Clinical record
A 40‐year‐old man was referred to the neuromuscular clinic with generalised fasciculations, raising the concerns for amyotrophic lateral sclerosis (ALS).1 Previously, he was a physically fit gym enthusiast without intercurrent medical problems. He was not prescribed medications and consumed supplements linked to his fitness program (Box 1). His presenting complaint was muscle twitching (Supporting Information video 1 and Supporting Information video 2), which evolved over an eight‐month period. Over the next weeks, he noticed undulating movements (myokymia) and twitching of muscles in his feet. This lasted for hours and continued while asleep. The twitching became increasingly frequent and intense, progressing to involve his proximal limbs, trunk, neck, and facial muscles. He described intermittent cramping of abdominal muscles. Several months later, he developed allodynia and hyperaesthesia of his feet, described as a burning sensation over the soles. He also reported intermittent paraesthesia involving his feet, back and trunk.
On neurological examination, fasciculations were evident over the chest and in all limbs. Cranial nerves were intact. Limb examination confirmed normal tone, power and reflexes. Sensory examination was normal. Nerve conduction studies were normal. F‐wave latencies confirmed normal proximal (spinal) function. Electromyography revealed fasciculation potentials of normal morphology (motor unit amplitude and duration were normal), as opposed to the “malignant” fasciculation potentials typically seen in ALS (highly polyphasic of long duration). Investigation for autoimmune diseases linked to hyperexcitability including antibodies to voltage‐gated potassium channel subsets (CASPR‐2 and LGI1) were negative.2 Neuroimaging of the brain and spine did not reveal structural abnormality. Serum electrolytes, B12, folate and cerebrospinal fluid analysis were normal.
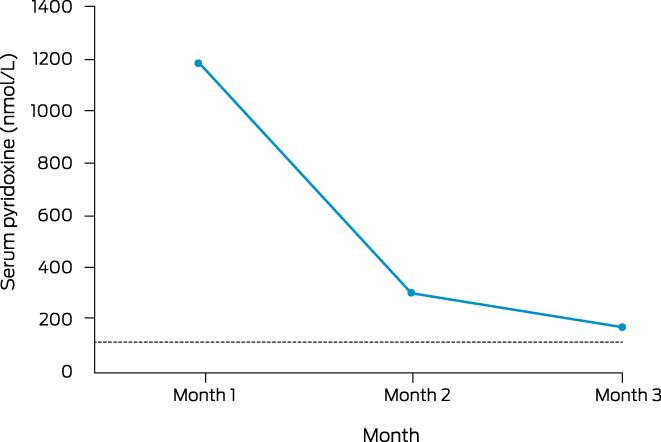
On further discussion about causes of fasciculations and neuropathic symptoms, the patient advised he was taking vitamin supplements, containing a total B6 amount of 95 mg daily. As a consequence, serum B6 level was measured and returned a markedly elevated value at 1179 nmol/mL (reference interval, 35–110 nmol/mL). All supplements were ceased, with precipitous decline of serum pyridoxine (Box 2) associated with a remarkable improvement in symptoms.
On clinical review four months later, there were no fasciculations. However, during this period, he developed dermatitis patches and desquamation involving his feet, leading to a diagnosis of probable pyridoxine toxicity (Naranjo score 5/13).3 In further support, there was resolution of fasciculations after cessation of vitamins.
Discussion
Vitamin B6 is water‐soluble and exists in six forms, namely pyridoxine, pyridoxal, pyridoxamine and their respective phosphorylated forms.4 Pyridoxine‐related neurotoxicity was first reported in 1983, and there have been case reports of sensory‐predominant peripheral neuropathy resulting from excessive pyridoxine consumption.5 However, to date, features of fasciculations and peripheral nerve hyperexcitability as the primary manifestation of pyridoxine toxicity have not been reported.
Pyridoxine toxicity classically manifests as sensory‐predominant polyneuropathy affecting large and small fibres.6 In contrast, the patient developed fasciculations and myokymia, before developing sensory symptoms. Electromyographic characteristics of fasciculations may aid in distinguishing benign fasciculation syndromes from ALS.1 However, there remains a potential overlap between ALS and benign fasciculation syndromes. Serial assessment may help to distinguish the underlying process, particularly in the context of disease progression.7
In terms of pathogenesis of pyridoxine‐related neuropathy, a single mechanism cannot explain the myriad of symptoms observed in each patient. This case adds to that diversity, with symptoms of peripheral nerve hyperexcitability preceding small fibre dysfunction by months. One potential mechanism for pyridoxine‐related neurotoxicity is disruption of γ‐aminobutyric acid (GABA) signalling in the dorsal root ganglion resulting in excitotoxicity.8
Upon cessation of pyridoxine, the patient developed desquamation of his soles, likely a coasting effect observed in pyridoxine toxicity, whereby there is progression of symptoms after withdrawal of the offending agent.9 Even though symptoms of dermatitis have been reported with pyridoxine toxicity, evidence suggests that this is usually related to a phototoxic reaction.10 Alternatively, pyridoxine may cause cellular toxicity and neuronal death through competitive inhibition of the active vitamer (an active form of the vitamin that performs its essential function), pyridoxal phosphate, resulting in apparent B6 deficiency, known as the B6 paradox.4
Dosage or serum pyridoxine concentration to induce toxicity remains unclear. The Netherlands Pharmacovigilance Centre Lareb determined the dosage of vitamin B6 in patients with neuropathy following B6 supplementation varied between 1.5 mg and 100 mg.4 A review by the European Food Safety Authority suggested that toxic effects of pyridoxine usually occurred at doses greater than 100 mg.11 Presently, a definitive threshold for neuronal injury remains unknown, such that a no observed adverse effect level for pyridoxine is yet to be established.11 Susceptibility to neuronal dysfunction from similar pyridoxine doses is likely related to intrinsic genetic variability in gut absorption.8,9
With development of neuropathic symptoms, enquiry about the use of over‐the‐counter preparations may identify causes. Pharmaceutical companies should be encouraged to warn consumers of the potential side effects of pyridoxine, including nerve hyperexcitability. Generalised fasciculations may lead patients and general practitioners to raise concerns about ALS.12 Neurophysiological confirmation of benign fasciculations may suggest an alternative process and promote patient recovery.12
Lessons from practice
- Vitamin B6 is frequently found in high doses, either directly as a pure vitamin or as a component in supplements and in fortified foods.
- Pyridoxine, a major form of vitamin B6, when in excess, is well known to cause an axonal polyneuropathy.
- Pyridoxine may induce generalised fasciculations, mimicking the early features of amyotrophic lateral sclerosis.
- As the exact toxic dosage of pyridoxine is not known, prescribers and retail pharmaceutical stores should exercise caution when initiating vitamin B6 to consumers.
Box 1 – Ingredients of supplements consumed by patient*
|
Super B complex |
Migraine care |
Zinc+ |
|||||||||||||
|
|
|||||||||||||||
|
Thiamine nitrate (50 mg) |
Riboflavin (200 mg) |
Zinc (25 mg) |
|||||||||||||
|
Nicotinamide (100 mg) |
Pyridoxine hydrochloride (15.2 mg) |
Vitamin A (375 μg) |
|||||||||||||
|
Riboflavin (30 mg) |
Calcium folinate (347.2 μg) |
Pyridoxine hydrochloride (30 mg) |
|||||||||||||
|
Calcium pantothenate (50 mg) |
Cyanocobalamin (200 μg) |
Magnesium (35 mg) |
|||||||||||||
|
Pyridoxine hydrochloride (50 mg) |
Magnesium citrate (150 mg) |
Manganese (2 mg) |
|||||||||||||
|
Cyanocobalamin (50 μg) |
Ubidecarenone (75 mg) |
Phenylalanine (amount not stated) |
|||||||||||||
|
Folic acid (300 μg) |
Tanacetum parthenium (40 mg) |
Soya beans (amount not stated) |
|||||||||||||
|
Ascorbic acid (100 mg) |
|
Sulfite (amount not stated) |
|||||||||||||
|
Choline bitartrate (50 μg) |
|
|
|||||||||||||
|
Inositol (50 μg) |
|
|
|||||||||||||
|
Biotin (50 μg) |
|
|
|||||||||||||
|
|
|||||||||||||||
|
* Supplements consumed by patient on a daily basis. |
|||||||||||||||
Provenance: Not commissioned; externally peer reviewed.
- 1. de Carvalho M, Kiernan MC, Swash M. Fasciculation in amyotrophic lateral sclerosis: origin and pathophysiological relevance. J Neurol Neurosurg Psychiatry 2017; 88: 773‐779.
- 2. Park SB, Lin CSY, Krishnan AV, et al. Axonal dysfunction with voltage gated potassium channel complex antibodies. Exp Neurol 2014; 261: 337‐342.
- 3. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30: 239‐245.
- 4. Vrolijk MF, Opperhuizen A, Jansen EHJM, et al. The vitamin B6 paradox: supplementation with high concentrations of pyridoxine leads to decreased vitamin B6 function. Toxicol In Vitro 2017; 44: 206‐212.
- 5. Schaumburg H, Kaplan J, Windebank A, et al. Sensory neuropathy from pyridoxine abuse. A new megavitamin syndrome. N Engl J Med 1983; 309: 445‐448.
- 6. Bacharach R, Lowden M, Ahmed A. Pyridoxine toxicity small fiber neuropathy with dysautonomia: a case report. J Clin Neuromuscul Dis 2017; 19: 43‐46.
- 7. Mills KR. Characteristics of fasciculations in amyotrophic lateral sclerosis and the benign fasciculation syndrome. Brain 2010; 133: 3458‐3469.
- 8. Hadtstein F, Vrolijk M. Vitamin B‐6‐induced neuropathy: exploring the mechanisms of pyridoxine toxicity. Adv Nutr 2021; 12: 1911‐1929.
- 9. Berger AR, Schaumburg HH, Schroeder C, et al. Dose response, coasting, and differential fiber vulnerability in human toxic neuropathy: A prospective study of pyridoxine neurotoxicity. Neurology 1992; 42: 1367‐1370.
- 10. Murata Y, Kumano K, Ueda T, et al. Photosensitive dermatitis caused by pyridoxine hydrochloride. J Am Acad Dermatol 1998; 39: 314‐317.
- 11. Scientific Committee on Food; Scientific Panel on Dietetic Products, Nutrition and Allergies; European Food Safety Authority. Tolerable upper intake levels for vitamins and minerals, February 2006. https://www.efsa.europa.eu/sites/default/files/efsa_rep/blobserver_assets/ndatolerableuil.pdf (viewed Jan 2023).
- 12. Kiernan MC. Fasciculation anxiety syndrome in clinicians: FASICS. Pract Neurol 2020; 20: 433‐434.






Open access
Open access publishing facilitated by The University of Sydney, as part of the Wiley ‐ The University of Sydney agreement via the Council of Australian University Librarians.
No relevant disclosures.