Reported general practitioner vaccination procedures, 1994 and 1996
Ana Herceg, Monica B Johns and Helen M Longbottom
For editorial comment, see Kable & Harris
Readers may print a single copy for personal use. No further reproduction or distribution of the articles should proceed without the permission of the publisher. For permission, contact the Australasian Medical Publishing Company
Journalists are welcome to write news stories based on what they read here, but should acknowledge their source as "an article published on the Internet by The Medical Journal of Australia <http://www.mja.com.au/>".
Abstract - Introduction - Methods - Results - Discussion - References - Authors' details
- - ©MJA1997
Abstract |
Objective: To determine general practitioner (GP)
vaccination procedures during the first two years of implementation
of the National Childhood Immunisation Program. Design: Two cross-sectional self-completion mail surveys of Australian GPs, in October/November 1994 (Survey 1) and April/May 1996 (Survey 2). Participants: 1417 systematically selected (Survey 1) and 1482 randomly selected (Survey 2) GPs. Intervention: Mailing of a package of publications on immunisation to all GPs, a community education campaign, and the introduction of the Australian Childhood Immunisation Register. State and Territory and local initiatives on immunisation, changes in vaccine distribution and funding, and media coverage of vaccination issues may also have affected GPs' procedures. Main outcome measures: Reported vaccination procedures consistent with National Health and Medical Research Council (NHMRC) recommendations. Results: Response rates were 77% (Survey 1) and 78% (Survey 2). There were statistically significant rises in the proportions of GPs who reported vaccination procedures consistent with NHMRC recommendations for simultaneous administration of vaccines, injection site, vaccination of children taking antibiotics or those with mild upper respiratory tract infection, and vaccine storage. There was a reduction in reported opportunistic vaccination, and no change in reported procedures regarding pertussis vaccination. Conclusions: Many factors may have contributed to these changes in GP vaccination procedures. While the changes mostly show an improvement in adherence to NHMRC guidelines, further progress needs to be made, particularly in the areas of opportunistic vaccination, pertussis vaccination and vaccine storage. The best method of informing GPs of current vaccination recommendations needs to be explored. |
Introduction |
Vaccine-preventable diseases of childhood continue to occur in
Australia, with large outbreaks of measles, pertussis and rubella
occurring in 1993 and 1994.1
Vaccination coverage rates in Australia are still not high enough to
prevent these outbreaks, with reported coverage varying from 60% for
pertussis to 92% for measles.2
More than 60% of childhood vaccinations in Australia are given by general practitioners (GPs),2 and vaccination guidelines are published by the National Health and Medical Research Council (NHMRC) in The Australian immunisation handbook, which is updated regularly.3-5 Studies in the United States have shown that health care systems and immunisation provider services contain major barriers to childhood vaccination, including inaccessibility, insufficient staff, insufficient clinic hours, missed opportunities to vaccinate (i.e., not vaccinating children who present for another reason and are due or overdue for vaccination), and misconceptions about true and false contraindications to vaccination.6 The existence or otherwise of these barriers to childhood vaccination in Australian health care has not been well researched, but one study showed that many immunisation providers in Victoria were unaware of the NHMRC recommendations for diphtheria-tetanus-pertussis (DTP) vaccine and combined diphtheria-tetanus (CDT) vaccine.7 Other studies found that GPs frequently do not follow the NHMRC recommendations for site of injection of vaccines and vaccine storage.8,9 The National Childhood Immunisation Program was devised to address the problem of low vaccination cover - age through a coordinated national approach. The Program, which commenced in July 1994, involved Commonwealth, State and Territory health authorities, as well as professional and consumer bodies. Aspects of the Program included improving surveillance and reporting for vaccine-preventable diseases and vaccination coverage; improving vaccine purchasing, distribution and delivery; and educating parents, the general public and vaccination providers about vaccination. We conducted two national surveys of GPs to estimate changes in vaccination procedures during the first two years of the National Childhood Immunisation Program. |
Methods | |
Sample selection and survey method |
Providers were systematically (Survey 1) and randomly (Survey 2)
selected from GPs registered with the Health Insurance Commission
who had lodged more than 50 services in the preceding quarter. A 10%
sample was selected from each group, comprising 1843 GPs in Survey 1
and 1888 GPs in Survey 2. The sample for Survey 1 was created by
selecting every tenth GP on the list; a modified classic synchronised
sampling method was used for random selection for Survey 2.10 Both surveys were anonymous self-completion mail surveys. For each survey, an initial questionnaire and covering letter was sent to each GP, followed a week later by a reminder letter and a duplicate questionnaire. Practitioners were asked to ignore the reminder if they had already completed the questionnaire, and to return only one blank questionnaire if they did not wish to participate in the survey. The two surveys contained identical questions about GPs and their practices, vaccine storage methods and vaccination procedures. Questions on vaccination procedures and vaccine refrigeration were based on the NHMRC recommendations in the fifth edition of The Australian immunisation procedures handbook (1994). No identifying information was collected. |
Interventions |
Survey 1 was conducted in October/November 1994, one month before the
National Childhood Immunisation Program sent a package of
publications to all vaccination providers, including all GPs. The
package included The Australian immunisation procedures
handbook (fifth edition) and other publications on vaccine
provision and storage.
Survey 2 was conducted in April/May 1996, after the National Childhood Immunisation Program had been in place for almost two years. Activities of the Program during that time included distribution of the publications mentioned above (November 1994), a community education campaign (April to November 1995), and the introduction of the Australian Childhood Immunisation Register (January 1996). During the period between the two surveys, other activities which may have affected GP immunisation practices included State and Territory initiatives to inform immunisation providers about vaccination procedures, changes in vaccine distribution and funding systems, the development of school-entry vaccination record requirements in some jurisdictions, media coverage, and local promotion of vaccination issues through Divisions of General Practice and other agencies. |
Statistical analysis | Data entry and analysis were done with Epi Info version 6. 11 The chi-squared test for independence in 2 x 2 tables was used to compare the results of the two surveys. Analyses were based on the number of respondents who completed each question rather than the total number of respondents. |
Results | |
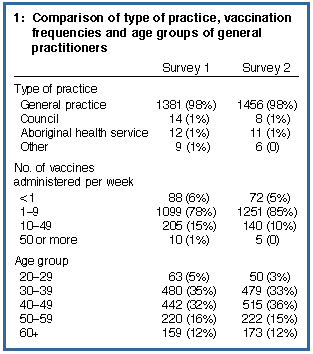
Response rates | After exclusion of respondents who indicated they did not provide childhood vaccinations, response rates were 77% for Survey 1 and 78% for Survey 2. Ninety-eight per cent of respondents worked in general practice. The two samples did not differ significantly in type of practice or age groups, but, for number of vaccines given per week, significantly more respondents in Survey 2 gave between one and nine (Box 1, below). |
Vaccine administration procedures |
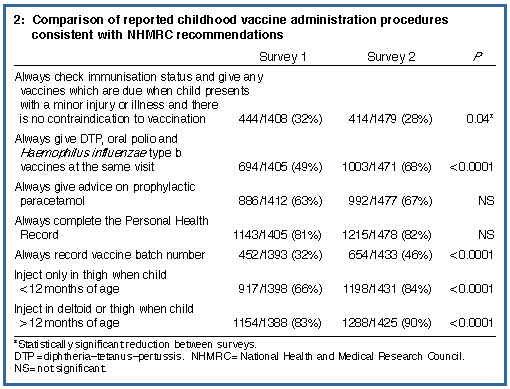
Reported vaccine administration procedures consistent with the
NHMRC recommendations are compared in Box 2. The proportion of
practitioners who would always opportunistically vaccinate fell
from 32% to 28%, although an additional 47% of GPs in 1996 said they
would frequently vaccinate children opportunistically.
Simultaneous administration of DTP, oral polio vaccine and
Haemophilus influenzae type b (Hib) vaccine increased from 49%
to 68%. Recording of vaccine batch number and use of recommended
injection sites also increased.
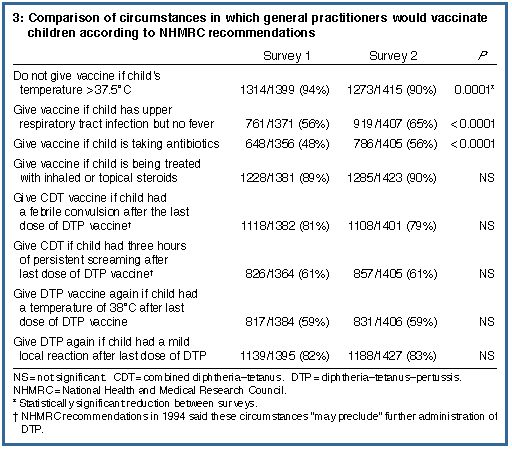
There were statistically significant rises from 1994 to 1996 in reported willingness of GPs to vaccinate children with mild upper respiratory tract infections or who were taking antibiotics (Box 3), and there was a fall from 94% to 90% in the proportion of GPs who would not vaccinate a febrile child. There was no change between the two surveys regarding when CDT should be used to replace DTP, or vaccination for a child taking inhaled or topical steroids.
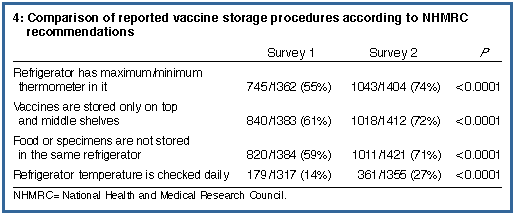
Reported storage of vaccines according to NHMRC recommendations improved between the two surveys, including use of a maximum/minimum thermometer, storing vaccines only on the top and middle shelves of the refrigerator and not storing food or specimens in the same refrigerator (Box 4). Although reported daily checking of the vaccine refrigerator temperature increased, in 1996 only 27% of GPs reported that this was done. A further 30% reported that the refrigerator temperature was checked weekly. In November 1994, 15% of respondents reported their practices had immunisation recall/reminder systems functioning. In April 1996, 78% of GPs reported always contributing to the Australian Childhood Immunisation Register. |
Discussion |
Our study has a number of limitations, including the sampling
methods, the number of non-respondents and the fact that it was based
on self-reported behaviour. While the sampling methods for the two
surveys were not identical, the two groups were demographically
comparable, and we believe both samples are representative of the GP
population. It is estimated that around 170 GPs would have received
both surveys; however, as the surveys were conducted anonymously we
have no record of this overlap. Thus, it is possible that a small
proportion of the reported change in behaviour was because of this
previously surveyed group who would have been more aware of the
issues. Finally, self-reported behaviour does not necessarily
correlate with actual behaviour. Hence, these surveys probably
provide a conservative estimate of behaviour contrary to NHMRC
guidelines.
Our study showed a significant improvement in several reported GP vaccination procedures between 1994 and 1996. The reasons for these changes are probably multiple, and include the information provided through the National Childhood Immunisation Program, State and Territory immunisation provider information and other professional education sources, as well as community and media interest in the topic. The degree to which each of these sources may have influenced the change cannot be assessed from our study. However, the coordination and information dissemination role of the National Childhood Immunisation Program meant that the information conveyed from health authorities and professional organisations to GPs between the two surveys was consistently based on NHMRC recommendations. Our results indicate, however, that more progress needs to be made in some areas. Opportunistic vaccination, including in hospital emergency departments, has frequently been identified as a way of improving immunisation coverage.12 In 1996, many GPs potentially missed opportunities to vaccinate by infrequently or never checking children's immunisation status when they presented for another reason. The high use of the parent-held Personal Health Record identified in our survey was also found by Bailey et al.13 Recording of batch number on the Record and/or in practice records improved between the two surveys; this is recommended by the NHMRC to ensure that children potentially at risk of illness can be found if a faulty batch of vaccine is identified. Misconceptions about true and false contraindications to vaccination have previously been recognised as barriers to full vaccination; this especially applies to when it is appropriate to replace DTP with CDT.6,7 The NHMRC guidelines have previously not been clear about pertussis vaccination, and this is likely to have contributed to the confusion. Most GPs in our study would replace DTP with CDT if a child had a febrile convulsion or persistent screaming after the last dose of DTP. These are situations which were previously classed by the NHMRC as contraindications to DTP and which, in the 1994 recommendations, "may preclude" another dose of DTP. The 1997 sixth edition of The Australian immunisation handbook provides new, clear guidelines on pertussis vaccination.5 GPs need to be aware of these changes; following the new recommendations should increase pertussis vaccination rates and prevent more disease. Vaccine storage procedures have been previously shown in New South Wales to be inadequate in many immunisation provider practices.9,14 Although there were improvements between our two surveys, in 1996 only 27% of practices checked and recorded the refrigerator temperature daily. Correct vaccine storage is essential to maintain vaccine potency. Our study looked only at private vaccination providers. We currently have no information about immunisation procedures of public providers, who administer around 40% of vaccines, and further studies are needed to assess procedures in this sector. While many GPs have kept abreast of changing recommendations, further improvements are possible. In addition, because of the impending availability of new vaccines and combinations of vaccines, further changes to the vaccination schedule and recommendations are likely in the coming years. Our study has not identified the best way to inform GPs of changes in recommendations, and this must be determined. The 78% participation rate of GPs in the Australian Childhood Immunisation Register five months after its commencement shows that good communication channels with GPs are available. Further improvements in vaccination provider practices will result in more children receiving the correct, efficacious vaccines safely and on time. |
References |
|
Authors' details
National Centre for Disease Control, Department of Health and Family Services, ACT.Ana Herceg, MPH, MAppEpid, Medical Epidemiologist, Surveillance and Management Section.
Monica B Johns, BAppSc; formerly, Senior Project Officer, National Childhood Immunisation Program.
Helen M Longbottom, MPH, FAFPHM; formerly, Director, Surveillance and Epidemiology Section.
Reprints will not be available from the authors.
Correspondence: Ana
Herceg, Surveillance and Management Section, National Centre for
Disease Control, MDP 6, Department of Health and Family Services, GPO
Box 9848, ACT 2601.
©MJA 1997
<URL: http://www.mja.com.au/>
© 1997 Medical Journal of Australia.
Received 22 May 2026, accepted 22 May 2026
- Ana Herceg
- Monica B Johns
- Helen M Longbottom