The difference between treatment recommended on the basis of clinical trials and that which actually occurs in the community has often been referred to as the "treatment gap".1 Evidence from international surveys indicates that only a minority of patients with coronary heart disease (CHD) achieve the target levels for their particular modifiable risk factors or even receive treatment.2-5 There are no published data available for Australia.
In this observational study we measured the treatment gap in patients with CHD who completed follow-up in the control groups of two randomised controlled trials of a risk-factor intervention called the "Coach Program", conducted in 1996–1998 and 1999–2000, respectively.6-7 Here, we compare the treatment gap in Victoria within these two time periods and contrast this gap with that in the United Kingdom,2 Europe3-4 and the United States.5
VIC-I represents the survey of the usual-care (control) group in the first study, conducted in 1996–1998.6 VIC-II represents the survey of the usual-care (control) group in the second study, conducted in 1999–2000.7 These patients represent a full spectrum of patients from different metropolitan areas, socioeconomic groups and rural areas. VIC-I included patients from the cardiology and cardiothoracic units of a single tertiary referral hospital in Melbourne. These patients had been hospitalised for revascularisation procedures — either coronary artery bypass grafting (CABG) or percutaneous coronary intervention (PCI). Patients were excluded if they were aged over 75 years or if they were immobilised by conditions such as severe heart failure or multiple sclerosis. VIC-II included patients from the cardiology and cardiothoracic units of six teaching hospitals in Melbourne. Patients were included if they had been hospitalised for CABG, PCI, postacute myocardial infarction or unstable angina and then discharged on medical therapy, or for coronary angiography with planned (elective) revascularisation. Patients were excluded if they were not contactable by telephone; could not speak or read English; had no fasting blood sample taken within 24 hours of hospitalisation; were participating in another study involving lipids; lived too far away from or were unwilling to travel to the hospital for follow-up visits; were too ill in hospital for interview; or would not provide signed consent.
The study protocols were approved by the human research ethics committee of each centre.
Data in VIC-I were collected from July 1996 to March 1997 by a dietitian. The patients in VIC-I were contacted twice after random allocation: once after two weeks to ask them how they were, and once after 24 weeks to ask them to obtain a fasting serum lipid level profile within the next two weeks and to document reported use of lipid-lowering medication.
Data in VIC-II were collected from May 1999 to April 2000 by four nurses and two dietitians (one health professional per hospital). VIC-II patients were discharged with a one-page chart of risk-factor targets for the secondary prevention of CHD, which was attached to their hospital discharge summary. This discharge summary and chart were given to the patient's usual medical carer by the patient after discharge. Patients were contacted only once after discharge, at 24 weeks to arrange a follow-up assessment within the next two weeks. The following data were obtained at the six-month interview: fasting serum lipid level profile, fasting glucose level; blood pressure and body weight measurement; dietary intake determined by means of a validated food-frequency questionnaire,8 self-reported smoking behaviour, alcohol intake, physical activity, performance of cardiac rehabilitation, and use of lipid-lowering medication.
Venous blood was drawn for the biochemical measurements. Low-density lipoprotein cholesterol (LDL-C) concentration was calculated by the Friedewald equation, except when the triglyceride concentration exceeded 4.5 mmol/L.9 Analyses were performed by one central commercial laboratory in VIC-I and within each hospital's own pathology laboratory in VIC-II. Blood pressure was measured in a sitting position. Two measurements were taken, and the lower systolic and diastolic measurements were used. Weight was measured on electronic scales in light indoor clothes without shoes. The height used for calculation of body mass index (BMI) was recorded from the patient's medical history.
Risk factors for Victorian patients were compared against the following Australian target levels for modifiable risk factors: total cholesterol concentration (TC), < 4.0 mmol/L;10 TC, < 4.5 mmol/L;11 complete smoking cessation; blood pressure, < 140/90 mmHg;12 fasting blood glucose level (FBG), < 7.0 mmol/L;13 BMI < 25 kg/m2;14 saturated fat intake ≤ 8% total energy intake;12 alcohol intake, not more than two standard drinks per day for women, and four standard drinks per day for men;12 and 30 minutes or more of moderate-intensity activity on most or all days of the week.12 International comparisons were made, where available, with UK (ASPIRE),2 European (EUROASPIRE I and II)3-4 and US (L-TAP)5 studies.
The treatment gap, expressed as a percentage, is 100%, minus the percentage of patients achieving the target level for a particular modifiable risk factor.
The 95% confidence intervals for the lipid results were calculated with Stata.15 The number of patients achieving the lipid targets in VIC-I and VIC-II were compared with χ2 tests.
Of the 124 patients assigned to the control group in the first study, 112 (90.3%) completed the trial (VIC-I) and 348 of the 398 patients assigned to the control group in the second study (87.4%) completed the trial (VIC-II). In this report, comparisons are based only on the data obtained from patients who attended the six-month assessment.
Box 1 shows the number of patients followed for six months by diagnostic category, sex, age and place of residence. It should be noted that all the patients studied in VIC-I had myocardial revascularisation, compared with only half of the patients in VIC-II (P = 0.0001). In VIC-I 60% of patients lived rurally or interstate, compared with 13% in VIC-II (P = 0.0001).
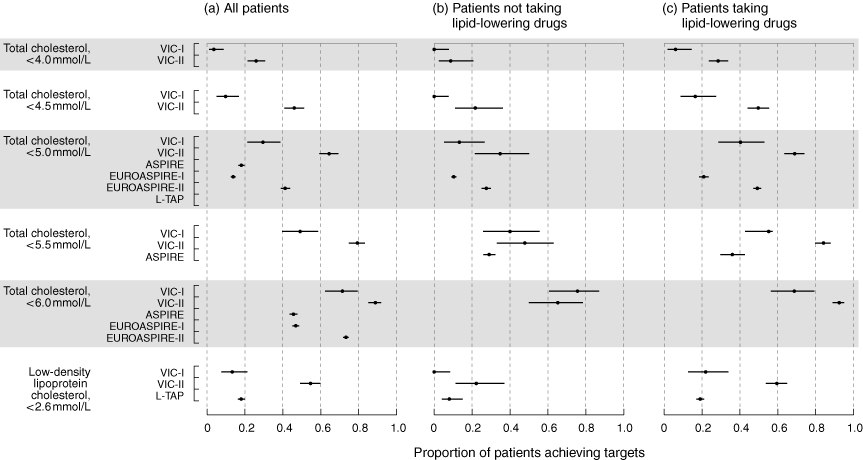
Box 2 compares the proportion of patients achieving lipid targets in the two Victorian studies, and the four overseas studies.2-5
Box 2 (a) shows that there was a reduction in the treatment gap from VIC-I to VIC-II by both the new (TC, < 4.0 mmol/L10) or old (TC, < 4.5 mmol/L11) National Heart Foundation (NHF) criteria. Thus, the treatment gap declined from 96.4% (95% CI, 91%–99%) to 74.1% (95% CI, 69%–79%) for TC < 4.0 mmol/L (P = 0.0001) and from 90.2% (95% CI, 83%–95%) to 54.0% (95% CI, 49%–59%) for TC < 4.5 mmol/L (P = 0.0001). However, almost three-quarters of patients are still not achieving the target TC level of < 4.0 mmol/L.
It should be noted that no patient achieved a TC level of < 4.0 mmol/L without medication in VIC-I and only 8.7% reached this level without medication in VIC-II (Box 2 [b]). Similarly, no patient achieved a TC level of < 4.5 mmol/L in VIC-I without medication and only 21.7% of patients reached this target in VIC-II without medication. Even when lipid-lowering medication was prescribed, most patients did not achieve the target (Box 2 [c]).
For comparison with overseas studies, treatment gaps for a TC level < 5.0 mmol/L were 70.5% for VIC-I, 35.6% for VIC-II, 82% for ASPIRE, 86.2% for EUROASPIRE I and 58.8% for EUROASPIRE II. The treatment gap for an LDL cholesterol level < 2.6 mmol/L was 86.7% in VIC-I, 45.4% in VIC-II and 82% in L-TAP.
As shown in Box 3, the use of lipid-lowering medication increased from 59.8% in VIC-I to 86.8% in VIC-II. Statins accounted for 92.5% of all lipid-lowering drugs in VIC-I and 97.4% in VIC-II. Similarly, there was an increase in the use of lipid-lowering drugs from 32.0% in EUROASPIRE I to 62.9% in EUROASPIRE II, with statins accounting for 57.8% and 91.7%, respectively. In L-TAP 92.6% of patients were prescribed lipid-lowering drugs, with statins accounting for 76.6%. Use of lipid-lowering drugs was lowest in ASPIRE (15.7%).
In VIC-I and VIC-II the same median doses of statins were used to achieve all the lipid thresholds shown in Box 2 (a) and (c). There was no evidence that the statin doses were titrated up to achieve target lipid levels.
In VIC-II, 127 of the 342 patients (37.1%) had high systolic blood pressure (≥ 140 mmHg) and 72 (21.1%) had high diastolic blood pressure (≥ 90 mmHg). The number of patients with systolic blood pressure of ≥ 140 mmHg or diastolic blood pressure ≥ 90 mmHg, or both, in VIC-II was 135 (39.5%). This compares with more than half the patients in EUROASPIRE I (55.4%) and II (53.9%). Similarly, the proportion of patients in VIC-II with blood pressure ≥ 160/95 mmHg was 6.7% (23), compared with 27.6% in EUROASPIRE I and 28.0% in EUROASPIRE II. In ASPIRE, 17.8% (349/1966) of patients had systolic blood pressure ≥ 160 mmHg and 26.4% (518/1961) had diastolic pressure ≥ 90 mmHg.
In VIC-II the prevalence of self-reported diabetes mellitus at baseline was 17.5% (61/348). At six months, the fasting blood glucose concentration (FBG) in 43 (70.5%) of these 61 patients was ≥ 7.0 mmol/L. Twenty-eight patients who did not report diabetes at baseline were found to have diabetes, defined by an FBG ≥ 7.0 mmol/L. The prevalence of diabetes at six months (defined by FBG ≥ 7.0 mmol/L or self-reported diabetes at baseline) was 25.6% (89/348).
In VIC-II, 14.2% (49/346) of patients had impaired fasting glucose levels (> 6.0 to < 7.0 mmol/L), while 65.3% (226/346) had normal fasting glucose levels (≤ 6.0 mmol/L).
The prevalence of self-reported diabetes mellitus was 18.0% (641/3569) in EUROASPIRE I, 21.9% (740/3379) in EUROASPIRE II and 23.6% (344/1460) in L-TAP.
The proportion of patients who were overweight (BMI ≥ 25 kg/m2) was 75.1% (257/342) in VIC-II and 72.3% (1419/1962) in ASPIRE. Obesity (BMI ≥ 30 kg/m2) affected 29.8% (102/342) of patients in VIC-II, 27.4% (544/1982) in ASPIRE, 25.3% (900/3557) in EUROASPIRE I and 32.8% (1101/3356) in EUROASPIRE II.
Prevalence of self-reported smoking was 14.7% (51/348) in VIC-II, 19.1% (378/1974) in ASPIRE, 19.4% (693/3569) in EUROASPIRE I and 20.8% (703/3379) in EUROASPIRE II.
VIC-II shows that high dietary fat intake (> 30% of energy) was reported in 203 of 336 patients (60.4%), with a high saturated fat intake (> 8% of energy) in 273 (81.3%), and high dietary cholesterol intake (> 300 mg) in 26 (7.7%).
In VIC-II 90 of 348 patients (25.9%) said they were not walking regularly for exercise. Of the 258 who reported walking regularly, 92 (35.7%) reported walking less than five times per week.
In VIC-II, 29.1% (101/347) of patients reported consuming more than two alcoholic drinks daily; 16.0% (42/263) of men reported consuming more than four standard drinks a day, and 15.5% (13/84) of women reported consuming more than two standard drinks a day.
Half of patients in VIC-I (56/112), compared with 42.8% (149/348) of patients in VIC-II, did not attend a cardiac rehabilitation program.
The data from the two Victorian surveys indicate that there has been a marked reduction in the treatment gap for lipid targets between 1996–1998 and 1999–2000. This may be attributable to better uptake of lipid-lowering drug therapy based on evidence from trials, a change in Pharmaceutical Benefits Scheme prescribing criteria for lipid-lowering drugs, and extensive sponsored medical education from the pharmaceutical industry. Also, the usual medical carer for each patient in VIC-II was provided with a one-page chart of goals for secondary prevention of CHD. Providing this chart was not usual practice and may have had an enhancing effect on usual care.
We have shown that many more patients in VIC-II achieved the NHF total cholesterol level targets of < 4.0 mmol/L10 (current target levels) or < 4.5 mmol/L11 (target levels at the time of the study) compared with VIC-I. We found that the use of lipid-lowering medication increased from 59.8% in VIC-I to 86.8% in VIC-II, although median doses of statins remained the same. Further, Box 2 (b) indicates that few patients achieved the recommended targets without the use of drugs and there was no improvement between VIC-I and VIC-II.
We therefore conclude that the reduction in the treatment gap between VIC-I and VIC-II has been the result of an increase in the number of patients prescribed lipid-lowering drugs, rather than an increased dose of these drugs or better nutritional counselling provided by usual medical care. Titration of statins and better advice on nutrition appear to be the next important focus for the achievement of lipid targets.
We have concentrated on lipid data in this report. However, the results for VIC-II for the other modifiable risk factors show that these also need more attention. Large treatment gaps exist for risk factors such as high blood pressure (≥ 140/90 mmHg), diabetes (FBG level, ≥ 7.0 mmol/L), overweight (BMI, 25 to < 30 kg/m2) and obesity (BMI ≥ 30 kg/m2), smoking, physical activity, dietary intake of saturated fat (> 8% of total energy intake) and alcohol intake.
One way of helping patients achieve their risk-factor targets for secondary prevention is the Coach Program, an aggressive training program for patients with CHD whereby a health professional coach trains patients to "take ownership" of their risk-factor levels and to "drive" the process of achieving and maintaining the target levels, while working in association with their doctors. The program has been tested,6-7 and the results have shown that coaching has had a major impact on these risk factors and may be the bridge across the treatment gap.
1: Participation rates, by clinical characteristics and place of residence
Number of patients |
|||||||||||
First survey (VIC-I) |
Second survey (VIC-II) |
||||||||||
Diagnosis |
|||||||||||
Coronary artery bypass grafting |
62 (55.4%) |
34 (9.8%) |
|||||||||
Percutaneous coronary intervention |
50 (44.6%) |
142 (40.8%) |
|||||||||
Medical therapy |
— |
128 (36.8%) |
|||||||||
Coronary angiography |
— |
44 (12.6%) |
|||||||||
Sex |
|||||||||||
Male |
84 (75.0%) |
264 (75.9%) |
|||||||||
Female |
28 (25.0%) |
84 (24.1%) |
|||||||||
Age at index event |
|||||||||||
≤60 years |
54 (48.2%) |
193 (55.5%) |
|||||||||
> 60 years |
58 (51.8%) |
155 (44.5%) |
|||||||||
Place of residence |
|||||||||||
Metropolitan |
45 (40.2%) |
302 (86.8%) |
|||||||||
Rural and interstate |
67 (59.8%) |
46 (13.2%) |
|||||||||
2: Proportion of all patients, those not taking lipid-lowering drugs and those taking lipid-lowering drugs who achieved cholesterol and low-density lipoprotein cholesterol level targets: international comparison
VIC-I6 and VIC II7 are the two Victorian studies under consideration. ASPIRE (UK),2 EUROASPIRE I and II (European)3-4 and L-TAP (US)5 are studies used for international comparison.
3: Use of lipid-lowering medication (number and % of patients): international comparison
VIC-I |
VIC-II |
ASPIRE |
EUROASPIRE I |
EUROASPIRE II |
L-TAP |
||||||
67 (59.8%) |
302 (86.8%) |
311 (15.7%) |
1143 (32.0%) |
2125 (62.9%) |
1352 (92.6%) |
||||||
Received 29 August 2001, accepted 11 January 2002
- Margarite J Vale1
- Michael V Jelinek2
- James D Best3
- on behalf of the COACH study group
- 1 The COACH Study: Coaching patients on Achieving Cardiovascular Health.
- 2 Department of Cardiology and the University of Melbourne Department of Medicine, St Vincent's Hospital, Melbourne, VIC.
- 3 The University of Melbourne Department of Medicine, St Vincent's Hospital, Melbourne, VIC.
We gratefully acknowledge the invaluable assistance of Dr John Sanatamaria in creating the graphs for presenting our lipid data with international comparisons. This study was supported by an unconditional project grant from Merck Sharp and Dohme (Australia) Pty Limited and by a research program grant awarded by VicHealth (Victorian Health Promotion Foundation). The study sponsors had no part in the study design, data collection, data analysis, interpretation or in the writing of the manuscript. The dietary survey (VIC-II) was supported by a project grant from the Research and Grants Unit, St Vincent's Hospital Melbourne. This study was presented at the 49th Annual Scientific Meeting of the Cardiac Society of Australia and New Zealand, Auckland, New Zealand, 5–8 August 2001.
None declared.
- 1. Pearson TA, Peters TD. The treatment gap in coronary artery disease and heart failure: community standards and the post-discharge patient. Am J Cardiol 1997; 80(8B): 45H-52H.
- 2. Bowker TJ, Clayton TC, Ingham JE, et al. A British Cardiac Society survey of the potential for secondary prevention of coronary disease: ASPIRE (Action on Secondary Prevention through Intervention to Reduce Events). Heart 1996; 75: 334-342.
- 3. The EUROASPIRE Study Group. A European Society of Cardiology survey of secondary prevention of coronary heart disease: principal results. Eur Heart J 1997; 18: 1569-1582.
- 4. EUROASPIRE II Steering Group. Clinical reality of coronary prevention guidelines: a comparison of EUROASPIRE I and II in nine countries. Lancet 2001; 357: 995-1001.
- 5. Pearson TA, Laurora I, Chu H, Kafonek S. The lipid treatment assessment project (L-TAP). A multicenter survey to evaluate the percentages of dyslipidaemic patients receiving lipid-lowering therapy and achieving low-density lipoprotein cholesterol goals. Arch Intern Med 2000; 160: 459-467.
- 6. Vale MJ, Jelinek MV, Best JD, Santamaria JD. Coaching patients with coronary heart disease to achieve the target cholesterol: a method to bridge the gap between evidence-based medicine and the "real world". Randomized controlled trial. J Clin Epidemiol 2002; 55: 245-252.
- 7. Vale MJ, Jelinek MV, Best JD, et al, for the COACH study group, Melbourne, Victoria. Multicenter randomized controlled trial of coaching patients on achieving cardiovascular health (COACH); a proven method for achieving risk factor targets in patients with coronary heart disease [abstract]. Circulation 2001; 104 Suppl: II-391.
- 8. Ireland P, Jolley D, Giles G, et al. Development of the Melbourne FFQ: A food frequency questionnaire for use in an Australian prospective study involving an ethnically diverse cohort. Asia Pacific J Clin Nutr 1994; 3: 19-31.
- 9. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low density lipoprotein cholesterol in plasma, without the use of the preparative centrifuge. Clin Chem 1972; 18: 499-502.
- 10. Lipid management guidelines 2001. National Heart Foundation of Australia, The Cardiac Society of Australia and New Zealand. Med J Aust 2001; 175 (5 November Suppl.): S57-S85.
- 11. Guide for the use of lipid-lowering drugs in adults. Position statement prepared by the Nutrition and Metabolism Advisory Committee, National Heart Foundation of Australia. Melbourne: NHFA, 1998.
- 12. National Heart Foundation of Australia homepage. <http://www.heartfoundation.com.au>.
- 13. Zimmet P, Alberti G, de Courten MP. New classification and criteria for diabetes: moving the goalposts closer. Med J Aust 1998; 168: 593-594.
- 14. Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee. Technical report series 854. Geneva: World Health Organization, 1995.
- 15. Stata statistical software [computer program]. Release 6.0. College Station, TX: Stata Corporation, 1999.





Abstract
Objectives: To determine the proportion of patients with established coronary heart disease (CHD) in two Australian studies (VIC-I in 1996–1998, and VIC-II in 1999–2000) who achieved their risk-factor targets as recommended by the National Heart Foundation of Australia, and to compare this proportion with those in studies from the United Kingdom (ASPIRE), Europe (EUROASPIRE I and II) and the United States (L-TAP).
Design and setting: Prospective cohort study with VIC-I set in a single Melbourne university teaching hospital and VIC-II set in six university teaching hospitals in Melbourne, Victoria.
Participants: 460 patients (112 in VIC-I, 348 in VIC-II) who completed follow-up in the control groups of two randomised controlled trials of a coaching intervention in patients with established CHD.
Main outcome measures: The treatment gap (100%, minus the percentage of patients achieving the target level for a particular modifiable risk factor) at six months after hospitalisation.
Results: The treatment gap declined from 96.4% (95% CI, 91%–99%) to 74.1% (95% CI, 69%–79%) for total cholesterol concentration (TC) < 4.0 mmol/L (P = 0.0001) and from 90.2% (95% CI, 83%–95%) to 54.0% (95% CI, 49%–59%) for TC < 4.5 mmol/L (P = 0.0001). This reduction in the treatment gap between VIC-I and VIC-II appears to be entirely explained by an increase in the number of patients prescribed lipid-lowering drugs. The treatment gaps in the UK and two European studies were substantially greater. The treatment gap for blood pressure (systolic ≥ 140 mmHg and/or diastolic ≥ 90 mmHg) in VIC-II was 39.5%, again less than corresponding European data. There were 8.1% of patients who had unrecognised diabetes in VIC-II (fasting glucose level ≥ 7 mmol/L), making a total of 25.6% of VIC-II patients with diabetes, self-reported or unrecognised. The proportion of patients in VIC-II who were obese (body mass index ≥ 30 kg/m2) was similar to the overseas studies, while fewer patients in VIC-II smoked compared with those in the UK and European studies.
Conclusions: A substantial treatment gap exists in Victorian patients with established CHD. The treatment gap compares well with international surveys and, at least in the lipid area, is diminishing.