||
Neonatal encephalitis |
+ |
++ |
|||||||||
Myelitis |
+ |
++ |
|||||||||
Erythema multiforme |
++ |
+ |
|||||||||
+++ = frequent. ++ = moderately frequent. + = uncommon. – = very rare. |
|||||||||||
3: Primary infection with herpes simplex virus type 1
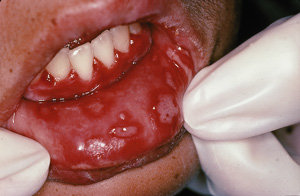
Mucosal ulceration and gingivitis typical of primary herpes simplex virus type 1 stomatitis in a child.
4: Genital herpes caused by herpes simplex virus type 2
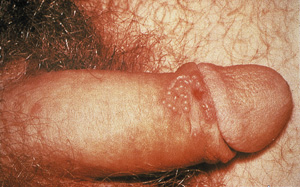
Recurrent genital herpes caused by herpes simplex virus type 2, showing typical small vesicles.
5: Neonatal infection with herpes simplex virus type 1
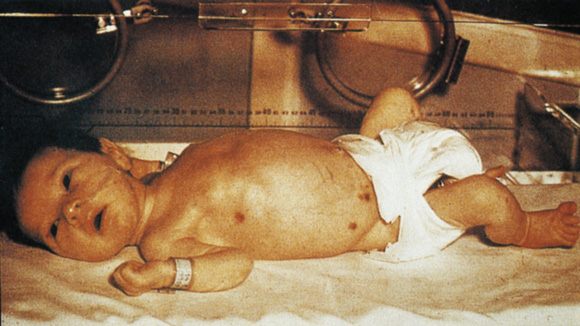
Neonatal infection with herpes simplex virus type 1, showing ulcerating and vesicular skin lesions. Lesions may be present in small numbers. Type 1 HSV was detected by direct immunofluorescence tests of cells collected from the base of a lesion. The virus was transmitted during birth.
6: Antiviral agents used to treat herpes simplex virus (HSV) and varicella zoster virus (VZV) infections
Infection |
Treatment |
||||||||||
Non-genital HSV (initial or recurrent) |
Aciclovir (200 mg orally 5 times daily for 5–10 days) or Aciclovir (5% topically 5 times daily for 5 days) (herpes labialis) or Aciclovir (3% ophthalmic ointment 5 times daily for 14 days) (HSV keratitis) |
||||||||||
Genital HSV (initial) |
Aciclovir (200 mg orally 5 times daily for 5–10 days) or Valaciclovir (500 mg orally twice daily for 5–10 days) |
||||||||||
Genital HSV (recurrent episode) |
As for initial genital HSV, or Famciclovir (125 mg orally 2 times daily for 5 days) |
||||||||||
Genital HSV (suppression of recurrences) |
Aciclovir (200 mg orally 3 times daily or 400 mg orally twice daily for up to 6 months) or Valaciclovir (500 mg orally 1–2 times daily for up to 6 months) or Famciclovir (250 mg orally twice daily for up to 6 months) |
||||||||||
Herpes zoster |
Aciclovir (800 mg orally 5 times daily for 7 days) or Valaciclovir (1000 mg orally 3 times daily for 7 days) or Famciclovir (250 mg orally 3 times daily for 7 days) |
||||||||||
Severe initial HSV, HSV encephalitis, or disseminated VZV |
Aciclovir (5–10 mg/kg intravenously 3 times daily for 7–21 days). Long-term aciclovir may be needed for recurrent encephalitis |
||||||||||
7: Case history — genital herpes in pregnancy
Presentation: A 25-year-old woman presented for routine antenatal care at 12 weeks' gestation in her first pregnancy. She had a history of recurrent genital herpes, with symptomatic episodes about every six months, but said that the lesions had never been swabbed. She was concerned about the effects of the infection on the baby.
Examination: Genital examination showed no abnormalities.
Investigations: Type-specific antibody testing showed antibodies to both herpes simplex virus types 1 and 2 (HSV-1 and HSV-2).
Management: The patient was advised to return if she had a clinical recurrence so that the lesions could be swabbed for viral culture. She was counselled that HSV transmission to the baby usually occurs at delivery, and that the risks of transmission are very low if no lesions are present during labour. If symptoms or signs of a recurrence occur at delivery, then a caesarean section is usually performed to minimise risk of transmission. The baby is closely monitored for HSV infection (including swabs taken for viral culture or polymerase chain reaction) and treated with antiviral therapy if there is evidence of this infection.
It is always important to confirm a clinical diagnosis of genital herpes.
Neonatal HSV infection occurs in 1 in 11 000–13 000 deliveries in Australia, with the risk highest in primary infection at delivery (about 33%) compared with recurrences (about 3%) and asymptomatic infection.
Clinical examination at the time of delivery is probably the best way to determine if there is active genital infection, athough asymptomatic viral shedding may occur.
Aciclovir therapy from 36 weeks of pregnancy can be considered in women with confirmed recurrent genital herpes.
8: Pneumonitis caused by varicella–zoster virus
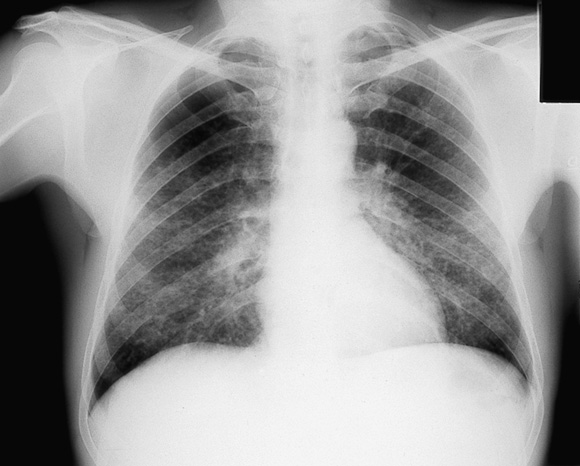
Chest x-ray showing interstitial pneumonitis in an otherwise healthy adult with chickenpox. Pneumonitis occurs in about 1 in 400 cases of adult infection and may be particularly severe in pregnancy.
9: Herpes zoster
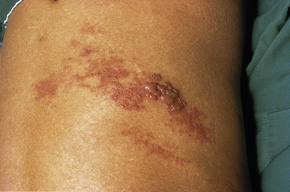 Typical papulovesicular rash of herpes zoster affecting thoracic dermatomes. |
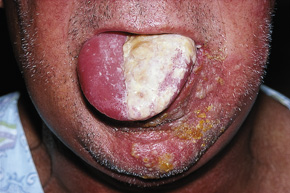 Herpes zoster affecting the third division of the fifth cranial (trigeminal) nerve, including the lingual nerve, in an elderly patient, showing necrotic tissue. |
10: Case history — delayed diagnosis of herpes zoster
Presentation: A 35-year-old woman with stage IV Hodgkin's disease in remission for over a year was referred from a regional hospital with severe right posterior thigh pain.
Investigations and course: During investigations for nerve root infiltration, including magnetic resonance imaging, an erythematous papulovesicular rash developed in the right L4–S1 dermatomal distribution. Pain worsened over the next few days and was associated with allodynia and patches of anaesthesia.
Diagnosis and management: Herpes zoster was diagnosed clinically. Intravenous aciclovir was begun 72 hours after the patient first noticed the rash, and halted its spread. However, pain remained extremely severe and required epidural anaesthesia for several days, followed by intramuscular narcotic analgesia.
Course: Pain subsided over the next three weeks, but persisted, together with allodynia, for five months, and required management at a pain clinic. Patches of anaesthesia slowly resolved.
This patient had severe, persistent pain associated with herpes zoster; ideally, antiviral therapy should have been started empirically as soon as lesions were noted. Severe pain before or at presentation is more likely to be associated with postherpetic neuralgia.
Postherpetic neuralgia is unusual in immunocompromised patients and, at worst, requires narcotic analgesia; corticosteroids do not help persistent pain and are not recommended for routine use in herpes zoster.
Laboratory confirmation of herpes zoster (eg, by virus culture, antigen or genome detction, or measurement of varicella–zoster IgM) is usually indicated when there is any doubt about the clinical diagnosis.
- Dominic E Dwyer1
- Anthony L Cunningham2
- 1 Centre for Infectious Diseases and Microbiology Laboratory Services, ICPMR, Westmead Hospital, Wentworthville, NSW.
- 2 Centre for Virus Research, Westmead Millennium Institute, Westmead Hospital, Wentworthville, NSW.
Both authors participate in clinical trials of antiviral agents and vaccines and sit on advisory boards for pharmaceutical companies that produce drugs and vaccines mentioned in this article.
- 1. Cunningham AL, Mindel AL, Dwyer DE. Global epidemiology of STD. In: Stanberry LR, Bernstein DI, editors. Sexually transmitted diseases, vaccines, prevention and control. New York: Academic Press, 2000.
- 2. Corey L, Wald A. Genital herpes. In: Holmes KK, Sparling PF, Mardh P-A, et al, editors. Sexually transmitted diseases. New York: McGraw-Hill, 1999: 285-312.
- 3. Johnson RE, Nahmias AJ, Magder LS, et al. A seroepidemiologic survey for the prevalence of herpes simplex virus type 2 infection in the United States. N Engl J Med 1989; 321: 7-12.
- 4. Stanberry L, Cunningham AL, Mindel A, et al. Prospects for control of herpes simplex virus disease through immunization. Clin Infect Dis 2000; 30: 549-566.
- 5. Koutsy LA, Stevens CE, Holmes KK, et al. Underdiagnosis of genital herpes by current clinical and viral isolation procedures. N Engl J Med 1992; 326: 1533-1539.
- 6. Brown ZA, Selke S, Zeh J, et al. The acquisition of herpes simplex during pregnancy. N Engl J Med 1997; 337: 509-515.
- 7. Mindel A, Taylor J, Seifert C, et al. Neonatal herpes prevention: A minor public health problem in some communities. Sex Transm Infect 2000; 76: 287-291.
- 8. Garland SM. Neonatal herpes simplex: Royal Women's Hospital 10-year experience with management guidelines for herpes in pregnancy. Aust N Z J Obstet Gynaecol 1992; 32: 331-334.
- 9. Whitley RJ, Cobbs CG, Alford CA Jr, et al. Diseases that mimic herpes simplex encephalitis: diagnosis, presentation and outcome. NIAD Collaborative Antiviral Study Group. JAMA 1989; 262: 234-239.
- 10. Siegal FP, Lopez C, Hammer GS, et al. Severe acquired immunodeficiency in male homosexuals, manifested by chronic perianal ulcerative herpes simplex lesions. N Engl J Med 1981; 305: 1439-1444.
- 11. Whitley RJ, Lakeman F. Herpes simplex virus infections of the central nervous system: therapeutic and diagnostic considerations. Clin Infect Dis 1995; 20: 414-420.
- 12. Wald A, Corey L, Cone R, et al. Frequent genital herpes simplex virus 2 shedding in immunocompetent women: effect of acyclovir treatment. J Clin Invest 1997; 99: 1092-1097.
- 13. Ashley RL. Sorting out the new HSV type-specific antibody tests. Sex Transm Infect 2001; 77: 232-237.
- 14. Stanberry L, Cunningham AL, Mertz G, et al. New developments in the epidemiology, natural history and management of genital herpes. Antiviral Res 1999; 42: 1-14.
- 15. Straus SE, Croen KD, Sawyer MH, et al. Acyclovir suppression of frequently recurring genital herpes. Efficacy and diminishing need during successive years of treatment. JAMA 1988; 260: 2227-2230.
- 16. Tyring SK, Douglas JM Jr, Corey L, et al. A randomized, placebo-controlled comparison of oral valaciclovir and acyclovir in immunocompetent patients with recurrent genital herpes infections. The Valaciclovir International Study Group. Arch Dermatol 1998; 134: 185-191.
- 17. Reitano M, Tyring S, Lang W, et al. Valaciclovir for the suppression of recurrent genital herpes simplex virus infection: A large-scale dose ranging study. J Infect Dis 1998; 178: 603-610.
- 18. Whitley RJ, Gnann JW. Acyclovir: A decade later. N Engl J Med 1992; 327: 782-789.
- 19. Safrin S. Treatment of acyclovir-resistant herpes simplex and varicella zoster virus infections. Adv Exp Med Biol 1996; 394: 59-66.
- 20. Scott LL, Sanchez PJ, Jackson GL, et al. Acyclovir suppression to prevent caesarean delivery after first episode genital herpes. Obstet Gynecol 1996; 87: 69-73.
- 21. Libman MD, Dascal A, Kramer MS, Mendelson J. Strategies for the prevention of neonatal infection with herpes simplex virus: a decision analysis. Rev Infect Dis 1991; 13: 1093-1104.
- 22. Cunningham AL, Dworkin RH. The management of post-herpetic neuralgia. BMJ 2000; 321: 778-779.
- 23. Arvin AM. Management of varicella-zoster virus infections in children. Adv Exp Med Biol 1999; 458: 167-174.
- 24. Enders G, Miller E, Cradock-Watson J, et al. Consequences of varicella and herpes zoster in pregnancy: prospective study of 1739 cases. Lancet 1994; 342: 1548-1551.
- 25. Dworkin RH, Carrington D, Cunningham AL, et al. Assessment of pain in herpes zoster: lessons learned from antiviral trials. Antiviral Res 1997; 33: 73-85.
- 26. Cohen JI, Brunell PA, Straus SE, Krause PR. Recent advances in varicella-zoster virus infection. Ann Intern Med 1999; 130: 922-932.
- 27. Whitley RJ, Gnann JW Jr. Herpes zoster in patients with human immunodeficiency virus infection — an ever expanding spectrum of disease. Clin Infect Dis 1995; 21: 989-990.
- 28. Rawlinson WD, Dwyer DE, Gibbons VL, Cunningham AL. Rapid diagnosis of varicella-zoster virus infection with a monoclonal antibody based direct immunofluorescence technique. J Virol Methods 1989; 23: 13-18.
- 29. Beutner KR, Friedman DJ, Forszpaniak C, et al. Valaciclovir compared with acyclovir for improved therapy for herpes zoster in immunocompetent adults. Antimicrob Agents Chemother 1995; 39: 1546-1553.
- 30. Tyring S, Barbarash RA, Nahlik JE, et al. Famciclovir for the treatment of acute herpes zoster: Effects on acute disease and associated post-herpetic neuralgia. Ann Intern Med 1995; 123: 89-96.
- 31. National Health and Medical Research Council. The Australian immunisation handbook. 7th ed. Canberra: AGPS, 2000.
- 32. Vazquez M, LaRussa PE, Gershon A, et al. The effectiveness of the varicella vaccine in clinical practice. N Engl J Med 2001; 344: 955-960.
- 33. Diaz-Mitoma F, Sibbald RG, Shafran SD, et al. Oral famciclovir for the suppression of recurrent genital herpes. A randomized controlled trial. JAMA 1998; 280: 887-892.





Abstract
Any new patient with suspected genital herpes should have diagnostic testing with virus identification.
Type-specific serological tests that distinguish between antibodies for type 1 and type 2 herpes simplex virus (HSV) may be useful to determine previous exposure but cannot be used to diagnose recurrences of genital herpes.
Initial episodes of genital herpes usually require antiviral therapy, while recurrences may be treated with continuous antiviral suppression (if frequent) or episodic therapy; patient counselling and education (including how to recognise lesions) are essential.
Topical or systemic therapy is available for initial and recurrent non-genital herpes simplex.
Primary varicella infection (chickenpox) and herpes zoster (shingles) are usually diagnosed clinically, but can be confirmed by detection of varicella–zoster virus antigens or nucleic acid from swabs of lesions or by antibody tests.
Antiviral therapy should be considered in chickenpox if disease is complicated or the patient is immunocompromised.
In herpes zoster, antiviral therapy should be given within 72 hours of onset to patients aged over 50 years or with severe pain or neurological abnormalities to reduce the likelihood and duration of postherpetic neuralgia.