An outbreak of Japanese encephalitis in the Torres Strait, Australia, 1995
Jeffrey N Hanna, Scott A Ritchie, Debra A Phillips, Jack Shield, M Clare Bailey, John S Mackenzie, Michael Poidinger, Bradley J McCall and Phillip J Mills
MJA 1996; 165: 256-260
Readers may print a single copy for personal use. No further
reproduction or distribution of the articles in whole or in part
should proceed without the permission of the publisher. For
copyright permission, contact the
Australasian Medical Publishing Company
Journalists are welcome to write news stories based on what they read here, but should acknowledge their source as "an article published on the Internet by The Medical Journal of Australia <http://www.mja.com.au/>".
Abstract - Introduction - Methods - Results - Discussion - Acknowledgements - References - Authors' details
- Register to be notified of new articles by email - - ©MJA1996
Abstract Objectives: To determine the distribution of virus infection during an outbreak of Japanese encephalitis (JE) in the Torres Strait, and to describe the environmental factors facilitating the outbreak.
Design: Human and porcine serological surveys for JE virus activity throughout the Torres Strait, and mosquito and household surveys on the island of Badu.
Setting: The island of Badu (where the clinical cases occurred) and the other islands of the Torres Strait, Australia, during April-May 1995.
Results: The serological surveys identified recent JE virus infection among residents or domestic pigs on at least nine outer Torres Strait islands. A JE virus, confirmed by nucleotide sequencing, was isolated from two asymptomatic Badu residents. Virus isolations and mosquito surveys implicated Culex annulirostris as the major vector involved in the outbreak. There was prolific Cx. annulirostris breeding in a variety of water bodies close to and within the Badu community. Over half (53%) of the households kept pigs in pens, and many (63%) of the pigpens were situated near standing water; in 56% of these "wet" pigpens Cx. annulirostris was breeding.
Conclusions: There was evidence of widespread JE virus activity throughout the outer islands of the Torres Strait. We suggest that migratory birds and/or wind-blown mosquitoes could have imported the virus into the Torres Strait from a focus of viral activity, possibly in Papua New Guinea, thereby initiating the outbreak. A combination of environmental factors, with large numbers of domestic pigs in close proximity to human dwellings and mosquito breeding sites, undoubtedly facilitated the outbreak on Badu.
MJA 1996; 165: 256-260
Introduction
Over a two-week period in March-April 1995, three cases of Japanese encephalitis (JE) occurred among residents of the island of Badu in the Torres Strait, Australia 1 ( Box 1). Although JE is widespread throughout Asia, 3 the Torres Strait outbreak is the first time that it has been recognised in Australia.J E is caused by a mosquito-borne flavivirus, and results in an acute illness characterised by headache, fever, convulsions, depressed level of consciousness and coma. It has a high case-fatality rate and there is a high prevalence of neurological sequelae in those who survive the acute illness. However, infection with the JE virus does not invariably cause disease; there is a high ratio of asymptomatic to symptomatic infections. 3
The JE virus is maintained in a natural cycle involving water birds such as egrets and herons. Pigs are very efficient amplifying hosts for the virus and therefore almost always contribute to outbreaks of JE in human populations. However, humans and other large verte brates, such as horses, are not efficient amplifying hosts, and are therefore "dead-end" hosts for the JE virus. 3
The outbreak was assumed to have been caused by a mosquito-borne virus, and Murray Valley encephalitis (MVE) virus (the major cause of arboviral encephalitis in Australia) was first suspected. Although the current status of flavivirus infections in the Torres Strait is uncertain, MVE and other Australian flaviviruses (Alfuy, dengue, Kokobera, Kunjin, Stratford and Sepik viruses) have been reported from either Cape York or Papua New Guinea (PNG). 4 Once laboratory findings confirmed the outbreak as JE, further investigations were undertaken.
Methods Serological survey
In April 1995, a serological survey was undertaken among residents of Badu. A non-random convenience sample was used. Human sera were screened for the presence of (i) flavivirus IgG and IgM by enzyme-linked immunosorbent assay (ELISA) 5 and haemagglutination inhibition assay, 6 and (ii) antibodies to JE, MVE and Kunjin viruses by plaque reduction neutralisation assay. 7 Sera testing positive for flavivirus IgM by ELISA were fractionated by ultracentrifugation over a sucrose gradient. IgM fractions were then tested for haemagglutination-inhibiting antibodies against JE, MVE and Kunjin viruses. 8 A diagnosis of a recent JE virus infection was made if there was a fourfold or greater rise in haemagglutination-inhibiting antibody titres in paired sera tested in parallel, or if JE virus IgM antibody and JE neutralising antibody were detected in serum at significantly higher titres than antibody to MVE and Kunjin viruses.
Blood samples for serological studies were also collected from domestic animals, in particular pigs, on Badu. The animal sera were screened for the presence of total (i.e., IgM and IgG) antibody to JE, MVE and Kunjin viruses by haemagglutination inhibition assay. A portion of the sera positive for total JE antibody by haemagglutination inhibition assay was then tested by plaque reduction neutralisation assay. The presence of neutralising antibody at significantly higher titres to JE virus than to MVE and Kunjin viruses was considered evidence of exposure of an animal to JE virus.
Isolation of virus
Virus isolations were attempted from human sera obtained from residents of Badu: 100 m L of serum was inoculated onto confluent monolayers of C6/36 ( Aedes albopictus ) cells. 9 Viral growth was monitored by testing the culture super natant for haemagglutinating ability. Box 2 details the method used for identification of virus isolates.
Mosquito survey
Because the outbreak was initially suspected to be MVE, mosquito surveillance focused on the primary vector of MVE in Australia, Culex annulirostris . 4 Commencing on 7 April, larval and adult mosquito surveys were conducted within a 1-km radius of the centre of the Badu community. Mosquito larvae were sampled with a 350 mL dipper and adult mosquitoes were trapped with Centers for Disease Control light traps -- eight, each baited with 1 kg of dry ice (carbon dioxide) and 1-octen-3-ol (octenol; release rate 10 mg/h), 16 were run overnight at locations throughout the community on four occasions throughout April 1995.
Household survey
In May 1995, environmental health staff undertook a house-to-house survey to interview one adult resident of each household on Badu about household characteristics that may have contributed to the outbreak of JE. These included the presence of backyard pigs and horses and the state of repair of waste disposal (drainage and sewerage) systems.
Survey of other communities
To determine the extent of recent JE infection, blood was taken from convenience samples of people residing on 12 other outer islands, three inner islands and seven Cape York communities. Blood was also taken from pigs from eight other outer islands, three inner islands and two Cape York communities.
Results
Serological survey
Two hundred and fifteen Badu residents were tested for JE infection:
35 people had serological evidence of recent JE infection (Box 3), and
21 of these had a fourfold or greater rise in JE-specific antibody
titres in paired sera. All 11 pigs from Badu (tested by plaque
reduction neutralisation assay) showed serological evidence of JE
infection (Box 3). Most of the horses (7/10) and dogs (10/16) tested
also showed serological evidence of JE infection, but not one of six
chickens was positive.

Isolation of virus
JE virus was isolated from the sera of two Badu residents, both of whom remained asymptomatic. Nucleotide sequences obtained by reverse transcriptase-polymerase chain reaction (RT-PCR) amplification showed that the two viruses (designated FU and NO) were definitely strains of JE, with a 90% nucleotide homology with other strains of JE virus but with less than 70% homology with other flaviviruses. Both Badu isolates were very closely related, with 99% homology with each other.
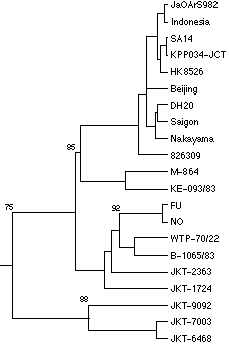
Detailed sequence comparison of the prM region of the viruses with
that of other JE virus isolates showed that the Badu viruses were most
closely related (92% homology) to other viruses of genotype III,
12,13 especially WTP-70/22
(from Malaysia) and B1065 (from southern Thailand) (Figure 1).
The nucleotide sequences of JE-FU and JE-NO have been deposited in
GenBank (the international computerised depository of
genomic-sequence information), with accession numbers L43565 ( prM
) and L48968 ( NS5 ) for JE-FU, and L43566 ( prM ) and L48967 ( NS5 ) for
JE-NO.
Mosquito survey
Numerous water bodies contained large numbers of Cx. annulirostris larvae. There were extensive swampy areas close to the community, and those contaminated by horse faeces had high larval densities ( >= 10 larvae/dip). A waterhole on the inland side of the community contained a large amount of grass clippings, rubbish and horse faeces; high larval densities were found along the margins. Many of the concrete-lined drains running through the community were overgrown with vegetation and contained mosquito-infested accumulations of water. High densities were also found in some of the defective household waste disposal systems (see below), and in pools in horse hoofprints.
A total of 22 190 adult mosquitoes were trapped in the Badu community in April. Aedes kochi , Aedes culiciformis and Cx. annulirostris comprised 99% of the collection, with means of 364, 243 and 127 adult mosquitoes per trap, respectively. While the largest collections of Cx. annulirostris (up to 607 per trap) were taken from swampy areas near the community, collections of more than 100 per trap were taken within the community and close to houses. Eight JE viruses were isolated from 2871 Cx. annulirostris mosquitoes collected at Badu, but not from any other species (S A Ritchie, D A Phillips and A K Broom, unpublished data). Six of the isolates were from mosquitoes collected from within the community.
Household survey
An adult resident from 97 of the 102 houses in the community was interviewed for the household survey. Fifty-one (53%) of the households kept pigs; 35 (69%) of the pigpens were within 50 metres of the house. There were 179 domestic pigs in the community, an average of 3.5 (range, 1-11) per pig-rearing household. On inspection, 32 (63%) of the pigpens were situated either over or surrounded by standing water, and in 18 (56%) of these "wet" pigpens Cx. annulirostris was breeding. The householders of 15 (15%) of the houses owned a total of 18 horses; they were all kept in paddocks within 500 metres of the houses.
The interviewees reported that 62 (64%) of the 97 houses had defective waste disposal systems: 49 of these houses had either waste water or raw sewage overflowing from septic tanks either into the house or into the yard, with 10% of the defective septic tanks containing Culex larvae.
Survey of other communities
A total of 1242 human serum samples were collected from the other communities. Twenty people from three other outer islands had serological evidence of recent JE virus infection. There was no evidence of JE infection in those tested from the inner islands or from the Cape York communities (Box 3). There was no evidence of any prior infection (i.e., only IgG JE antibodies) in any individual.
Of the 182 pig sera collected from the other communities, 121 (66%) were tested by plaque reduction neutralisation assay. There was serological evidence of JE infection in pigs from all eight other outer islands, but no evidence of infection in pigs from the inner islands and Cape York Peninsula (Box 3).
Discussion The human serological survey indicated that people from four outer islands had been recently infected with the JE virus. The porcine serological survey found evidence of JE activity in pigs from another five outer islands. Therefore, the two surveys indicated widespread, and presumably recent, JE virus activity in at least nine of the outer Torres Strait islands. There was no evidence of either human or porcine infection in the communities surveyed on the inner islands or on Cape York. The lack of evidence of prior infection in those tested suggests that the 1995 outbreak was the first incursion of the JE virus into the Torres Strait.
Nucleotide sequencing studies clearly defined the virus strains isolated from the two people from Badu (and presumably the virus that infected people and pigs throughout the outer Torres Strait islands) as JE virus, and that the isolates were distinct from, but related to, virus strains that were circulating in southern Thailand, Malaysia and Indonesia between 1968 and 1983. 12 The virus differed, however, from a newly recognised genotype known to be circulating in Indonesia in 1980-1981. A strain (JKT-6468, Figure 1) of this latter genotype has been isolated from culicine mosquitoes collected from Flores, east of Bali, and is therefore the closest known isolate to Australia. 13
JE virus has not been isolated or reported as causing human disease in either PNG or Irian Jaya. Although no evidence of JE virus has been demonstrated conclusively in a number of serological surveys, there was possible JE seropositivity in a few single-serum specimens collected in 1956-1957 in the Western Province of PNG. 17 More recently, antibody to JE virus was detected by competitive ELISA in at least 23% of human sera collected in the Western Province in 1989 (R A Hall and J S Mackenzie, unpublished data), and 49% of porcine sera collected in the Western Province in 1995 were positive for neutralising antibody to JE virus (J Shield and R A Lunt, unpublished data). Thus, it seems possible that JE virus has become enzootic in parts of southwestern PNG.
The northwestern islands (Boigu, Dauan and Saibai), because of their closeness to PNG, receive Papuan visitors virtually every day, raising the question of whether a viraemic visitor could have brought the JE virus across from PNG to the Torres Strait. However, humans, as "dead-end" hosts of the JE virus, have a low level of JE viraemia of short duration. 3 On the other hand, a viraemic pig imported from PNG, being a very efficient amplifying host, might have initiated the Torres Strait outbreak. However, the Australian Quarantine Act 1908 (Cwlth) prohibits the movement of live animals from PNG to the Torres Strait. The ban is actively enforced and respected; since the appointment of the first indigenous Quarantine Officers in 1982 they have not had occasion to seize even one illegally imported pig (P Stephen, Australian Quarantine and Inspection Service, personal communication). Therefore, we believe it most unlikely that an imported pig initiated the outbreak.
Over 100 species of bird migrate annually between Australia and New Guinea, usually in a predictable seasonal pattern. 18 A further 63 species cross the Torres Strait at irregular intervals; these include numerous aquatic species, including wading birds. 18 The rufous night heron ( Nycticorax caledonicus ), for example, is a common nomad found throughout the Torres Strait; 18 it is closely related to the black-crowned night heron ( N. nycticorax ), a principal bird species implicated in the natural JE virus bird-mosquito cycle in Asia. 19 Experimental JE virus infection of the rufous night heron produces levels of viraemia that are quite adequate to infect the most efficient JE vector in Asia ( Culex tritaeniorhynchus ). 20
From December to April the prevailing wind in the region is from the northwest, raising the possibility that the dispersal of "wind-blown" mosquitoes could have carried the JE virus from New Guinea to the Torres Strait. Indeed, it has been reported that female Cx. annulirostris mosquitoes "can disperse at least 12 km and probably further"; 21 the northernmost outer islands are all less than 10 km from the PNG coastline (Box 1). We therefore suggest that two natural phenomena -- viraemic migratory birds and/or infectious wind-blown mosquitoes -- are plausible mechanisms for the importation of the JE virus from New Guinea to the Torres Strait, thereby initiating the outbreak.
Cx. annulirostris was undoubtedly the major vector at Badu, and presumably at the other outer islands. Multiple JE virus isolations were made from Cx. annulirostris but not from any other mosquito species. The very small (fewer than one adult mosquito per trap) collections of Culex quinquefasciatus and Culex bitaeniorhynchus (both recognised as being either "marginal" or "occasional" vectors of the JE virus in Asia) 3 indicate that these species were of no importance in this outbreak.
Cx. annulirostris was breeding in abundance in a variety of sites close to the community. However, the natural surface waters were extensive, and produced the most mosquitoes. The waterhole with run-off watercourses passing through the community meant that there was extensive mosquito breeding close to the $omestic pigs and to the people. The close proximity of the blocked drains to many of the houses also made them a significant risk. Defective waste disposal systems may also have contributed to the outbreak.
The most striking combination of environmental factors contributing to the outbreak was the large number of domestic pigs adjacent to human dwellings and prolific mosquito breeding sites (Figure 2). The density of pigs, the abundance of the vector species and the human population density are all critical factors in determining the risk of human infection. 3,22 Horses, although "dead-end" hosts for the JE virus, 3 nevertheless contributed to the outbreak by providing numerous hoofprint breeding sites, nutrient (i.e., faeces) to the larvae and bloodmeals for female mosquitoes. 23
We need to determine the likelihood of future incursions of the JE virus into the Torres Strait. At the same time there is a clear need not only to reduce the mosquito breeding potential but also to improve the environmental conditions on the islands. Meanwhile, an inactivated JE vaccine has been offered to the inhabitants of the outer islands to confer protection while these risk assessment studies and risk reduction interventions are being implemented. 24
Acknowledgements
Many people assisted with the investigation of the outbreak. We thank
the staff of the Community Health Centers throughout the Torres
Strait, Torres Strait Public Health Program, Tropical Public Health
Unit, Australian Quarantine and Inspection Service, Queensland
Department of Primary Industries, Laboratory of Microbiology and
Pathology and the Australian Animal Health Laboratory. We are
particularly grateful to Dr Ted Tsai (Division of Vector-Borne Viral
Diseases, Centres for Disease Control and Prevention, USA) for his
expert advice and support.
References
- Hanna J, Ritchie S, Loewenthal M, et al. Probable Japanese
encephalitis acquired in the Torres Strait. Commun Dis Intell
1995; 19: 206-208.
-
The Torres Strait Health Workshop Working Party. Torres Strait
Health Strategy. Thursday Island: Torres Strait Health Council,
1993.
-
Vaughn DW, Hoke CH Jr. The epidemiology of Japanese encephalitis:
prospects for prevention. Epidemiol Rev 1992; 14: 197-221.
-
Mackenzie JS, Lindsay MD, Coelen RJ, et al. Arboviruses causing
human disease in the Australasian zoogeographic region. Arch
Virol 1994; 136: 447-467.
-
Burke D, Nisalak A, Ussery M. Antibody capture immunoassay
detection of Japanese encephalitis virus immunoglobulin M and G
antibodies in cerebrospinal fluid. J Clin Microbiol 1982;
16: 1034-1042.
-
Clarke DH, Cassals J. Techniques for haemagglutination and
haemagglutination inhibition with arthropod borne viruses. Am J
Trop Med Hyg 1958; 7: 561-573.
-
Gorman BM, Leer JR, Filippich C, et al. Plaquing and neutralization
of arboviruses in the PS-EK line of cells. Aust J Med
Technol 1975; 6: 65-71.
-
Field PR, Murphy AM. The role of specific IgM globulin estimations
in the diagnosis of acquired rubella. Med J Aust 1972; 2:
1244-1248.
-
Igarashi A. Isolation of a Singh's Aedes albopictus cell
clone sensitive to dengue and chikungunya viruses. J Gen Virol
1978; 40: 531-544.
-
Pierre V, Drout M-T, Deubel V. Identification of mosquito-borne
flavivirus sequences using universal primers and
reverse-transcriptase-polymerase chain reaction. Res Virol
1994; 145: 93-104.
-
Sellner LN, Coelen RJ, Mackenzie JS. A one-tube, one manipulation
RT-PCR reaction for detection of Ross River virus. J Virol Methods
1992; 40: 255-264.
-
Chen W-R, Tesh RB, Rico-Hesse R. Genetic variation of Japanese
encephalitis virus in nature. J Gen Virol 1990; 71:
2915-2922.
-
Chen W-R, Rico-Hesse R, Tesh RB. A new genotype of Japanese
encephalitis virus from Indonesia. Am J Trop Med Hyg 1992; 47:
61-69.
-
Ni H, Barrett ADT. Nucleotide and deduced amino acid sequence of
the structural protein genes of Japanese encephalitis viruses from
different geographical locations. J Gen Virol 1995; 76:
401-407.
-
Sumiyoshi H, Mori C, Fuke I, et al. Complete nucleotide sequence of
the Japanese encephalitis virus genome RNA. Virology 1987;
161: 497-510.
-
Ritchie SA, Kline DL. Comparison of CDC and EVS light traps baited
with carbon dioxide and octenol for trapping mosquitoes in Brisbane,
Queensland (Diptera: Culicidae). J Aust Entomol Soc 1995;
34: 215-218.
-
Anderson SG, Price AVG, Nanadai-Koia, Slater K. Murray Valley
encephalitis in Papua and New Guinea: II. Serological survey,
1956-1957. Med J Aust 1960; 2: 410-413.
-
Draffan RDW, Garnett ST, Malone GJ. Birds of the Torres Strait: an
annotated list and biogeographical analysis. The Emu 1983;
83: 207-234.
-
Buescher EL, Scherer WF, McClure HE, et al. Ecologic studies of
Japanese encephalitis virus in Japan. IV. Avian infection. Am J
Trop Med Hyg 1959; 8: 678-688.
-
Boyle DB, Dickerman RW, Marshall ID. Primary viraemia responses
of herons to experimental infection with Murray Valley
encephalitis, Kunjin and Japanese encephalitis viruses. Aust J
Exp Biol Med Sci 1983; 61: 655-664.
-
Bryan JH, O'Donnell MS, Berry G, Carvan T. Dispersal of adult
female Culex annulirostris in Griffith, New South Wales,
Australia: a further study. J Am Mosq Control Assoc 1992; 8:
398-403.
-
Gingrich JB, Nisalak A, Latendresse JR, et al. Japanese
encephalitis virus in Bangkok: factors influencing vector
infections in three suburban communities. J Med Entomol
1992; 29: 436-444.
-
Kay BH, Boreham PFL, Fanning ID. Host-feeding patterns of
Culex annulirostris and other mosquitoes (Diptera: Culicidae)
at Charleville, southwestern Queensland, Australia. J Med
Entomol 1985; 22: 529-535.
-
Hanna J, Barnett D, Ewald D. Vaccination against Japanese
encephalitis in the Torres Strait. Commun Dis Intell 1996;
20: 188-190.
(Received 9 Feb, accepted 27 May 1996) o
Authors' details
Tropical Public Health Unit, Queensland Health, Cairns, QLD.
Jeffrey N Hanna, MPH, FAFPHM, Public Health Physician.
Scott A Ritchie, PhD, Medical Entomologist.
World Health Organization Collaborating Centre for Arbovirus
Reference and Research, Laboratory of Microbiology and Pathology,
Queensland Health, Brisbane, QLD.
Debra A Phillips, BSc, MASM, Supervising Scientist.
Queensland Department of Primary Industries, Cairns, QLD.
Jack Shield, BVSc, Veterinary Officer.
Torres Strait Public Health Program, Queensland Health, Thursday
Island, QLD.
M Clare Bailey, MAIEH, Environmental Health Officer.
Department of Microbiology, The University of Queensland,
Brisbane, QLD.
John S Mackenzie, PhD, FASM, Professor of Microbiology.
Michael Poidinger, PhD, Research Officer (NHMRC).
Southern Zone Public Health Unit, Queensland Health, Upper Mount
Gravatt, QLD.
Bradley J McCall, MPH, FAFPHM, Public Health Physician.
Queensland Health, Thursday Island, QLD.
Phillip J Mills, Executive Officer, Torres Sector.
Reprints: Dr J Hanna, Tropical Public Health Unit,
Queensland Health, PO Box 1103, Cairns, QLD 4870.
Email:
troppubATcitec.qld.gov.au
- Register to be notified of new articles by email - - To top of article - ©MJA1996
< URL: http://www.mja.com.au/> © 1996 Medical Journal of Australia.
Received 24 May 2026, accepted 24 May 2026
- Jeffery H Hanna
- Scott A Ritchie
- Debra A Phillip
- Jack Shield
- John S Mackenzie
- Michael Poidinger
- Bradley J McCall
- Phillip J Mills




