Burden and outcomes of hospitalisation for congestive heart failure
Fiona M Blyth, Ross Lazarus, David Ross, Michael Price, Gary Cheuk and Stephen R Leeder
For editorial comment see Krum
Readers may print a single copy for personal use. No further reproduction or distribution of the articles should proceed without the permission of the publisher. For permission, contact the Australasian Medical Publishing Company
Journalists are welcome to write news stories based on what they read here, but should acknowledge their source as "an article published on the Internet by The Medical Journal of Australia <http://www.mja.com.au/>".
Abstract - Introduction - Methods - Results - Discussion - Acknowledgements - References - Authors' details
- ©MJA1997
Abstract |
Objective: To describe the hospital burden and
health outcomes associated with admission for congestive heart
failure (CHF). Design and setting: Descriptive follow-up study in a tertiary-level metropolitan teaching hospital. Patients: Acute adult inpatients with a clinical diagnosis of CHF for more than 24 hours admitted to Westmead Hospital, Sydney, during the four months from September 1993 to January 1994. At baseline, 122 patients were assessed; 88 patients were assessed at four-month follow-up. Interventions: Usual clinical care. Main outcome measures: Length of stay; hospital bed-days; readmissions; mortality; health related quality of life (SF-36); patient knowledge. Results: The average age of subjects was 73.4 years. Many were using informal domiciliary care before admission. Mean length of stay for the baseline admission was 13.8 days, accounting for 7.6% of hospital separations and 1683 hospital bed-days, or 4.2% of bed-days for all inpatients aged 65 years and over. Fifteen patients were readmitted for CHF during the following four months, with a total of 26 CHF-related admissions. Twenty-one patients (17.2%) died during the course of the study. Quality of life at baseline was poor compared with population normative data, with a slight improvement among survivors at four-month follow-up. Patient knowledge of CHF was poor in a subsample survey ( n = 24). Conclusions: CHF represents a significant burden to patients (through morbidity and mortality), their carers (through provision of daily care), and hospitals (through multiple admissions for acute decompensation). It is difficult to monitor the hospital burden of CHF using routine data sources. |
Introduction |
Congestive heart failure (CHF) has been estimated to affect 3%-5% of
those aged over 65 years, and 10% of those over 75 years.1 It is the fastest growing
cardiovascular disorder in the United States,2 the only one increasing in incidence
and prevalence,3 and the
leading cause of hospital admission and readmission in Americans
aged over 65 years.4 In 1990,
CHF cost the US economy $US8 billion, and accounted for five million
hospital bed-days.5,6 In Scotland, CHF hospital discharge rates have risen in a decade to be almost equal to those for myocardial infarction.7 In the United Kingdom, the National Health Service spends £360 million per year in diagnosis and management, on a par with spending on stroke or asthma. Most of this expenditure is on hospital admissions.8 As there is little information available on the outcomes of current hospital management of CHF patients,9,10 we describe the health outcomes of a cohort of patients hospitalised with CHF, and the impact of their hospitalisation on a health service. |
Methods |
All acute adult patients admitted to Westmead Hospital during four
months (September 1993 to January 1994) with a clinical diagnosis of
CHF, or who developed CHF of more than 24 hours' duration during an
admission, were eligible for inclusion in the study. The diagnosis of
CHF was made by medical staff on clinical grounds. Additional
eligibility criteria were fluency in English and absence of
significant cognitive impairment.
Two cardiology-trained research nurses implemented an active daily case-finding strategy to overcome the difficulties encountered in identifying CHF patients from existing record systems. Diagnostic codes are generally not added to the patient's medical record until some time after discharge. Patients were identified by review of the computerised admissions log, medical record chart audit in the relevant ward areas, and consultation with medical staff. Potential subjects were assessed for their general condition and fluency in English and, if appropriate, screened for cognitive impairment by means of the Mini-Mental State examination.11 Informed consent was then sought. If patients were unable to participate fully, consent was sought to review their medical record and follow them up four months later to ascertain vital status. Baseline data were collected by interview, medical record review and self-administered questionnaires. Data were collected on demographic characteristics, domiciliary arrangements, current and past medical history, clinical severity of heart failure (according to the New York Heart Association [NYHA] criteria for grading functional incapacity of patients with cardiac disease), investigations, drug treatment, hospital resource use, formal and informal use of domiciliary care, and health-related quality of life (HRQOL) before admission (measured with the SF-36 [Medical Outcomes Study 36 Item Short Form Health Survey]).12 Patient knowledge about CHF was assessed by a short questionnaire in a subset of 24 consecutive patients. Four months after baseline admission, subjects were contacted by telephone to arrange follow-up by questionnaire. Data were sought on health status, HRQOL, current treatment, and domiciliary arrangements. The hospital's computer system was searched for any readmissions during the follow-up period. All analysis was performed using SAS version 6.08 for Windows.13 Two-sample t tests were used to assess the statistical significance of differences between groups, with adjustments made for multiple comparisons. SF-36 data were scored and subscales were calculated with the recommended scoring algorithm.12 Westmead Hospital Human Research Ethics Committee approved the study, and informed consent was obtained from all participating patients. |
Results |
There was no "gold standard" available for checking the accuracy and
completeness of study case ascertainment methods. However, a list
was assembled of all separations from Westmead Hospital during the
study recruitment period with a principal diagnosis code for CHF
(ICD-9 codes 428.0, 428.1, 428.9). These were then cross-checked
against a list of study subjects. This list identified seven patients
who were not identified by study case-finding methods. These were
considered "missed" potential cases, but represented a small
proportion of this group (4.3%).
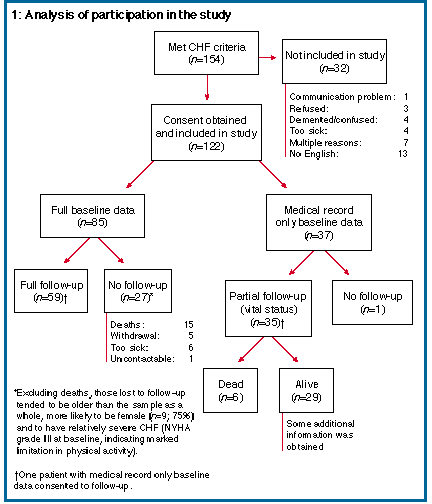
During the study recruitment period, 154 patients met the required clinical criteria and 122 (79%) consented to participate. Participation and follow-up are detailed in Box 1.
Sample characteristics: Women made up 54.9% of the study participants. The mean age was 73.4 years (range, 24-97 years); women were older on average than men (76.8 compared with 69.4 years; P = 0.0001). Most patients had a history of established CHF; 35.2% were undergoing their first hospital admission for CHF. Only 1.8% of patients were NYHA grade I on baseline admission, 12.3% grade II, 64.4% grade III, and 17.5% grade IV (4% lacked data for NYHA classification). Thirty-eight patients had their ejection fraction measured at the baseline admission, with a mean value of 35% (range, 9%-74%). Ischaemic heart disease (58.2%) and hypertension (39.3%) were major aetiological factors in CHF. The cohort was cared for by cardiologists (n = 74; 60.7%), and geriatricians (n = 48; 39.3%). Admission diagnoses are shown in Box 2 (below). When admission diagnoses were compared with ICD-9 codings on the medical record, 39 (86.6%) of the 45 with heart-failure-specific admission diagnoses and 30 (78.9%) of the 38 with suggestive diagnoses had a CHF-related ICD-9 code (428, 428.0, 428.1, 428.9) in one of the first six ICD-9 coding positions.
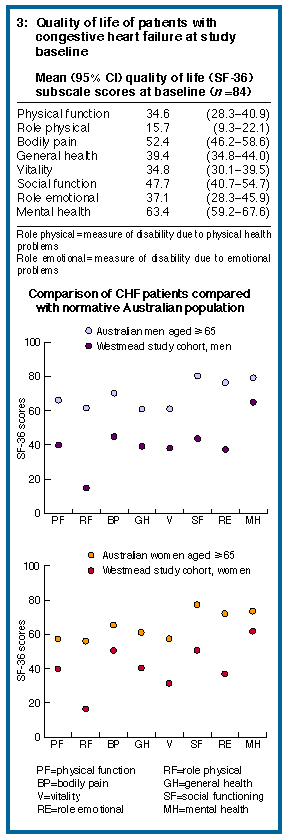
Preadmission characteristics: Data were available for 99 subjects: 88 lived in private accommodation, 50 reported needing regular help from family or friends with general housekeeping, and 23 reported needing help with supervision of medication. Length of stay: The study participants accounted for 7.6% of hospital separations (excluding day-only patients). The mean length of stay during the baseline admission was 13.8 days (SD = 12.1). Length of stay had a markedly skewed distribution (median, 10 days; range, 2-66 days). Overall, the sample accounted for 1683 bed-days during baseline admissions and 10.6% of hospital bed-days associated with the geriatricians and cardiologists who participated in the study. Of those patients aged 65 years and over admitted during the same period, the CHF cohort accounted for 4.2% of bed-days. Readmissions: During the follow-up period, there were 73 readmissions to Westmead Hospital from the study cohort, 26 due to a further episode of CHF in 15 patients. A total of 171 bed-days were used for CHF-related readmissions by the 14 patients for whom data were available. Deaths: Twenty-one patients died during the study (17.2%). Changes in domicile: Eight patients (8.1%) were discharged to a higher level of domiciliary care, suggesting a decline in independence and increased use of health and/or community services. Quality of life: Data on HRQOL before admission were obtained from 84 of the 85 subjects at baseline (Box 3). Mean SF-36 subscale scores for these subjects were generally low, particularly for subscales with physical health components. For each subscale there was a wide range of scores, indicating substantial variability in health-related quality of life before admission. Compared with Australian normative SF-36 data for men and women aged 65 years and over,14 the study cohort reported significantly lower mean HRQOL for all subscales.
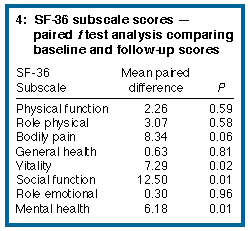
Follow-up SF-36 data were obtained for 58 subjects (Box 4). Patients with missing follow-up data had either died before follow-up (n = 15) or were too unwell to fill in the form (n = 6). In that sense, the SF-36 results at follow-up represent the "survivor" population within the study cohort. Mean follow-up subscale scores in survivors were somewhat higher compared with their baseline scores. The smallest improvements were in the subscales related to physical health. Improvements in the Social Function, Vitality and Mental Health subscales were statistically significant.
Patient knowledge: The 24 patients who completed the questionnaire were similar to the CHF cohort in age, sex, length of stay, and type of treating specialist. Nineteen had been admitted to hospital previously for CHF. Only 11 knew that they had been diagnosed as having heart failure. Although 16 agreed that patients with heart failure would need to take medication permanently, six thought that CHF was unlikely to recur. Nineteen agreed that shortness of breath and 15 agreed that ankle swelling were important symptoms, but only four recognised that rapidly increasing weight was important. |
Discussion |
CHF has a significant impact on hospital services. The study cohort
contributed significantly to adult bed-days attributable to
participating clinicians. A substantial proportion (12.3%) of the
cohort was readmitted with CHF within four months of their baseline
admission, suggesting an annual readmission rate of around 36%. This
compares with an annual readmission rate of nearly 20% in the SOLVD
(Studies of Left Ventricular Dysfunction) registry study cohort,
who were younger.10 CHF
admissions show seasonal variations, with a winter peak that is
probably associated with chest infection. Therefore, the
impact of CHF on acute hospital services may be even greater at that
time of year.
The 48 subjects (39.3%) who were admitted under the care of geriatricians were, by hospital admission policy, those aged over 65 with serious comorbidity and/or likely to represent a placement problem after discharge. Many required substantial help with tasks essential for independent living, and most of this help was provided informally. This was clearly a group that would require increased resources with deteriorating health. HRQOL was generally poor in the study participants compared with normative data,14 probably reflecting the combined effects of disease severity and comorbidity. There was limited improvement between baseline and follow-up, particularly in the subscales related to physical health, perhaps indicating that at baseline the lower limits of some SF-36 subscales did not adequately reflect the subjects' condition (i.e., a "floor" effect was operating). The mean changes in scores over time were small, but underlying this was great variability between individuals. At the time of this study there were few educational resources for CHF patients. This is surprising, as CHF is a chronic condition with acute exacerbations that may be ameliorated by early recognition and intervention, and which requires compliance with medication. Some CHF patients may avoid hospital admission by timely intervention prompted by self-monitoring of signs and symptoms. Seventy-six subjects (62.3%) had a recorded history of acute deterioration lasting for more than 24 hours, suggesting a possible opportunity for early recognition and intervention. The patient knowledge survey was limited, but it demonstrated an apparent lack of understanding of key features of CHF. Recent acute illness could have contributed to this. That almost a quarter of the cohort required daily help with taking medications has implications for the targeting of educational interventions. For elderly patients experiencing readmissions for CHF, the risk of dying in the near future and the palliative nature of treatment are additional issues which may need to be explored. This study was constrained, for practical reasons, by reliance on a clinical diagnosis of CHF. In a larger study with more resources, verification of diagnosis and more detailed examination of diagnostic coding practices would be desirable, and would allow identification of subgroups of particular clinical interest. Diagnostic coding practices in routine record keeping do not facilitate monitoring of the public health impact of CHF. A hospital admission for CHF may be assigned a principal ICD-9 code which reflects the underlying cause of CHF, and secondary ICD-9 codes are sometimes used to identify a past medical history of CHF rather than a feature of the current admission. In this study, poor health represented a further barrier to study participation and monitoring outcomes. Most losses to follow-up resulted from death or worsening health. During the study 21 subjects (17.2%) died. This represents a poor survival rate, worse than for many malignancies of adulthood. Cause of death was not ascertained, but in the SOLVD registry cohort10 most deaths were due to progressive CHF. In addition, sudden death occurs in CHF patients at five times the rate in the general population of the same age.15 Chronic CHF represents a significant burden to patients (through morbidity and mortality), their carers (through provision of daily care), and the hospital system (through multiple admissions for acute decompensation). |
Acknowledgements | We acknowledge the contribution of Moira Hewitt, RN, and Jeanette Bunn, RN, to the Westmead CHF Outcomes study, and study coinvestigator Dr Julia Lowe from the Newcastle Heart Failure Group. This study was funded by the NSW Health Department's Health Outcomes Program. |
References |
(Received 8 Oct 1996, accepted 10 Apr 1997) |
Authors' details
Westmead Hospital, Sydney, NSW.Fiona M Blyth, FAFPHM, Registrar, Department of Public Health and Community Medicine.
Ross Lazarus, FAFPHM, Senior Lecturer in Epidemiology, Department of Public Health and Community Medicine, University of Sydney at Westmead Hospital.
David Ross, FRACP, Head, Department of Cardiology.
Michael Price, FRACP, Head, Geriatric Medicine Unit.
Gary Cheuk, FRACP, Registrar, Geriatric Medicine Unit.
Stephen R Leeder, FRACP, PhD, FAFPHM, Professor of Public Health and Community Medicine, University of Sydney at Westmead Hospital.
Reprints will not be available from the author. Correspondence: Dr F M Blyth, Department of Public Health and Community Medicine, Westmead Hospital, Westmead, NSW 2145.
©MJA 1997
<URL: http://www.mja.com.au/> © 1997 Medical Journal of Australia.
Received 9 June 2026, accepted 9 June 2026
- Fiona M Blyth
- Ross Lazarus
- David Ross
- Michael Price
- Gary Cheuk
- Stephen R Leeder