Attitudes to and use of a modified prescription form by general practitioners and pharmacists
Merilyn J Liddell and Sue P Goldman
MJA 1998; 168: 322-325
For editorial comment see Roberts
Abstract -
Introduction -
Methods -
Results -
Discussion -
Acknowledgements -
References -
Authors' details
-
-
©MJA1998
Abstract |
Objective: To evaluate the rate of use and acceptance
of a new prescription form designed to provide more information to
pharmacists and patients. Design: Prospective descriptive study. Setting: A semi-rural community outside Melbourne, Victoria, in November 1994. Participants: GPs and pharmacists working three or more sessions per week in the locality, and 21 consumers who formed two consumer focus groups. Intervention: An education session for GPs and pharmacists, followed by a one-month trial of new prescription forms which included notations to facilitate interprofessional communication. Main outcome measures: Rate of use and acceptability of the new prescription notations. Results: Eighteen GPs and 10 pharmacists participated; 3600 forms were issued and 2521 prescriptions, including 3464 prescription items, were analysed. GPs and pharmacists used at least one of the new prescription notations in 45% (1559) of prescription items; 35% of prescription items (1222) were notated with the general purpose of the medication. Qualitative findings suggested that GPs, pharmacists and consumers considered the new prescription form to be beneficial and useful. Conclusions: A modified prescription form to increase communication between GPs and pharmacists is acceptable in clinical practice. |
Introduction |
Over recent years professional standards for prescribing have
emphasised the need for a more patient-centred approach,1 as shown in such documents as the
National Health and Medical Research Council's guidelines for
medical practitioners on providing information to patients,2 the Policy on the Quality Use of
Medicines developed by the Commonwealth Government,3 and regulations on the provision of
written consumer information with dispensed medication.4 Research on how to provide more
patient-centred pharmaceutical information and counselling shows
that closer collaboration between general practitioners (GPs) and
pharmacists would help to ensure that medication information is both
comprehensive and relevant for consumers.5,6 A joint working party of the Royal Australian College of General Practitioners (RACGP) Victoria Faculty and the Victorian Branch of the Pharmaceutical Society of Australia (PSA) was formed in 1993 to consider how to improve communication between GPs, pharmacists and consumers. A series of recommendations were developed and endorsed by the national executives of the RACGP and the PSA, and published in a joint statement.7 The major recommendation was the use of a new prescription form to aid communication between GPs, pharmacists and patients. Here, we describe a pilot study to determine the rate of use of, and the attitudes of GPs, pharmacists and patients to, a modified prescription form for use in normal clinical practice. No similar study has been reported in the literature (based on the following searches: MEDLINE, January 1982 - June 1997; International Pharmaceutical Abstracts, January 1970 - December 1996; HEALTH, January 1975 - December 1996; EMBASE, January 1994 - December 1996; Pharmaceutical News Index, January 1974 - December 1996). |
Methods |
Ethical approval for this study was received from the Monash
University Standing Committee on Ethics in Research on Humans and all
subjects gave informed consent for their par ticipation. The study
was conducted in November 1994 in a semi-rural community on the
outskirts of Melbourne with a population of 25 000.
All 24 GPs and 13 pharmacists working three or more sessions per week in the locality were invited to participate; 18 GPs and 10 pharmacists agreed. Twenty-one consumers from existing heart and arthritis support groups at the local community health centre were recruited for two focus groups of eight and 13 participants. Before the intervention, the 18 participating GPs and 10 pharmacists completed separate questionnaires (29 questions for pharmacists and 28 for GPs), which covered demographics and assessed (on a scale of 1 to 5) attitudes to professional roles, interprofessional collaboration and consumers' needs for medication information and counselling. |
Intervention |
The intervention began with an educational session for GPs and
pharmacists to introduce the modified prescription form on which GPs
could notate the purpose of prescribed medications, changes to
existing medications and specific patient needs. Some of these
notations were unprompted, requiring freehand additions to the
prescription, while others were prompted, appearing as
abbreviations on the form which could be circled when appropriate
(see Box 1). A glossary of the new prescription notations was
provided. Box 1 shows the glossary which was provided to pharmacists.
During the education sessions, the new notations were explained as
follows. The major change was that GPs were encouraged, where they
considered it appropriate, to indicate the purpose of each
prescribed medication as part of the written directions. For
example, GPs were also encouraged to indicate to the pharmacist if therapy with an ongoing medication was to cease (enabling pharmacists to update their computer records to show that a drug was no longer to be taken, and attempt to withdraw any unpresented repeats from circulation), or if the directions intentionally specified an unusual dosage or quantity to be dispensed. Finally, GPs could circle abbreviations printed on the prescription form to indicate specific patient needs or changes in the medication regimen. Pharmacists could then check the patient had understood changes to directions and amend the computer records accordingly. At the end of the educational session, 200 triplicate prescription forms were issued to each GP for use over one month. The third copy of each prescription dispensed (minus identifying patient details) was collected to determine the use of the new notations. After one month, individual one-hour open-ended, semi-structured interviews were conducted with all participating GPs and pharmacists, covering their overall views on the reformatted prescription, specific issues of practical application and suggested changes. Two separate one-and-a-half-hour focus group discussions with consumers dealt with aspects of the use of the reformatted prescription which would directly affect patients. |
Data analysis | For frequency analysis of the questionnaires and of the prescription data, we used, respectively, the statistical package Genstat8 and the computer database program Access.9 Transcripts of interview data and focus group discussions were coded according to emerging themes around the prescription format and particular notations. The analysis, with the NUDIST program for qualitative analysis,10 concentrated on direct testimonies of views held about the new prescription form. |
Results | The median age of the 18 GPs was 35 years (range, 20-29 to >70 years) and 14 were male. Their median workload was 175 patients per week (range, 50-99 to >200) and 13 had postgraduate qualifications. The GPs' age and sex distribution was similar to that of respondents in a national general practice population survey.11 The median age of the 10 pharmacists was 40 years (range, 20-29 to 50-59 years) and eight were male. Their median workload was 125 prescriptions dispensed per day (range, 50-99 to >200) and three had postgraduate qualifications. |
Attitudes to interprofessional communication |
In the questionnaire, 16 GPs (89%) and all of the pharmacists stated
that they were in favour of more collaboration with one another. GPs
had a reasonably high regard for the local pharmacists -- 13 (72%) did
not agree that pharmacists' advice conflicted with their own; 17
(94%) agreed they had a good working relationship with the
pharmacists, and none believed that pharmacists contacted them
unnecessarily.
Seven pharmacists (70%) stated that they enjoyed a comfortable working relationship with GPs; only three (30%) agreed that GPs were not always receptive to inquiries about prescriptions, and five (50%) expected the GP to be responsive to contact about a potential drug interaction. Before the intervention, the concept of including the general purpose of a medication on the prescription was not recognised by seven GPs (39%) and seven pharmacists (70%) as useful or necessary for the pharmacist. However, nine pharmacists (90%) stated that to counsel patients adequately they often needed to ask a patient for more information than was on the prescription. |
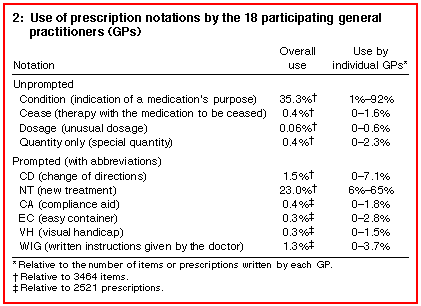
Use of the new prescription form | Of a total 3600 prescription forms distributed to the GPs, 70% (2521 prescriptions for 3464 individual prescription items) were collected and analysed. Forty-five per cent of individual prescription items (1559) included one of the new notations. The rate of use of the new notations by the GPs is shown in Box 2. The purpose of the medication was notated for 35.3% of all prescription items (1222), with use of this notation by individual GPs ranging from 1% for the lowest user to 92% for the highest user. In only 0.3% (range, 0-4.5%) of cases where this notation was used did doctors request that the information be omitted from the label of the medication container. |
Attitudes to the new prescription form |
Overall, the new system was positively received by most GPs and
pharmacists, and they suggested it should be widely implemented.
Also, most GPs and pharmacists saw indicating the purpose of the
medication on the prescription as the most substantial and important
focus of the new prescription notations, and suggested that this
would enhance the quality of information provided to consumers and
minimise irrelevant or inappropriate advice. Further, some
suggested that compliance may be improved because patients would
receive consistent advice from GPs and pharmacists.
Most GPs and pharmacists considered it useful to note when therapy with a long term medication was to be ceased, as patients may forget verbal instructions by the GP. Although the need did not often arise in practice, both GPs and pharmacists were in favour of specifically informing the pharmacist when an unusual dosage or a special quantity of medication was being prescribed. Most GPs and pharmacists thought it appropriate to indicate a new treatment, primarily for ongoing management. Noting a change of directions was considered useful by doctors and pharmacists alike, particularly with elderly patients or those who might still have unused prescriptions. The pharmacist could then be sure the change was intended, check the patient had understood the change, amend any unpresented repeats available, and note the change in the pharmacy computer record. Many GPs, pharmacists and consumers regarded preprinted abbreviations for specific patient needs as unnecessary; it was suggested that longhand could always be used for these or other specific requests to the pharmacist. The abbreviation for noting when separate written instructions had been given to the patient was considered a simple and useful quality control mechanism by doctors, pharmacists and consumers. The consumer focus groups indicated that pharmacists needed to know why someone was being prescribed a medication to enable them to provide appropriate counselling. They expressed reservations about conditions of a very personal nature; in such cases they expected the doctor to either ask their permission or omit the information from the prescription. In general, they considered it both appropriate and helpful to have the purpose of the medication included on the medication label. |
Discussion |
In this study the questionnaire findings gave basic data about
pre-existing attitudes of the GPs and pharmacists to their
respective roles, while the qualitative data explored, in much more
depth, the strengths and weaknesses of the new innovation. A 70%
return rate of prescriptions was satisfactory, as non-redemption of
prescriptions can range from 5%-20%.12 Our most notable finding was the utilisation rate of the new notations, with 35% of prescription items indicating the purpose of the medication to the pharmacist. This was somewhat unexpected, as such a strategy had not previously been considered necessary by GPs or pharmacists. Further, such a high utilisation rate requires considerable behaviour change, and behaviour change strategies generally have much more modest outcomes (eg, 6% for minimal intervention smoking cessation programs13 and 8%-25% for group counselling14). The benefits of asking GPs to indicate the purpose of the medication on the prescription were supported by the qualitative data suggesting that when this strategy was employed it was highly valued. Both GPs and pharmacists indicated that the new prescription form would improve the quality of information given to patients by ensuring that information given by pharmacists was consistent with the advice already given by the GP. It would also enable more information to be given to patients on the medication label. It was of particular interest that, while some of the GPs and pharmacists had very high workloads, they were able to use the new prescription conventions easily in their normal daily practice. There was a high level of utilisation of the new prescription form as a whole, with some notations being used frequently, and others barely at all. This is to be expected as the appropriate circumstance for the use of some notations (such as unusual dosage) would be infrequent. The notation to pharmacists that drug therapy was to be ceased was not widely used, possibly because this affected only a small number of patients, but more likely because there was no specific reminder on the prescription form itself and doctors may have simply forgotten to use it. While some variation in the use of notations may have related to differing perceptions of their value, it was probably also a result of the difficulty of altering ingrained behaviour. Some participants stated that they would try to increase their use of the strategies if they were implemented in the future. Indicating the purpose of the medication is easily misinterpreted as being the same as including the diagnosis, and thereby providing unnecessarily detailed and confidential information to the pharmacist. Participating GPs, however, reported that they provided general information at an appropriate level for the pharmacist and, subsequently, the patient. From the pharmacists' perspective, information about the purpose of a medication was most useful for drugs with multiple indications. It enabled them to target their information appropriately without asking questions to ascertain the precise indication for a particular patient. Generally, consumers thought that including the purpose of the medication on the container label was likely to enhance patients' understanding of their own medication. Confidentiality needs always to be considered, but there are clear advantages in having a drug's purpose included on the label in most situations. As we used only two small consumer focus groups, they could provide only a limited indication of what consumers may think of the proposed system. However, consumers did suggest that the new system would help them gain access to appropriate information and advice. Any future implementation should be made with adequate consumer input, to ensure attention to their requirements. In conclusion, this study showed that it is possible to modify the prescription form to include more information in a way that is acceptable to GPs and pharmacists in their daily practice. GPs, pharmacists and consumers found such changes worthwhile and believed they could lead to better medication management and patient care. |
Acknowledgements | We thank the Royal Australian College of General Practitioners and the Victorian Branch of the Pharmaceutical Society of Australia, and the members of the joint working party -- Val Constable, John Daffey, David Dammery, Chris Hogan, Alistair Lloyd, Mary Murray and Denise Ruth -- as well as the GPs, pharmacists and consumers in the study. Finally, we thank the Department of Health, Housing, Local Government and Community Services for financial support for the project. |
References |
(Received 5 Jun, accepted 10 Nov, 1997) |
Authors' details
Department of Community Medicine and Department of General Practice, Monash University, Melbourne, VIC.Merilyn J Liddell, MB BS, FRACGP, Senior Lecturer;
Sue P Goldman, BEd, GradDipSoc, Research Fellow.
Reprints will not be available from the authors.
Correspondence: Dr M
J Liddell, 867 Centre Road, East Bentleigh, VIC 3165.
E-mail:
merilyn.liddell AT med.monash.edu.au
Readers may print a single copy for personal use. No further
reproduction or distribution of the articles
should proceed without the permission of the publisher. For
permission, contact the
Australasian Medical Publishing Company
Journalists are welcome to write news stories based on what they read here, but should acknowledge their source as "an article published on the Internet by The Medical Journal of Australia <http://www.mja.com.au>".
<URL: http://www.mja.com.au/>
Received 25 May 2026, accepted 25 May 2026
- Merilyn J Liddell
- Sue P Goldman





 -adrenoceptor
blocking agents might be notated as "for arrhythmias" or "for blood
pressure". Pharmacists could then ensure that their medication
counselling was relevant to that particular indication and would
include the medication's purpose on the medication label for the
patient's information. There was provision for GPs to request that
the purpose not be written on the medication label by the pharmacist in
certain circumstances (such as to preserve patients' privacy if the
nature of their illness was not known to family members).
-adrenoceptor
blocking agents might be notated as "for arrhythmias" or "for blood
pressure". Pharmacists could then ensure that their medication
counselling was relevant to that particular indication and would
include the medication's purpose on the medication label for the
patient's information. There was provision for GPs to request that
the purpose not be written on the medication label by the pharmacist in
certain circumstances (such as to preserve patients' privacy if the
nature of their illness was not known to family members).