The known: Sleep disorders affect workplace productivity, but their significance for young working adults, for whom diagnosis and management rates are low, is not well understood.
The new: Annual workplace productivity loss was considerably greater among 22‐year‐old working Australians with sleep disorders (22% of participants), particularly undiagnosed insomnia, than among young adults without sleep disorders.
The implications: Screening for and managing sleep disorders in young working adults is inadequate. Occupational health providers and general practitioners could assist with identifying them, and awareness of sleep disorders and their consequences among young adults should be improved.
A recent editorial in The Lancet called for greater attention to sleep medicine, emphasising that “sleep disorders are an underappreciated public health problem” and that these disorders “are very common, cause much distress to both sufferers and their bed partners, and have far‐reaching effects on population health and economic wellbeing.”1 Sleep disorders can occur across the lifespan, but their impact on the productivity and safety of young adults has received only limited attention. The recent report that one in five young Australian adults has a clinically significant sleep disorder, as determined by laboratory polysomnography and validated questionnaires, was the first to draw attention to the high rates of common sleep disorders.2 These conditions, including moderate to severe obstructive sleep apnoea, chronic insomnia, and restless legs syndrome, are often undiagnosed in young working adults.3
Sleep disorders have a considerable impact on productivity and safety in middle‐aged and older adults. They are associated with increased likelihood of road4 and workplace safety incidents,5,6,7 as well as workplace productivity loss, including absenteeism8,9 and presenteeism (being at work but not functioning at full capacity).10 Substantial productivity losses are attributed to sleep disorders in developed countries.11 However, information on their impact on the productivity and safety of young Australian adults is limited.12
The few studies that have examined the relationship between sleep disorders and workplace productivity loss in young adult workers have generally been cross‐sectional studies based on self‐reports of sleep disorders. We prospectively examined workplace productivity losses across a year among young adult workers in whom the presence of the three most prevalent sleep disorders was assessed in overnight laboratory‐based sleep studies as well as by detailed sleep screening.
Methods
Our prospective observational study is part of the Raine Study, a longitudinal birth cohort study in Perth, Western Australia. Generation 1 mothers were recruited during 1989–1992; their children comprise the Generation 2 cohort. We collected follow‐up data when the Generation 2 participants were 22 years of age. The Raine Study is the first to undertake in‐laboratory sleep assessments in a representative sample of young adults as well as to prospectively collect workplace productivity data over twelve months.13
Clinically significant sleep disorders
We analysed data for participants who were currently employed, and who completed at least four hours of laboratory (level 1) polysomnography and detailed sleep assessments, as well as at least three of four quarterly workplace productivity assessments during the twelve calendar months following polysomnography assessment (during 2012–2014).14
Validated methods were used to screen for insomnia and restless legs syndrome. Insomnia was assessed with the Pittsburgh Sleep Symptom Questionnaire for Insomnia (PSSQ‐I);15 insomnia was defined by symptoms being present for at least three months. Restless legs syndrome was defined according to the International Restless Legs Syndrome Study group definition.16 Specifically, the Raine Study asked about the urge to move the legs when sitting or lying down, co‐existing dysesthesia, whether the symptoms were relieved by movement, and whether the symptoms were worse during the evening or at night. Participants were determined to have restless legs syndrome if they met all four symptoms at least five times a month.2 Clinically significant obstructive sleep apnoea was defined by a polysomnography‐derived apnoea–hypopnea index (AHI) of more than fifteen events per hour, or obstructive sleep apnoea syndrome (AHI greater than five and daytime sleepiness symptoms).
Workplace productivity loss
The primary dependent variable was total workplace productivity loss, expressed as hours per worker per year, assessed with the World Health Organization Work Performance Questionnaire (HPQ).17 Total workplace productivity loss comprised total absenteeism (including absenteeism due to sickness) and presenteeism. Participants were asked to complete the HPQ every three months during the year following their overnight sleep study. The conversion of presenteeism scores into lost work time has been described previously.18 Briefly, we subtracted the HPQ productivity rating (0–10, with ten indicating no presenteeism) from ten, converted the result to a percentage, and multiplied it by the mean number of hours worked daily over the past seven days to calculate lost time over the preceding four weeks. Excess total productivity loss was defined as the difference in total productivity loss between people with and without sleep disorders. The population burden of productivity loss associated with having a sleep disorder was calculated as the excess loss multiplied by the sleep disorder prevalence rate, and expressed as hours per 1000 workers per year.
Covariates
Sex was defined as biological sex at birth. Participants were asked whether they were shift workers, and whether they currently had any of the following health and medical conditions or problems, diagnosed by a health professional: anxiety, arthritis or joint pain, asthma, attention problems, back pain, behavioural problems, impaired bladder control, chronic respiratory or breathing conditions, coordination difficulties or clumsiness, coeliac disease, depression, diabetes, eating disorder or weight problems, hay fever or allergies, hearing impairment or deafness, heart conditions, haemochromatosis, intellectual disability, learning difficulties, menstrual problems, migraine or severe headache, neck pain, speech or language problems, thyroid gland disease, and vision deficits. Usual alcohol consumption was reported by participants as number of days per week.19
Statistical analysis
Statistical analyses were conducted in RStudio 2020 (Posit) and R 4.2.3 (R Foundation for Statistical Computing), using the pscl package.20 Incidence rate ratios (IRRs) with 95% bias‐corrected and accelerated confidence intervals (BCa CIs) comparing workplace productivity loss endpoints (total workplace productivity loss, total absenteeism, sickness absenteeism, and presenteeism) in the two participant groups (sleep disorders, no sleep disorders) were estimated in zero‐inflated negative binomial regression models; bias‐corrected parameter estimates were used because of the high number of meaningful zero counts for productivity loss.18 Vuong tests confirmed that these models were superior to zero‐inflated Poisson regression models. Final models were adjusted for sex, shift work status, and number of current medical conditions.
Ethics approval
The Raine Study was approved by the human research ethics committee of the University of Western Australia (RA/4/20/5722), and project approval for this study was obtained from Flinders University (4717).
Results
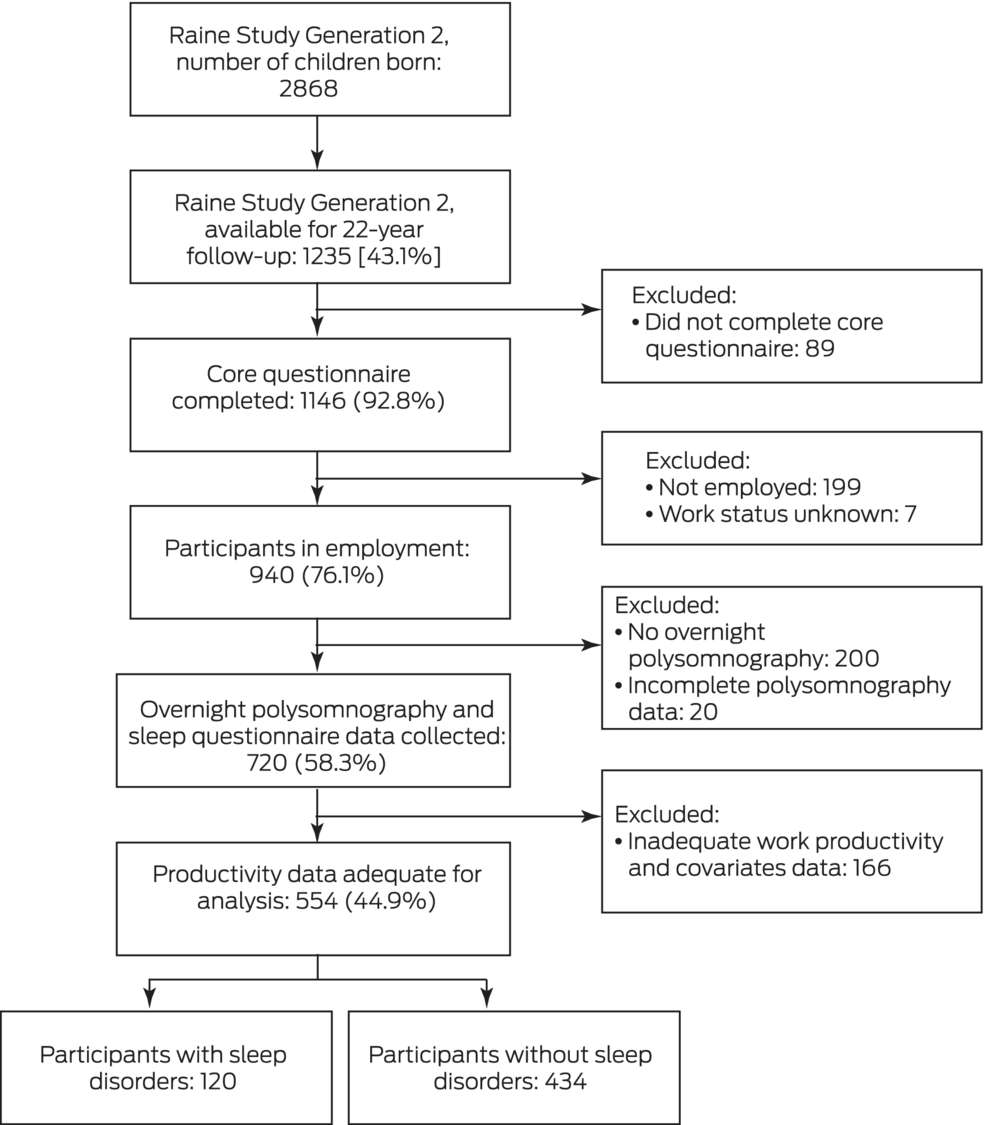
Of the 2868 children in the Raine Study Generation 2, 1235 were available for 22‐year follow‐up data collection (43.1%). A total of 720 employed young adults underwent overnight polysomnography and completed the baseline sleep questionnaire (58.3%) and 554 also completed at least three workplace productivity assessments (44.9%) (Box 1, Box 2). The demographic characteristics of the participants for whom complete sleep and HPQ data were available were similar to those of people who completed only the sleep assessments (Supporting Information, table 1). The education and occupational characteristics of the 554 participants who provided full data were also similar to those derived from Western Australian 2011 population census data for 22‐year‐old people (Supporting Information, table 2).
At least one clinically significant sleep disorder was identified in 120 participants (21.7%): 69 women (23% of female participants) and 51 men (20% of male participants). Ninety participants had insomnia (17%), thirty had clinically significant obstructive sleep apnoea (5.4%), and two restless legs syndrome (0.4%) (Box 2). Seventeen people (14% of those with sleep disorders) had previously been diagnosed with a sleep disturbance by a health professional: fourteen with insomnia, two with obstructive sleep apnoea, and one with both insomnia and restless legs syndrome.
Sleep disorders and workplace productivity loss
The median total workplace productivity loss was greater for participants with sleep disorders (164 hours/year; interquartile range [IQR], 0–411 hours/year) than for those without sleep disorders (30 hours/year; IQR, 0–202 hours/year. Total workplace productivity loss was 40% greater among participants who had sleep disorders than those who did not (adjusted IRR, 1.40; BCa 95% CI, 1.10–1.76). This was predominantly attributable to greater presenteeism (76 [IQR, 0–230] v 10 [IQR, 0–115] hours/year; adjusted IRR, 1.52; BCa 95% CI, 1.18–1.95) and greater total absenteeism (48 [IQR, 0‐142] v 0 [IQR, 0–71] hours/year; adjusted IRR, 1.34; BCa 95% CI, 1.01–1.79) (Box 3). Post hoc analyses identified no statistically significant interaction effects for productivity loss between sleep disorders and shift work status, sleep disorders and sex, or sleep disorders and number of medical conditions (Supporting Information, table 3).
Sensitivity analyses adjusted for numbers of clustered mental health, pain, and respiratory conditions (see footnotes to Box 2) instead of count of medical conditions, or additionally adjusted for body mass index and alcohol consumption, returned similar results to the main adjusted analyses (data not shown).
Excess total productivity loss and population burden of productivity loss attributable to sleep disorders
We estimated that the excess total productivity loss was 134 hours per worker per year (Box 3) and that the population burden of total productivity loss was 28 644 hours per 1000 young workers per year for participants with clinically significant sleep disorders. The total productivity loss associated with insomnia was 28 730 hours/1000 workers/year, and the loss associated with restless legs syndrome was 1968 hours/1000 workers/year. Obstructive sleep apnoea was associated with less productivity loss than those without sleep disorders (–1566 hours/1000 workers/year).
Discussion
We found that total workplace productivity loss in young adults was 40% greater among those meeting the diagnostic criteria for at least one sleep disorder than for those who did not. Young people with sleep disorders lost a median of about four working weeks’ productivity per year, while those without sleep disorders lost less than one working week. In terms of population burden of total productivity loss, this is equivalent to 28 644 hours per 1000 workers with sleep disorders per year, with the burden predominantly attributable to insomnia. Sleep disorders need to be identified and managed in early adulthood to mitigate their impact on workforce productivity.
Self‐reported sleep problems are associated with reduced workplace productivity,10 including in young adults.18 However, prospective evidence from studies in representative population samples including young adult workers is scarce, and prospective studies using the reference standard for identifying obstructive sleep apnoea, in‐laboratory polysomnography, have not previously been reported for young adult workers.
Our finding that total workplace productivity loss was lower in young people with obstructive sleep apnoea than in those without it is novel. Sleepiness in obstructive sleep apnoea is associated with productivity loss.21 The participants in the Raine Study comprise a community‐based cohort, not a clinical sample. Our 22‐year‐old participants did not have high sleepiness scores (as previously reported2), and the rate of severe obstructive sleep apnoea was not high;2 these factors perhaps explain why finding differed from those of studies with middle‐aged and older adults. This association should be further explored in larger samples of young workers with obstructive sleep apnoea to better determine the implications for productivity loss and to quantify any change over time, as it is associated with productivity loss in older adults.21 Productivity loss was reported by the two participants with restless legs syndrome, but this finding should naturally be interpreted with caution, given the small number of people with the condition in our study.
Our findings indicate that total workplace productivity loss in young adults was attributable to presenteeism and total absenteeism, rather than to sickness absenteeism. This may reflect the nature of employment of young adults, which often includes temporary and casual contracts with limited or no leave provisions,22 both of which are associated with presenteeism.23 Further, the low rates of sleep disorder diagnoses reported at baseline by our participants2 may explain why workers were less likely to attribute absenteeism to sleep disorder symptoms (ie, that they missed work because of poor sleep); this may be because of limited awareness that these symptoms could reflect an undiagnosed medical condition.
Presenteeism has financial implications for employers and is prospectively associated with sickness absences.24 By middle age (45–65 years), the prevalence of clinically significant sleep disorders reaches 43%, but these disorders remain underdiagnosed and undertreated.25 The effects of sleep disorders on productivity may consequently increase with age and time in the workforce. The most frequent sleep disorder in young adults is insomnia, but by middle age it is obstructive sleep apnoea.21 It is important to determine whether tailored interventions across adulthood are required to reduce productivity losses associated with sleep disorders more broadly.
Early interventions for identifying and managing sleep disorders are likely to benefit workplace productivity, safety, and individual wellbeing. While one in five of the young working adults in our study met the criteria for a sleep disorder, only 14% of these participants reported that it had been previously diagnosed.2 This suggests that many young workers with sleep disorders are either unaware of their sleep disorder, or they may be aware but have not discussed their sleep with medical care providers. Strategies are needed for increasing awareness of sleep disorders among young adults that encourage them to consult relevant primary care and occupational health providers.
Workplace‐focused strategies can improve the productivity of people with insomnia.26 However, young adults are often underrepresented in relevant studies, and robust, evidence‐based approaches are needed for people in this age group. Young workers who indicate their interest in improving their sleep often report barriers related to time demands, their use of technology during sleep opportunity times,27 and unpredictable habits that limit changes to sleep behaviour.28 These factors are likely to limit the opportunity to engage with health care providers and receive diagnostic advice for sleep. Co‐designing and implementing sleep disorder awareness, diagnosis and management approaches with young adults could improve their engagement. Employers and occupational health care providers could invest in such approaches to improve the diagnosis and management of sleep disorders in young employees, and consequently their workplace productivity.
Limitations
Treatment effects may explain why obstructive sleep apnoea was not associated with workplace productivity loss in this cohort. The Raine Study is a non‐interventional study, but participants were provided with their overnight sleep study results, and some may have subsequently sought treatment for obstructive sleep apnoea. Productivity losses may therefore be greater than reported. Future studies should also take into consideration the potentially confounding effects on workplace productivity of substance use and other self‐medication (eg, with sedatives or stimulants) by people with sleep disorders.
Conclusion
Total workplace productivity loss was 40% greater among 22‐year‐old Australian workers with clinical sleep disorders, and was predominantly related to insomnia. Total workplace productivity loss across twelve months was about four weeks for young people with clinically significant sleep disorders. Our findings highlight the need for interventions in the workplace (and possibly in primary care and other health care settings) for facilitating the diagnosis and management of sleep disorders in younger people to improve workplace productivity.
Box 1 – Selection and participation of Raine Study Generation 2 cohort members in the 22‐year follow‐up study of clinically significant sleep disorders and work productivity
Box 2 – Demographic, sleep, occupational, and health characteristics of the 554 participants in the Raine Study Generation 2 study of clinically significant sleep disorders and work productivity
|
Characteristic |
All participants |
Participants with sleep disorders |
Participants without sleep disorders* |
||||||||||||
|
|
|||||||||||||||
|
Participants |
554 |
120 |
434 |
||||||||||||
|
Age (years), mean (SD) |
22.1 (0.6) |
22.1 (0.6) |
22.1 (0.6) |
||||||||||||
|
Sex (women) |
294 (53%) |
69 (57%) |
225 (52%) |
||||||||||||
|
Body mass index (kg/m2), median (IQR) |
23.9 (21.7–27.1) |
23.9 (21.5–27.6) |
23.8 (21.8–27.1) |
||||||||||||
|
Sleep characteristics † |
|
|
|
||||||||||||
|
Insomnia |
90 (17%) |
90 (76%) |
— |
||||||||||||
|
Missing data |
13 |
1 |
12 |
||||||||||||
|
Clinically significant obstructive sleep apnoea |
30 (5.4%) |
30 (25%) |
— |
||||||||||||
|
Missing data |
0 |
0 |
0 |
||||||||||||
|
Clinically significant restless legs syndrome |
2 (0.4%) |
2 (1.7%) |
— |
||||||||||||
|
Missing data |
3 |
0 |
3 |
||||||||||||
|
Doctor‐diagnosed sleep problem |
21 (3.8%) |
17 (14%) |
4 (0.9%) |
||||||||||||
|
Missing data |
3 |
1 |
2 |
||||||||||||
|
Occupational characteristics |
|
|
|
||||||||||||
|
Shift workers |
156 (28%) |
31 (26%) |
125 (29%) |
||||||||||||
|
Work hours/week, median (IQR) |
35 (15–40) |
32 (18–38) |
35 (15–40) |
||||||||||||
|
Current occupation |
|
|
|
||||||||||||
|
Managers |
19 (3.4%) |
4 (3%) |
15 (3.5%) |
||||||||||||
|
Professionals |
82 (15%) |
11 (9.2%) |
71 (16%) |
||||||||||||
|
Technicians/trade |
79 (14%) |
14 (12%) |
65 (15%) |
||||||||||||
|
Community/personal services |
102 (18%) |
20 (17%) |
82 (19%) |
||||||||||||
|
Clerical/administrative |
96 (17%) |
24 (20%) |
72 (17%) |
||||||||||||
|
Sales |
102 (18%) |
27 (23%) |
75 (15%) |
||||||||||||
|
Machinery operators/drivers |
16 (2.9%) |
4 (3%) |
12 (2.8%) |
||||||||||||
|
Labourers |
56 (10%) |
15 (13%) |
41 (9.5%) |
||||||||||||
|
Missing data |
2 |
1 |
1 |
||||||||||||
|
Health characteristics |
|
|
|
||||||||||||
|
Typical alcohol consumption |
|
|
|
||||||||||||
|
None |
28 (5.1%) |
7 (6%) |
21 (4.8%) |
||||||||||||
|
< 1 day/week |
210 (38%) |
40 (33%) |
170 (39%) |
||||||||||||
|
1–4 days/week |
295 (53%) |
68 (57%) |
227 (52%) |
||||||||||||
|
5–7 days/week |
21 (3.8%) |
5 (4%) |
16 (3.7%) |
||||||||||||
|
Missing data |
0 |
0 |
0 |
||||||||||||
|
Medical conditions, median number (IQR) |
1 (0–2) |
2 (0–3) |
1 (0–2) |
||||||||||||
|
Mental health problems‡ |
84 (15%) |
38 (32%) |
46 (11%) |
||||||||||||
|
Missing data |
2 |
1 |
1 |
||||||||||||
|
Pain§ |
143 (26%) |
48 (40%) |
95 (22%) |
||||||||||||
|
Missing data |
2 |
1 |
1 |
||||||||||||
|
Respiratory conditions¶ |
153 (28%) |
42 (35%) |
111 (26%) |
||||||||||||
|
Missing data |
2 |
1 |
1 |
||||||||||||
|
|
|||||||||||||||
|
IQR = interquartile range; SD = standard deviation. * Includes participants for whom questionnaire responses regarding insomnia and restless legs syndrome were missing and complete polysomnography detected no evidence of obstructive sleep apnoea. † Multiple disorders possible for an individual participant. ‡ Anxiety, depression, or eating or weight disorder. § Arthritis, back pain, migraine, or neck pain. ¶ Asthma, allergy, or general respiratory condition. |
|||||||||||||||
Box 3 – Annual workplace productivity loss, by clinical sleep disorder status
|
Characteristic |
Median productivity loss |
Productivity loss:* |
All participants |
Participants with sleep disorders |
Participants without sleep disorders |
Unadjusted model |
Adjusted model† |
||||||||
|
Number of participants |
554 |
120 |
434 |
|
|
||||||||||
|
Total workplace productivity loss (hours), median (IQR) |
54 (0–246) |
164 (0–411) |
30 (0–202) |
1.48 (1.19–1.85) |
1.40 (1.10–1.76) |
||||||||||
|
Total absenteeism |
0 (0–89) |
48 (0–142) |
0 (0–71) |
1.38 (1.05–1.87) |
1.34 (1.01–1.79) |
||||||||||
|
Sickness absenteeism |
0 (0–48) |
25 (0–90) |
0 (0–42) |
1.37 (1.01–2.13) |
1.23 (0.90–1.76) |
||||||||||
|
Lost performance (presenteeism) |
29 (0–143) |
76 (0–230) |
10 (0–115) |
1.61 (1.27–2.01) |
1.52 (1.18–1.95) |
||||||||||
|
|
|||||||||||||||
|
BCa 95% CI = bias‐corrected and accelerated 95% confidence intervals; IQR = interquartile range. * Incident rate ratios compare the productivity loss across the four endpoints for all 120 participants with sleep disorders and all 434 participants without sleep disorders. † Adjusted for sex, number of diagnosed health conditions, and shift work status. |
|||||||||||||||
Received 1 November 2022, accepted 19 April 2023
- Amy C Reynolds1
- Pieter Coenen2,3
- Bastien Lechat1
- Leon Straker4
- Juliana Zabatiero4
- Kath J Maddison5,6,7
- Robert J Adams8
- Peter Eastwood1
- 1 Flinders Health and Medical Research Institute, Flinders University, Adelaide, SA
- 2 Vrije Universiteit Amsterdam, Amsterdam, The Netherlands
- 3 Amsterdam Public Health Research Institute, Amsterdam, The Netherlands
- 4 Curtin University, Perth, WA
- 5 The University of Western Australia, Perth, WA
- 6 West Australian Sleep Disorders Research Institute, Perth, WA
- 7 Sir Charles Gairdner Hospital, Perth, WA
- 8 Flinders University, Adelaide, SA
Open access:
Open access publishing facilitated by Flinders University, as part of the Wiley – Flinders University agreement via the Council of Australian University Librarians.
We acknowledge the Raine Study participants and their families for their ongoing participation in the study, and the Raine Study team for study coordination and data collection. We acknowledge the University of Western Australia Centre for Sleep Science for allowing us to use their facilities and the contributions of the sleep study technicians. We also thank the National Health and Medical Research Council (NHMRC) for its long term contribution to funding the study over the past thirty years. The core management of the Raine Study is funded by the University of Western Australia, Curtin University, the Telethon Kids Institute, the Women and Infants Research Foundation, Edith Cowan University, Murdoch University, the University of Notre Dame Australia, and the Raine Medical Research Foundation. The Raine Study Generation 2 22‐year follow‐up was funded by NHMRC project grants 1027449, 1044840, and 1021858; polysomnography measures were supported by NHMRC project grant 1084947. Funding was also generously provided by Safe Work Australia.
Amy Reynolds has received research funding from the Sleep Health Foundation, Flinders Foundation, Compumedics, and Sydney Trains, and speaker and consultancy fees from Teva Pharmaceuticals, Sealy Australia, and the Sleep Health Foundation for work unrelated to this study. Kath Maddison has received research funding from Nyxoah, Incannex Healthcare, and Zelira Therapeutics, and speaker and consultancy fees from the Sleep Health Foundation, Melius Consulting, and Invicta Medical for work unrelated to this study. Robert Adams has received research funding from the NHMRC, the Sleep Health Foundation, the Flinders Foundation, the ResMed Foundation, the Hospital Research Foundation, Philips, Sydney Trains, and the Australian government for work unrelated to this study.
- 1. The Lancet. Waking up to the importance of sleep [editorial]. Lancet 2022; 400: 973.
- 2. McArdle N, Ward SV, Bucks RS, et al. The prevalence of common sleep disorders in young adults: a descriptive population‐based study. Sleep 2020; 43: zsaa072.
- 3. Reynolds AC, Lechat B, Melaku YA, et al. Shift work, clinically significant sleep disorders and mental health in a representative, cross‐sectional sample of young working adults. Sci Rep 2022; 12: 16255.
- 4. Barger LK, Rajaratnam SMW, Wang W, et al; Harvard Work Hours Health and Safety Group. Common sleep disorders increase risk of motor vehicle crashes and adverse health outcomes in firefighters. J Clin Sleep Med 2015; 11: 233‐240.
- 5. Uehli K, Mehta AJ, Miedinger D, et al. Sleep problems and work injuries: a systematic review and meta‐analysis. Sleep Med Rev 2014; 18: 61‐73.
- 6. Reynolds AC, Ferguson SA, Appleton SL, et al. Prevalence of probable shift work disorder in non‐standard work schedules and associations with sleep, health and safety outcomes: a cross‐sectional analysis. Nat Sci Sleep 2021; 13: 683‐693.
- 7. Shahly V, Berglund PA, Coulouvrat C, et al. The associations of insomnia with costly workplace accidents and errors: results from the America Insomnia Survey. Arch Gen Psychiatry 2012; 69: 1054‐1063.
- 8. Reynolds AC, Appleton SL, Gill TK, et al. Sickness absenteeism is associated with sleep problems independent of sleep disorders: results of the 2016 Sleep Health Foundation national survey. Sleep Health 2017; 3: 357‐361.
- 9. Godet‐Cayré V, Pelletier‐Fleury N, Le Vaillant M, et al. Insomnia and absenteeism at work. Who pays the cost? Sleep 2006; 29: 179‐184.
- 10. Espie CA, Pawlecki B, Waterfield D, et al. Insomnia symptoms and their association with workplace productivity: cross‐sectional and pre‐post intervention analyses from a large multinational manufacturing company. Sleep Health 2018; 4: 307‐312.
- 11. Streatfeild J, Smith J, Mansfield D, et al. The social and economic cost of sleep disorders. Sleep 2021; 44: zsab132.
- 12. Kessler RC, Berglund PA, Coulouvrat C, et al. Insomnia and the performance of US workers: results from the America insomnia survey. Sleep 2011; 34: 1161‐1171.
- 13. Straker L, Mountain J, Jacques A, et al. Cohort profile: the Western Australian Pregnancy Cohort (Raine) Study–Generation 2. Int J Epidemiol 2017; 46: 1384‐1385j.
- 14. Straker LM, Hall GL, Mountain J, et al; Raine Study 22 Year Follow‐Up Investigator Group. Rationale, design and methods for the 22 year follow‐up of the Western Australian Pregnancy Cohort (Raine) Study. BMC Public Health 2015; 15: 663.
- 15. Okun ML, Kravitz HM, Sowers MF, et al. Psychometric evaluation of the Insomnia Symptom Questionnaire: a self‐report measure to identify chronic insomnia. J Clin Sleep Med 2009; 5: 41‐51.
- 16. Allen RP, Picchietti D, Hening WA, et al; Restless Legs Syndrome Diagnosis and Epidemiology workshop at the National Institutes of Health; International Restless Legs Syndrome Study Group. Restless legs syndrome: diagnostic criteria, special considerations, and epidemiology. A report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Med 2003; 4: 101‐119.
- 17. Kessler RC, Barber C, Beck A, et al. The World Health Organization Health and Work Performance Questionnaire (HPQ). J Occup Environ Med 2003; 45: 156‐174.
- 18. Troelstra SA, Straker L, Harris M, et al. Multimorbidity is common among young workers and related to increased work absenteeism and presenteeism: results from the population‐based Raine Study cohort. Scand J Work Environ Health 2018; 44: 521‐529.
- 19. Hebden L, Kostan E, O'Leary F, et al. Validity and reproducibility of a food frequency questionnaire as a measure of recent dietary intake in young adults. PLoS One 2013; 8: e75156.
- 20. Jackman S. pscl: classes and methods for R developed in the Political Science Computational Laboratory, Stanford University. Version 1.5.5.1. http://CRAN.R‐project.org/package=pscl (viewed Mar 2022).
- 21. Léger D, Stepnowsky C. The economic and societal burden of excessive daytime sleepiness in patients with obstructive sleep apnea. Sleep Med Rev 2020; 51: 101275.
- 22. Chesters J, Cuervo H. Adjusting to new employment landscapes: consequences of precarious employment for young Australians. Economic and Labour Relations Review 2019; 30: 222‐240.
- 23. Reuter M, Wahrendorf M, Di Tecco C, et al. Do temporary workers more often decide to work while sick? Evidence for the link between employment contract and presenteeism in Europe. Int J Environ Res Public Health 2019; 16: 1868.
- 24. Janssens H, Clays E, De Clercq B, et al. The relation between presenteeism and different types of future sickness absence. J Occup Health 2013; 55: 132‐141.
- 25. McArdle N, Reynolds AC, Hillman D, et al. Prevalence of common sleep disorders in a middle‐aged community sample. J Clin Sleep Med 2022; 18: 1503‐1514.
- 26. Bostock S, Luik AI, Espie CA. Sleep and productivity benefits of digital cognitive behavioral therapy for insomnia: a randomized controlled trial conducted in the workplace environment. J Occup Environ Med 2016; 58: 683.
- 27. Appleton SL, Reynolds AC, Gill TK, et al. Waking to use technology at night, and associations with driving and work outcomes: a screenshot of Australian adults. Sleep 2020; 43: zsaa015.
- 28. Paterson JL, Reynolds AC, Duncan M, et al. Barriers and enablers to modifying sleep behavior in adolescents and young adults: a qualitative investigation. Behav Sleep Med 2019; 17: 1‐11.






Abstract
Objective: To examine associations between three clinically significant sleep disorders (chronic insomnia, obstructive sleep apnoea, restless legs syndrome) and workplace productivity losses among young Australian adults.
Design, setting: Prospective, observational study; 22‐year follow‐up of participants in the longitudinal birth cohort Raine Study (Perth, Western Australia).
Participants: Currently employed 22‐year‐old Raine Study participants who underwent in‐laboratory sleep disorder screening for moderate to severe obstructive sleep apnoea (apnoea–hypopnea index of more than fifteen events/hour or obstructive sleep apnoea syndrome) and were assessed for insomnia and restless legs syndrome using validated measures.
Main outcome measures: Total workplace productivity loss over twelve months, assessed with the World Health Organization Health and Work Performance Questionnaire.
Results: Of 1235 contactable 22‐year‐old Raine Study cohort members, 554 people (44.9%; 294 women [53%]) underwent overnight polysomnography, completed the baseline sleep questionnaire, and completed at least three quarterly workplace productivity assessments. One or more clinically significant sleep disorders were identified in 120 participants (21.7%); 90 participants had insomnia (17%), thirty clinically significant obstructive sleep apnoea (5.4%), and two restless legs syndrome (0.4%). Seventeen people (14% of those with sleep disorders) had previously been diagnosed with a sleep disturbance by a health professional, including fourteen with insomnia. Median total workplace productivity loss was greater for participants with sleep disorders (164 hours/year; interquartile range [IQR], 0–411 hours/year) than for those without sleep disorders (30 hours/year; IQR, 0–202 hours/year); total workplace productivity loss was 40% greater for participants with sleep disorders (adjusted incidence rate ratio, 1.40; bias‐corrected and accelerated 95% confidence interval, 1.10–1.76). The estimated population total productivity loss (weighted for disorder prevalence) was 28 644 hours per 1000 young workers per year, primarily attributable to insomnia (28 730 hours/1000 workers/year).
Conclusion: Insomnia is a risk factor for workplace productivity loss in young workers. Tailored interventions are needed to identify and manage sleep disorders, particularly as most of the sleep disorders detected in the Raine Study had not previously been diagnosed.