Reduction of inappropriate use of blood products by prospective monitoring of transfusion request forms
Annabel Tuckfield, Michael N Haeusler, Andrew P Grigg and Jack Metz
For editorial comment, see Allain & Williamson
Readers may print a single copy for personal use. No further reproduction or distribution of the articles should proceed without the permission of the publisher. For permission, contact the Australasian Medical Publishing Company
Journalists are welcome to write news stories based on what they read here, but should acknowledge their source as "an article published on the Internet by The Medical Journal of Australia <http://www.mja.com.au/>".
Abstract - Introduction - Methods - Results - Discussion - References - Authors' details
- - ©MJA1997
Abstract |
Objective: To determine the effect of prospective
monitoring on appropriateness of transfusions of red cells,
platelets and fresh frozen plasma (FFP). Design: Prospective interventional study. Setting: Royal Melbourne Hospital (a tertiary teaching hospital), Melbourne, Victoria, March-May 1996. Intervention: The blood product request form was modified to incorporate indications for transfusion and clinical and laboratory data. Requests were monitored by blood bank laboratory staff for conformation with hospital transfusion guidelines; non-conforming requests were discussed with the requesting medical practitioner by the Haematology Registrar before blood products were issued. In cases of disagreement, blood products were always issued. Subjects: 200 consecutive transfusion episodes for each product (red cells, platelets and FFP). Outcome measures: Appropriateness of transfusion, assessed by a Consultant Haematologist according to hospital guidelines. Rates of inappropriate transfusion episodes after intervention were compared with rates in a previous study. Results: After intervention, rates of inappropriate transfusion episodes fell significantly (red cells, 16% to 3% [P=0.004]; platelets, 13% to 2.5% [P=0.02]; and FFP, 31% to 15% [P=0.02]). Almost all inappropriate FFP transfusion episodes post-intervention were due to failure to demonstrate prolongation of prothrombin or activated partial thromboplastin times more than 1.5 times the control value. Conclusion: Prospective monitoring of request forms can reduce rates of inappropriate transfusions. High rates of inapproriate FFP transfusions possibly reflect uncertainty about appropriate laboratory criteria for FFP transfusion. While results of large prospective randomised controlled clinical trials of FFP transfusions are awaited, current laboratory criteria can be retained, but should be applied with flexibility. |
Introduction |
In 1995, we reported results of an audit of blood product use at a
tertiary teaching hospital (Royal Melbourne Hospital, Victoria).1 Indications for
transfusion were analysed, mainly by retrospective review of
medical records. We found that blood product use was inappropriate
for 16% of red cell, 13% of platelet and 31% of fresh frozen plasma (FFP)
transfusion episodes. In a significant number of episodes, the
specific indication for transfusion was not documented in the
medical record.
Blood products must be used appropriately to minimise patient exposure to potential hazards, to conserve a limited resource and to contain costs. Measures to reduce inappropriate use of blood products have had varying results. While retrospective audit and education do not produce consistent and continuing improvements,2-8 prospective monitoring (monitoring of requests for blood products before issue) has usually been more successful.9-15 Consequently, in March 1996, the Royal Melbourne Hospital introduced prospective monitoring of transfusion requests with the aim of reducing inappropriate use of blood products. We present the results of the first three months of this intervention. |
Methods | |
Intervention |
Blood products request form: The blood products request form was redesigned to incorporate the indication for transfusion and relevant clinical and laboratory data. These included:
In addition, the redesigned request form stipulates that the indication for transfusion must conform with the hospital's transfusion guidelines, which are printed on the reverse of the form (Box 1). Criteria for appropriate transfusions: The criteria were the same as those used in the audit.1 For FFP use, the requirement for coagulation times to be prolonged by more than 50% translated to PT >20 seconds or APTT >40 seconds. For massive blood transfusion (10 or more units of red cells in less than 24 hours), blood products were issued before laboratory results were available, and these transfusions were accepted as appropriate. In view of the potentially devastating effects of minor haemorrhage associated with neurosurgical procedures, these were exempted from the criteria for FFP use.1 Monitoring: On receiving a completed blood product request form, the senior medical laboratory scientist determined that the necessary information had been provided, and telephoned the requesting doctor to obtain any missing information. The scientist then checked that the indications conformed with transfusion guidelines, and if so, blood products were issued. If clinical or laboratory indications did not conform, the request was referred to the Haematology Registrar who consulted the requesting doctor. There were three possible outcomes: they agreed (i) either that the transfusion was not indicated and the blood products were not issued or (ii) that it was indicated and the products were issued or (iii) they disagreed and the products were issued. The consultation was never confrontational, and the blood product was always issued when there was disagreement. However, for the study these cases were subsequently referred to the Consultant Haematologist who reviewed the haematological data, transfusion request and medical record to decide if the transfusion was indeed inappropriate. |
Analysis of outcomes | For each type of blood product (red cells, platelets and FFP), we analysed 200 consecutive transfusion episodes between March and May 1996. As in the initial audit, we excluded FFP transfusions for patients with thrombotic thrombocytopenic purpura, because of the different rationales for FFP use in this condition and in coagulation factor depletion. Rates of inappropriate blood product use after introduction of prospective monitoring were compared with those found during the previous audit (pre-intervention) with two-sided Fisher's exact tests. |
Results |
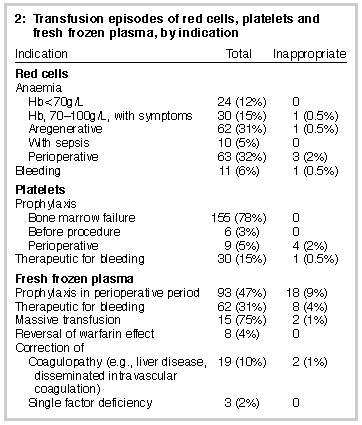
Indications for transfusion of red cells, platelets and FFP after
introduction of prospective monitoring are shown in Box 2 (below). They were
the same as in the pre-intervention audit.1
Numbers of transfusions referred to the Consultant Haematologist for review were: red cells, eight; platelets, 12; and FFP, 41. Final numbers of transfusion episodes deemed inappropriate were: red cells, six (3%); platelets, five (2.5%); and FFP, 30 (15%). Compared with the pre-intervention audit, rates of inappropriate transfusion episodes were significantly reduced, from 16% to 3% for red cells (P= 0.004), 13% to 2.5% for platelets (P=0.02) and 31% to 15% for FFP (P= 0.02). Of the six patients in whom red cell use was deemed inappropriate, five had a pretransfusion haemoglobin concentration greater than 100 g/L, the threshold for appropriate transfusion (range in these patients, 104-114 g/L). The sixth patient had a haemoglobin concentration of 98 g/L, but no clinical indications to warrant transfusion. In four of the five inappropriate platelet transfusion episodes, the platelet count was greater than 63x109/L (range, 63x109-159x109/L), a level at which platelets are not indicated except in the case of excessive bleeding with cardiac bypass surgery. Although three of these four transfusions were given in association with cardiac bypass surgery, there was no evidence of excessive bleeding. In 30 patients, FFP transfusion was deemed inappropriate as coagulation test results did not meet the criterion of a 50% prolongation of coagulation times. In one of these 30, this was because no coagulation tests had been performed. |
Discussion |
We found that the rate of inappropriate transfusion episodes fell
significantly after introduction of prospective monitoring, from
16% to 3% for red cells, from 13% to 2.5% for platelets and from 31% to 15%
for FFP.
The success of prospective monitoring in reducing rates of inappropriate transfusions for red cells and platelets was not matched for FFP. This agrees with results of three previous studies, which have found persistently high rates of FFP transfusions despite monitoring (>40%,8 33%13 and 27%,15 respectively). As in these studies, we found the reason for deeming FFP use inappropriate was almost invariably failure to show sufficient abnormality in coagulation test results. The need to document abnormal coagulation with a PT or APTT greater than 1.5 x the control value, before FFP transfusion is deemed appropriate, rests on the assumptions that abnormal bleeding is unlikely if coagulation times are shorter than this but likely if they are longer, and that, when coagulation times are greater than 1.5 x the control value, FFP transfusion is likely to prevent or control bleeding. Detailed analysis of published data suggests that these assumptions are questionable.16-23 It is of interest that we found that lowering the cut-off for appropriate FFP transfusions from PT >20 seconds (1.5 times the control value) to PT >17 seconds would have lowered the rate of inappropriate transfusions from 15% to 3%, comparable to the rate found for red cell and platelet transfusions. A PT of 17 seconds is still 3.5 seconds longer than the median control value. Further investigation is needed to establish the clinical guidelines for FFP transfusion. Two publications24,25 have pointed to the need for a large multicentre prospective randomised controlled clinical trial to provide a definitive answer for the role of FFP in patients with acquired multiple coagulation defects. In the interim, it would seem reasonable to require docu mentation of abnormal coagulation as a criterion for appropriate FFP use, but the available evidence does not warrant rigid application of a cut-off level, such as 50% prolongation of PT or APTT. Other issues that must be addressed before a system of prospective monitoring can be introduced include exemption criteria. Some protocols exempt operating room patients,6 "desperate situations"14 and haematology and oncology patients.13 Emergency situations are also usually exempted from the requirement for laboratory data if the laboratory cannot provide urgent results at all times. If the criteria are to be applied to emergency situations, laboratory facilit ies must be available to provide the data quickly. In some programs of prospective monitoring, blood products are never withheld, but apparently inappropriate transfusions are later reviewed.11,14 Refusal to issue blood products leads to an adversarial relationship between clinicians and laboratory staff, which may compromise patient care. Refusal might also have medicolegal implications if subsequent patient morbidity or death could be attributed to withholding of the transfusion. For sustained improvement in practice, prospective monitoring must be continued indefinitely.11 This is both time consuming and demanding of staff. The demand might be lessened by computerised audit of transfusion requests; clinical and laboratory data could be entered into a program which flags non-compliant requests for review by blood bank staff.26 In conclusion, prospective monitoring was successful in reducing inappropriate use of red cells and platelets, but only partly successful for FFP. This probably reflects uncertainty about the appropriate clinical guidelines for FFP use, and although current laboratory criteria for FFP use should probably be retained, they should be applied with flexibility. |
References |
(Received 20 Mar, accepted 1 Jul 1997) |
Authors' details
Department of Diagnostic Haematology, Royal Melbourne Hospital, Melbourne, VIC.Annabel Tuckfield, FRACP, FRCPA, Registrar;
Michael N Haeusler, FAIMS, Senior Scientist, Blood Bank;
Andrew P Grigg, FRACP, FRCPA, Haematologist;
Jack Metz, MD, FRCPA, Head.
Reprints will not be available from the authors. Correspondence: Dr A Tuckfield, Diagnostic Haematology, Post Office, Royal Melbourne Hospital, Parkville, VIC 3050.
E-mail: depATmis.medrmh.unimelb.edu.au
©MJA 1997
<URL: http://www.mja.com.au/>
© 1997 Medical Journal of Australia.
Received 4 June 2026, accepted 4 June 2026