Not all placebo interventions control for the placebo effect, potentially producing misleading results
Placebo‐controlled trials have traditionally been considered the gold standard when comparing the effect of an intervention with no intervention, as they allow the opportunity to differentiate between the therapeutic and placebo effects. However, the results are only valid if appropriate placebo controls are used; otherwise, the placebo control may be a “wolf in sheep's clothing”.
The placebo effect is present in all experiences of interventions, with the magnitude of the effect potentially influenced by several factors.1 The observed summary measure of the primary outcome (eg, mean/median) in each treatment arm or intervention is thus a combination of the real therapeutic effect, the placebo effect, and the natural progression of the condition since treatment initiation (Box 1). The magnitude of each of these components may vary within both person and group. Placebo controls are intended to control for the placebo effect, but where the placebo effect is not equal across interventions, this is unlikely the case and the magnitude of the therapeutic effect remains unknown.
To control for the placebo effect, a placebo control should have no specific therapeutic effect on outcomes of interest and be perceived as real and identical to the primary intervention.2 These placebo criteria are often not possible to achieve for all interventions (eg, lifestyle interventions). Despite this, several studies have compared such interventions with so‐called placebo controls that do not meet these criteria, potentially producing misleading results.
In this Perspective, we discuss three possible problems with so‐called placebo controls that potentially result in failure to control for the placebo effect:
- the placebo control having a specific, therapeutic effect on outcomes of interest;
- the placebo control being distinguishable from the intervention of interest; and
- an emerging issue of open‐label placebos being used to supposedly control for the placebo effect.
Specific therapeutic effects
Placebo controls should have no specific therapeutic effect on outcomes of interest, to control for the placebo effect. However, there are several examples of so‐called placebo controls that contravene these criteria. For example, although saline injections are pharmacologically inert and, therefore, often thought of as having no specific effects, saline injections may improve symptoms3,4 and may have specific physical and chemical effects that could improve outcomes of interest,5 and hence do not necessarily control for the placebo effect.
Importantly, the potential negative effects of so‐called placebo controls should also be considered. Recently, the REDUCE‐IT trial compared the effect of icosapent ethyl with a placebo control (a mineral oil) on cardiovascular disease, concluding that the drug improved cardiovascular outcomes.6 However, further analyses revealed that this difference was not likely due to the effectiveness of the icosapent ethyl but rather to the harm caused by the placebo control, which may have interacted with other medications being taken by the participants.7 It is thus crucial to ensure that the placebo control does not have specific therapeutic effects, even if pharmacologically inert, because such effects may lead to misinterpretation of the therapeutic effects of the drug being tested.
Distinguishable from the intervention of interest
A placebo control must be indistinguishable from the primary intervention. This point not only facilitates blinding of the participant (and potentially outcome assessors) thus reducing bias, but also allows for control of the magnitude of the placebo effect. There is a large body of evidence indicating that not all placebo interventions have the same effect, with the level of invasiveness, dosage and brand differentially affecting individual expectations around intervention outcomes.1 For example, a 2022 study compared an education and exercise program for people with knee osteoarthritis with a so‐called placebo control, where the placebo was a saline injection with arthrocentesis (where required) — two very distinct treatments.8 Having so‐called placebo controls that are distinguishable from the primary intervention does not necessarily control for the placebo effect because the magnitude of the placebo effect may differ from that of the primary intervention.
Open‐label placebos
A new suggested approach to allow for comparison of distinguishable interventions while apparently controlling for the placebo effect is the use of an open‐label placebo — an inert intervention where the participant is made aware of the inert status of the intervention. Open‐label placebos have gained increasing attention, with evidence indicating they are more effective than no treatment in some contexts.9,10,11 However, these findings do not indicate that open‐label placebos control for the placebo effect, and the manner in which they are delivered may also have an impact on the outcomes for the intervention group.
An open‐label placebo was employed in the abovementioned study regarding knee osteoarthritis. In that study, all participants had the candidate interventions described, with statements that the saline injections were “inert, yet with potential beneficial effects that may compare to those of exercise and education” and that “investigators had no treatment preference,“8 presumably in an attempt to make the placebo effect comparable. However, both statements mean the intervention is no longer perceived to be real and may negatively affect the outcomes of the education and exercise intervention. It is critical to appreciate that the participants’ expectations, particularly when manipulated by investigators, may not only increase the expectations of benefits but also reduce them and, therefore, affect measured outcomes. For instance, in a recent study, the same active medication was provided to all participants, but one group was told they had the real medication (the truth) and the other group that they had an “active placebo” (deception), resulting in statistically significant differences in measured effect between the groups, despite the therapeutic effect per se being identical in both groups.12 In the same way, in the 2022 knee osteoarthritis trial, the explanations to participants regarding the potential comparative effectiveness of the saline injection versus the exercise and education intervention may have influenced the measured effect by manipulating the participants’ expectations.8 Open‐label placebos do not typically control for the placebo effect adequately, as the magnitude of the placebo effect is still likely to differ between groups as well as at the individual level.
Implications and recommendations
For interventions involving lifestyle and psychosocial components, we do not and cannot know the mean/median magnitude of the placebo effect for each group. Based on the 2022 trial,8 we provide two hypothetical scenarios (Box 2). For simplicity of comparison, each scenario assumes that the measured mean/median effect of the two interventions is equal. The identical outcome scores may lead one to conclude that combined education and exercise programs have no real therapeutic value. Where the placebo effect is adequately controlled (Box 2, A), this conclusion would be correct. However, where the placebo effect is not equal across the two groups (Box 2, B), this conclusion would be misleading; the education and exercise intervention in fact has a greater therapeutic effect. We have no way of knowing the true magnitude of the placebo effect and, therefore, we are unable to assess the real therapeutic effect of the intervention. As such, we cannot make assumptions about the appropriateness of these interventions as placebo controls.
While placebo‐controlled trials may have value in differentiating the relative real and placebo effects of an intervention, we must accept that they are not possible for many interventions, including lifestyle and psychosocial interventions. It is not always possible to have a control intervention with no specific therapeutic effects that is perceived to be real and appears identical to the primary intervention, which are the requirements of a placebo control.2 Importantly, the impossibility of undertaking placebo‐controlled trials for some interventions does not diminish their potential role as part of the management of chronic conditions, nor the need for and value of pragmatic randomised controlled trials that compare such interventions with, for instance, usual treatment. Although these studies may be criticised for the potential benefits being driven by the placebo effect, we argue that the placebo effect is now an accepted part of any intervention. Indeed, prescribing placebos is common among medical professionals,13,14,15 despite knowing that the treatments they are administering have no therapeutic effect per se. The ethical implications of such practice are beyond the scope of this Perspective, but the practice highlights a willingness to accept that the placebo effect contributes to the effectiveness of therapies.
If we remain focused on placebo‐controlled trials as the gold standard, researchers may feel pressured to conduct trials that are no longer consistent with an acceptance in current practice that placebo effects are an integral part of effective therapy and/or trials that might not necessarily control for the placebo effect. There must be a clear rationale for conducting placebo‐controlled trials, and the limitations of this approach for guiding a necessary evidence base for clinical practice should be acknowledged.16,17,18 We must accept that we cannot have appropriate placebo controls for many interventions, and that calling comparison interventions “placebo controls”, when they do not necessarily control for the placebo effect, is misleading and may result in inappropriate recommendations from health professionals as well as false perceptions of treatment effectiveness by the general public. We should focus on the best available evidence that may also be the best possible evidence, even if that evidence does not consist of placebo‐controlled trials.
Box 1 – A hypothetical example comparing the difference in treatment outcome between three interventions: (1) no intervention, (2) a placebo intervention, and (3) an intervention of interest*
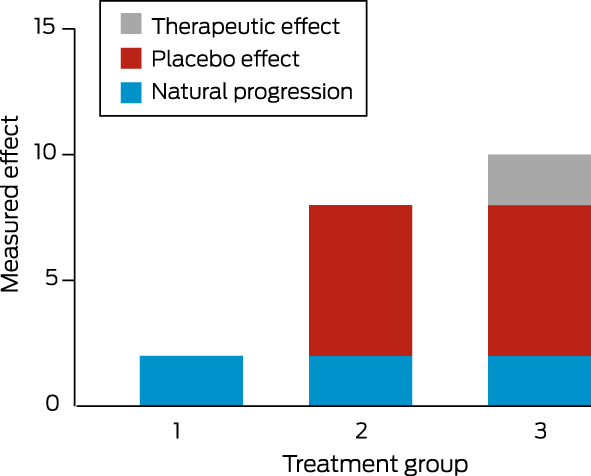
* Where appropriate, placebo interventions have been used, assuming no impact of treatment on the placebo effect.
Box 2 – Hypothetical examples comparing the pain reduction from saline injections with a combined education and exercise program, with both interventions having the same measured effect but different magnitudes of placebo effect*
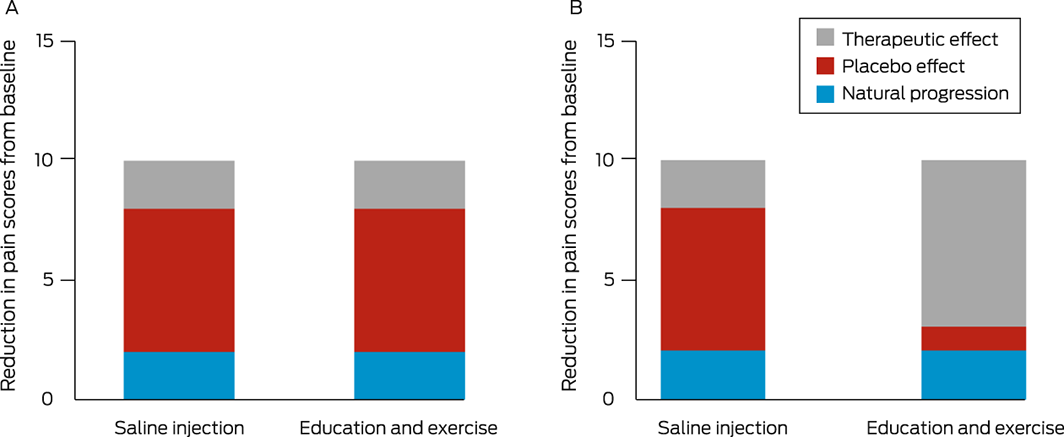
* (A) The placebo effect is adequately controlled. (B) The placebo effect is not equal across the two groups.
Provenance: Not commissioned; externally peer reviewed.
- 1. Meissner K, Linde K. Are blue pills better than green? How treatment features modulate placebo effects. Int Rev Neurobiol 2018; 139: 357‐378.
- 2. Fitzgerald GK, Hinman RS, Zeni J, et al. OARSI clinical trials recommendations: design and conduct of clinical trials of rehabilitation interventions for osteoarthritis. Osteoarthritis Cartilage 2015; 23: 803‐814.
- 3. Altman RD, Devji T, Bhandari M, et al. Clinical benefit of intra‐articular saline as a comparator in clinical trials of knee osteoarthritis treatments: a systematic review and meta‐analysis of randomized trials. Semin Arthritis Rheum 2016; 46: 151‐159.
- 4. Saltzman BM, Leroux T, Meyer MA, et al. The therapeutic effect of intra‐articular normal saline injections for knee osteoarthritis: a meta‐analysis of evidence level 1 studies. Am J Sports Med 2017; 45: 2647‐2653.
- 5. Fazeli MS, McIntyre L, Huang Y, Chevalier X. Intra‐articular placebo effect in the treatment of knee osteoarthritis: a survey of the current clinical evidence. Ther Adv Musculoskelet Dis 2022; 14: 1759720X211066689.
- 6. Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med 2019; 380: 11‐22.
- 7. Ridker PM, Rifai N, MacFadyen J, et al. Effects of randomized treatment with icosapent ethyl and a mineral oil comparator on interleukin‐1β, interleukin‐6, c‐reactive protein, oxidized low‐density lipoprotein cholesterol, homocysteine, lipoprotein(a), and lipoprotein‐associated phospholipase A2: a REDUCE‐IT biomarker substudy. Circulation 2022; 146: 372‐379.
- 8. Bandak E, Christensen R, Overgaard A, et al. Exercise and education versus saline injections for knee osteoarthritis: a randomised controlled equivalence trial. Ann Rheum Dis 2022; 81: 537‐543.
- 9. von Wernsdorff M, Loef M, Tuschen‐Caffier B, Schmidt S. Effects of open‐label placebos in clinical trials: a systematic review and meta‐analysis. Sci Rep 2021; 11: 3855.
- 10. Kaptchuk TJ, Miller FG. Open label placebo: can honestly prescribed placebos evoke meaningful therapeutic benefits? BMJ 2018; 363: k3889.
- 11. Blease CR, Bernstein MH, Locher C. Open‐label placebo clinical trials: is it the rationale, the interaction or the pill? BMJ Evid Based Med 2020; 25: 159‐165.
- 12. Faria V, Gingnell M, Hoppe JM, et al. Do you believe it? Verbal suggestions influence the clinical and neural effects of Escitalopram in social anxiety disorder: a randomized trial. EBioMedicine 2017; 24: 179‐188.
- 13. Faasse K, Colagiuri B. Placebos in Australian general practice: A national survey of physician use, beliefs and attitudes. Aust J Gen Pract 2019; 48: 876‐882.
- 14. Braga‐Simões J, Costa PS, Yaphe J. Placebo prescription and empathy of the physician: a cross‐sectional study. Eur J Gen Pract 2017; 23: 98‐104.
- 15. Tilburt JC, Emanuel EJ, Kaptchuk TJ, et al. Prescribing “placebo treatments”: results of national survey of US internists and rheumatologists. BMJ 2008; 337: a1938.
- 16. Anand R, Norrie J, Bradley J, et al. Fool's gold? Why blinded trials are not always best. BMJ 2020; 368: i6228.
- 17. Kaptchuk TJ. The double‐blind, randomized, placebo‐controlled trial: gold standard or golden calf? J Clin Epidemiol 2001; 54: 541‐549.
- 18. Karjalainen T, Heikkinen J, Busija L, et al. Use of placebo and nonoperative control groups in surgical trials: a systematic review and meta‐analysis. JAMA Netw Open 2022; 5: e2223903.






Open access
Open access publishing facilitated by The University of Adelaide, as part of the Wiley ‐ The University of Adelaide agreement via the Council of Australian University Librarians.
No relevant disclosures.