The known: Late termination of pregnancy is appropriate when fetal aneuploidy, genetic syndromes, or late evolving structural abnormalities are identified, or when the woman has a severe medical condition. Australian late terminations data are limited.
The new: During 2010–2020, the most frequent indications for late termination feticide were central nervous system or cardiac malformations, and aneuploidy (236 of 305 procedures, 77%). Maternal indications accounted for 28 procedures (9%), mostly requests for psycho‐social reasons.
The implications: Despite advances in imaging and prenatal screening, not all fetal abnormalities are detectable early in pregnancy. Safe late termination services are therefore essential for equitable reproductive health care.
The World Health Organization endorses legal, safe, and comprehensive abortion care as an essential component of sexual and reproductive health.1 However, access to safe abortion is difficult for many women, including in Australia,2 because of its limited availability. In Australia, termination of pregnancy care is regulated by the states and territories. Legislative changes to decriminalise abortion commenced only in 1998; South Australia was the last jurisdiction to do so (Termination of Pregnancy Act 2021).
Following an independent review of state legislation, the Termination of Pregnancy Act 2018 came into force in Queensland in December 2018, ending regulation of termination of pregnancy under the state criminal code.3 Termination is now available throughout the first 22 weeks of pregnancy; beyond this point, two medical practitioners must review and support the woman’s request before the procedure can be performed.
Late termination of pregnancy is generally defined as termination after 20 weeks’ gestation, as this is the gestational age beyond which a birth must be legally registered.4 Although most terminations in Australia are undertaken during the first trimester, a small proportion (about 1%) are performed after 20 weeks’ gestation, usually because of late diagnosed major structural anomalies, genetic syndromes, severe fetal growth restriction, or maternal conditions in which continuation of the pregnancy would be significantly detrimental to the mental or physical health of the woman.5 In the United Kingdom, about 1.3% of all terminations in 2021 were performed after 20 weeks’ gestation, and 1% of late terminations were performed because of a substantial risk of physical or mental harm to the woman were the child carried to term. These proportions had not changed markedly since 2011.6
Feticide is a procedure that ensures that the fetus is dead prior to birth, typically achieved by intracardiac injection of potassium chloride to cause asystole. There are no specific Australian guidelines regarding late terminations. Queensland maternity guidelines recommend that feticide should be preferred to surgical termination beyond 22+0 weeks’ gestation;4 in the United Kingdom, feticide is recommended from 21+6 weeks’ gestation.7 In contrast to first trimester abortions,8 late gestation termination of pregnancy requires practitioners with advanced ultrasound and in utero invasive procedural skills who can perform feticide.
The aim of our study was to review rates and indications for late pregnancy feticide at a major Queensland tertiary perinatal centre during the past decade.
Method
We reviewed all late terminations of singleton pregnancies requiring feticide at the Centre for Advanced Prenatal Care, the Royal Brisbane and Women’s Hospital (RBWH) during 1 January 2010 – 31 December 2020. The Centre for Advanced Prenatal Care, one of two tertiary maternal and fetal medicine centres in metropolitan Brisbane, accepts referrals for women with complex pregnancies from beyond its designated catchment region when other providers are unable or unwilling to perform feticides.
We retrieved the data for each case from the RBWH maternal–fetal medicine database (Viewpoint 5, GE Healthcare) and, when necessary, by individual chart review. We did not include cases of selective reduction in multiple pregnancies or very rare cases of late termination without prior feticide.
Before December 2018, women referred for potential feticide were assessed by a multidisciplinary team comprising maternal–fetal medicine and obstetric medicine specialists, a geneticist or genetics counsellor, social workers, neonatologists, paediatric neurologists, paediatric surgeons, and cardiologists, neurologists or mental health specialists. Detailed fetal ultrasound was followed by relevant further investigations, including fetal magnetic resonance imaging (MRI), echocardiography, and genetic testing when indicated. If the woman requested termination of pregnancy or it was appropriate to offer termination as a management option, the case was reviewed by the institutional ethics committee, which made the final decision about proceeding with a late termination.
After the change to Queensland law in December 2018, the initial maternal–fetal medicine/multidisciplinary assessment was followed by review of the termination request by a second maternal–fetal medicine specialist, who independently reviewed all investigations and consultations with other specialists and, when appropriate, undertook a second consultation with the woman. The request for feticide was approved only when both maternal–fetal medicine specialists concurred that it was appropriate. In cases of disagreement, further review by the institutional ethics committee was undertaken to reach a final decision.
At the RBWH, feticide is performed only by maternal–fetal medicine specialists or directly supervised fellows, using a standard technique.9 The procedure is performed under aseptic conditions with continuous ultrasound guidance. Following local anaesthetic infiltration of the mother’s abdominal skin, a 15 cm 20G needle is inserted trans‐abdominally into the uterus and into the fetal left ventricle. After confirming needle placement, 100 μg fentanyl and 3–5 mL 10% potassium chloride is injected. Asystole is confirmed by assessing cardiac activity for at least two minutes before removing the needle; departmental protocol also requires repeat ultrasound scanning 30 minutes after the procedure to confirm persistence of asystole. The woman is administered anti‐D immunoglobulin if appropriate. Following feticide, depending on the woman’s preference or obstetric indication, vaginal or caesarean birth is induced, at the woman’s local hospital when possible.
Indications for feticide were classified as maternal (medical or psycho‐social) or fetal. Fetal indications were further subdivided into cardiac, neurological, thoracic, aneuploidy/genetic syndrome, genito‐urinary, head and neck, infection, pre‐viable pre‐term premature rupture of membranes, severe early onset fetal growth restriction, multiple abnormalities, and other. When available, referral times and birth details (for deliveries in Queensland Health facilities) were also recorded.
Ethics approval
The RBWH Human Research Ethics Committee approved our study (HREC/2021/QRBW/81467).
Results
During 2010–2020, 305 feticides were performed at the RBWH at 22 weeks’ gestation or later (Box 1). All procedures were successful and complication‐free. The median gestational age at feticide was 24+6 weeks (range, 17+0 to 37+1 weeks); the median age of the women was 31 years (range, 15–44 years) (Box 2). The annual number of feticides rose from 20 in 2010 to 54 in 2020; the median gestational age was consistent across the study period.
Of 238 women for whom referral information was available, 201 (84%) had been referred by metropolitan Brisbane health practitioners and facilities; 129 referrals were from outside the usual RBWH catchment area (54%), including 34 from rural and regional Queensland (14%), 58 from tertiary maternity hospitals that do not routinely perform terminations (24%), and 15 from private obstetricians and ultrasound providers (6%) (Box 3).
Most feticides (236 of 305, 77%) were motivated by central nervous system abnormalities (including neural tube defects), chromosomal or genetic abnormalities, or cardiac malformations (Box 4). Aneuploidy was the indication in 40 cases (13%), most frequently trisomy 18 (19 cases) or trisomy 21 (ten cases). The median interval between referral and tertiary maternal–fetal medicine review was five days (range, 0–34 days); 154 of 197 women (78%) were seen within seven days of referral. Most feticides were undertaken within seven days of the initial RBWH review (136 of 197, 69%), but 46 women (23%) waited more than 14 days (Box 5). Delay of feticide pending clarification of fetal outcome was more frequent for cases involving severe fetal growth restriction or pre‐term premature rupture of membranes at pre‐viable gestation ages; feticide was undertaken more than 14 days after initial review in six of 13 cases.
Discussion
In our large Queensland study, we found that the most frequent indications for feticide during 2010–2020 were structural malformations (58%) and chromosomal and genetic abnormalities (22%). Feticide for purely psycho‐social reasons were relatively infrequent (8%).
In contrast to other studies that reported aneuploidy as the reason for 19–24% of feticides, including trisomy 21 for almost 9% of cases,9,10 we found that aneuploidy was the reason for only 40 feticides (13%), including ten instances of trisomy 21 (3%). The smaller proportion may reflect earlier diagnosis and termination following first trimester prenatal aneuploidy screening for cell‐free fetal DNA in the maternal circulation (non‐invasive prenatal testing) or more conventional ultrasound‐based screening.11 In contrast, suspicion of a genetic syndrome often arises later in pregnancy, typically because of the presence of multiple abnormalities.12
The annual number of feticides performed at the RBWH declined between 2010 and 2014. Practitioners may have feared prosecution for terminating pregnancies following a 2009 criminal prosecution for termination of pregnancy in Queensland,13 the first in several years. At this time, abortion was still regulated under the criminal code; in addition, the state government strictly regulated referrals for abortion from outside catchment zones. The decline during 2010–2014 probably reflected women who could not access the service at the RBWH being directly referred to interstate institutions or practitioners for termination of pregnancy services, rather than a decline in requests for this service in Queensland.14
During 2014–2020, the number of feticides performed at the RBWH steadily increased. The establishment of a dedicated ethics committee by the Metro North Hospital and Health Service during this period facilitated rapid reviews, accompanied by clear institutional support for the termination of pregnancy service from the Women’s and Newborn Services division of Metro North Health and the RBWH board. As a result, the number of referrals from other Queensland hospitals (from within and outside the RBWH catchment zone) and by general practitioners and private ultrasound and obstetric care providers increased. Women who might previously have been referred to private or interstate pregnancy termination providers were increasingly referred to the RBWH maternal–fetal medicine service, probably explaining the rise in the annual number of feticides at the RBWH since 2014.
The median gestational age at feticide did not vary markedly during 2010–2020, despite the increasing availability of antenatal genetic testing and the change in Queensland law. The complex nature of the cases detected by more advanced genetic screening probably explains this finding, as further imaging and other investigations, as well as counselling by other experts, are required before reaching a final decision about the appropriateness of feticide. Although first trimester aneuploidy and mid‐trimester fetal anomaly screening are now widely available in Australia, access and affordability can still be barriers for some women.15 Further, women who decline screening are sometimes confronted with the implications of late gestation diagnoses of fetal aneuploidy or of major structural malformation. Additionally, the prognosis associated with a specific abnormality often becomes clearer only at an advanced stage of gestation. For example, mild fetal ventriculomegaly early in pregnancy may develop into severe hydrocephalus over a period of weeks, at which point the severity of the condition and its prognosis is clearer. Further, some abnormalities (eg, duodenal atresia, some skeletal dysplasia) may only evolve later in gestation and consequently not be detected until the second or third trimester of pregnancy. Finally, given the distress for women and their families in these situations, they often need time to assimilate and understand complex medical information and to consider whether late termination is appropriate for them.
Queensland legislation, without a gestational age cut‐off for feticide, allows women adequate time to undergo relevant investigations and receive multidisciplinary counselling before making a final decision about their pregnancy. In our series, almost one in four women who proceeded with a feticide did so more than two weeks after their first consultation at the RBWH, probably because serial scans and further investigations and counselling were needed to aid their decision.
Limitations
The recorded indications for feticide were sometimes broadly expressed, and comparison with medical records was often needed for confirmation. For women who returned to their primary provider for care, post‐procedural data (eg, interval to birth, intrapartum complications) were often not available. We could assess neither the psychosocial effects nor the impact on subsequent pregnancies of feticide. The longer term psychological consequences of late termination of pregnancy should not be underestimated, and sensitive, family‐centred care, with closer than usual surveillance of mother and child, is often required during a subsequent pregnancy.16
Although our retrospective study specifically examined feticide in late termination of pregnancy, it did not include all late terminations at the RBWH. Some were undertaken without prior feticide, despite national and overseas guidelines recommending feticide for terminations after 21+6 weeks’ gestation. Some women and practitioners choose to proceed directly to late termination without feticide when it is clear that the fetus will probably die during the birth process or survive it only briefly. This includes scenarios, for example, of severe fetal pulmonary hypoplasia (following extreme pre‐term premature rupture of membranes or bilateral non‐functioning kidneys) or a major neural tube defect (eg, acrania or severe pre‐term fetal growth restriction) at pre‐viable gestational ages. These clinical scenarios were rare in our study, and usually arose at extremely pre‐term gestation ages when management, particularly regarding the advisability of feticide, was tailored to the individual. Finally, we could not investigate barriers to late termination of pregnancy service, such as financial, family, community, and social opposition, as well as domestic violence and mental health problems.17,18,19
Conclusion
Late termination of pregnancy is challenging for women, but also for health care providers and society.20,21 Advances in prenatal imaging and genomics22 mean that women now have much more information about their fetus and can make more informed decisions about late termination. It is vital that access to late termination is equitable and not restricted by fear of prosecution among medical practitioners. In contrast to other countries, such as the United Kingdom,6 notification of late termination is not mandatory in Australia. Collecting and publishing data about late pregnancy terminations from both public and private health care providers, including those outside maternal–fetal medicine and obstetric services (eg, social workers, mental health and other specialists) is essential for informing decisions about resource allocation and service provision. Equity of access to late termination of pregnancy has been restricted during the COVID‐19 pandemic, and this situation may persist in the face of further infection, economic, conflict, and policy challenges.23,24,25 It is incumbent upon health care providers and legislators to ensure that the infrastructure for termination of pregnancy care in Australia is secure.26
Box 1 – Feticides performed at the Royal Brisbane and Women’s Hospital, 1 January 2010 – 31 December 2020
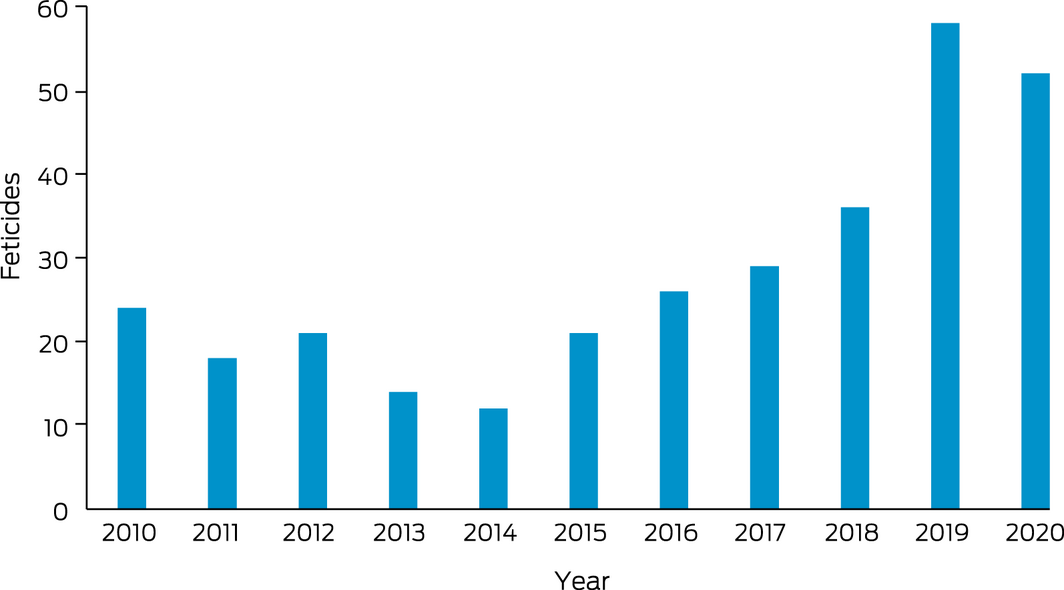
Box 2 – Gestational age at feticide, Royal Brisbane and Women’s Hospital, 1 January 2010 – 31 December 2020*
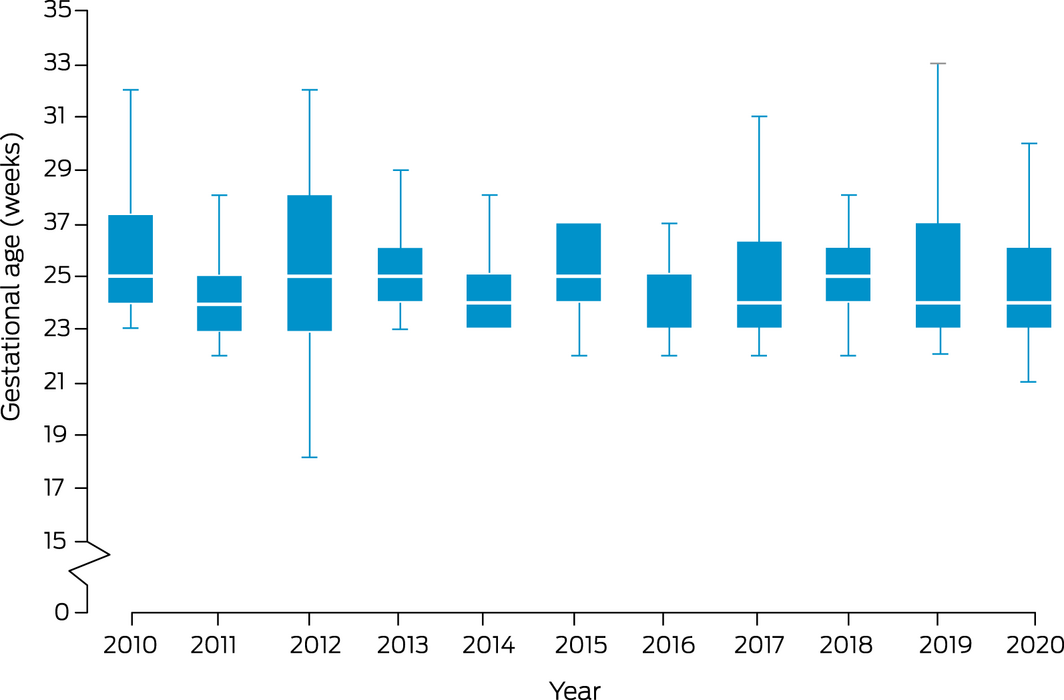
* The boxes depict the interquartile range, with the median marked by a white line; the whiskers depict the minimum and maximum values. For 2016, the median (25 weeks) coincided with the third quartile value.
Box 3 – Characteristics of 305 women who had feticides performed at the Royal Brisbane and Women’s Hospital, 1 January 2010 – 31 December 2020
|
Characteristic |
Value |
||||||||||||||
|
|
|||||||||||||||
|
Age (years), median (range) |
31 (15–44) |
||||||||||||||
|
Parity, median (range) |
1 (0–9) |
||||||||||||||
|
Ethnic background |
|
||||||||||||||
|
European |
153 (77%) |
||||||||||||||
|
Asian |
26 (13%) |
||||||||||||||
|
Aboriginal or Torres Strait Islander |
8 (4%) |
||||||||||||||
|
Other |
12 (6%) |
||||||||||||||
|
Missing data |
106 |
||||||||||||||
|
Referral source |
|
||||||||||||||
|
Metropolitan primary care |
39 (16%) |
||||||||||||||
|
Other tertiary hospital |
58 (24%) |
||||||||||||||
|
Queensland Health metropolitan |
61 (26%) |
||||||||||||||
|
Internal (Royal Brisbane and Women’s Hospital) |
28 (12%) |
||||||||||||||
|
Queensland Health rural/regional |
34 (14%) |
||||||||||||||
|
Interstate |
3 (1%) |
||||||||||||||
|
Private obstetrician |
8 (3%) |
||||||||||||||
|
Private ultrasound practice |
7 (3%) |
||||||||||||||
|
Missing data |
67 |
||||||||||||||
|
|
|||||||||||||||
|
|
|||||||||||||||
Box 4 – Indications for 305 feticides performed at the Royal Brisbane and Women’s Hospital, 1 January 2010 – 31 December 2020
|
Indication* |
Number |
Medical review within seven days of referral |
Median gestation age at feticide, weeks (range) |
||||||||||||
|
|
|||||||||||||||
|
Maternal indications |
28 (9%) |
21 (75%) |
24+0 (22+2 to 28+0) |
||||||||||||
|
Medical |
4 (1%) |
4 (100%) |
24+3 (17+0 to 26+1) |
||||||||||||
|
Psycho‐social |
24 (8%) |
17 (71%) |
24+4 (22+2 to 28+0) |
||||||||||||
|
Fetal indications |
281 (92%) |
221 (79%) |
24+6 (17+0 to 37+1) |
||||||||||||
|
Central nervous system |
110 (36%) |
87 (79%) |
25+3 (17+1 to 37+1) |
||||||||||||
|
Aneuploidy/syndromic |
67 (22%) |
53 (79%) |
25+1 (17+0 to 36+4) |
||||||||||||
|
Cardiac |
59 (19%) |
42 (71%) |
24+3 (22+0 to 30+4) |
||||||||||||
|
Multiple anomalies |
13 (4%) |
13 (100%) |
24+4 (20+4 to 31+2) |
||||||||||||
|
Genito‐urinary |
7 (2%) |
6 (86%) |
23+1 (22+0 to 27+2) |
||||||||||||
|
Severe early onset fetal growth restriction |
7 (2%) |
7 (100%) |
24+3 (23+5 to 26+1) |
||||||||||||
|
PPROM (< 22 weeks) |
6 (2%) |
4 (67%) |
24+3 (22+6 to 27+3) |
||||||||||||
|
Thoracic |
5 (2%) |
5 (100%) |
25+3 (24+1 to 33+0) |
||||||||||||
|
Other† |
7 (2%) |
7 (100%) |
25+6 (23+2 to 28+1) |
||||||||||||
|
|
|||||||||||||||
|
PPROM = pre‐term premature rupture of membranes. * More than one indication possible. † Includes two cases of infection (both cytomegalovirus), head and neck abnormalities, and idiopathic hydrops. |
|||||||||||||||
Box 5 – Time from maternal–fetal medicine review to feticide for 197 of 305 feticides at the Royal Brisbane and Women’s Hospital, 1 January 2010 – 31 December 2020
|
|
Referral to first maternal–fetal medicine review |
First maternal–fetal medicine review to feticide |
|||||||||||||
|
|
|||||||||||||||
|
Under seven days |
154 (78%) |
136 (69%) |
|||||||||||||
|
7–14 days |
8 (4%) |
15 (8%) |
|||||||||||||
|
More than 14 days |
35 (18%) |
46 (23%) |
|||||||||||||
|
|
|||||||||||||||
|
|
|||||||||||||||
Received 31 March 2022, accepted 19 July 2022
- Shannyn Rosser1
- Renuka Sekar1
- Johanna Laporte1
- Gregory J Duncombe2
- Alexa Bendall1
- Christoph Lehner1
- Carol Portmann3
- Pauline McGrath1
- Karin Lust1
- Peter Ganter1
- Sailesh Kumar1
- 1 Royal Brisbane and Woman's Hospital, Brisbane, QLD
- 2 Centre for Advanced Prenatal Care, Royal Brisbane and Woman's Hospital, Brisbane, QLD
- 3 Marie Stopes Australia, Brisbane, QLD
No relevant disclosures.
- 1. World Health Organization. Safe abortion: technical and policy guidance for health systems. Second edition. Geneva: WHO, 2019. https://apo.who.int/publications/i/item/safe‐abortion‐technical‐and‐policy‐guidance‐for‐health‐systems‐second‐edition (viewed July 2022).
- 2. LaRoche KJ, Wynn LL, Foster AM. “We’ve got rights and yet we don’t have access”: exploring patient experiences accessing medication abortion in Australia. Contraception 2020; 101: 256‐260.
- 3. Queensland Parliament. Termination of Pregnancy Act 2018. https://find.search.qld.gov.au/s/redirect?collection=qld‐gov&url=https%3A%2F%2Fwww.legislation.qld.gov.au%2Fview%2Fpdf%2Finforce%2Fcurrent%2Fact‐2018‐023&auth=D82cZxvmVXTl24Wet2iHTQ&profile=qld&rank=6&query=Termination+of+Pregnancy+Act+2018 (viewed May 2022).
- 4. Queensland Health. Maternity and neonatal clinical guidelines. Termination of pregnancy (Queensland clinical guidelines, MN19.21‐V6‐R24). Oct 2019. https://www.health.qld.gov.au/__data/assets/pdf_file/0029/735293/g‐top.pdf (viewed June 2022).
- 5. Australian Institute of Health and Welfare. 2004 National Drug Strategy household survey: detailed findings (Cat. no. PHE 66; Drug statistics series no. 16). Canberra: AIHW, 2005. https://www.aihw.gov.au/reports/illicit‐use‐of‐drugs/2004‐ndshs‐detailed‐findings/summary(viewed Jan 2022).
- 6. Office for Health Improvement and Disparities. Abortion statistics, England and Wales: 2021. 21 June 2022. https://www.gov.uk/government/statistics/abortion‐statistics‐for‐england‐and‐wales‐2021/abortion‐statistics‐england‐and‐wales‐2021 (viewed May 2022).
- 7. Royal College of Obstetricians and Gynaecologists. Termination of pregnancy for fetal abnormality in England, Scotland and Wales. May 2010. https://www.rcog.org.uk/media/21lfvl0e/terminationpregnancyreport18may2010.pdf (viewed May 2022).
- 8. Renner RM, Brahmi D, Kapp N. Who can provide effective and safe termination of pregnancy care? A systematic review. BJOG 2013; 120: 23‐31.
- 9. Pasquini L, Pontello V, Kumar S. Intracardiac injection of potassium chloride as method for feticide: experience from a single UK tertiary centre. BJOG 2008; 115: 528‐531.
- 10. Muin DA, Otte P, Scharrer A, et al. Temporal changes in epidemiological profile and fetal indications for late termination of pregnancy: a retrospective single‐center study. Arch Gynecol Obstet 2021; 304: 935‐942.
- 11. Lassey SC, Reiff ES, Dobson L, et al. The influence of noninvasive prenatal testing on gestational age at time of abortion for aneuploidy. Prenat Diagn 2017; 37: 635‐639.
- 12. Svirsky R, Pekar‐Zlotin M, Rozovski U, Maymon R. Indications for genetic testing leading to termination of pregnancy. Arch Gynecol Obstet 2019; 300: 1221‐1225.
- 13. Petersen K. Abortion laws and medical developments: a medico‐legal anomaly in Queensland. J Law Med 2011; 18: 594‐600.
- 14. Nickson C, Shelley J, Smith A. Use of interstate services for the termination of pregnancy in Australia. Aust N Z J Public Health 2002; 26: 421‐425.
- 15. Bowman‐Smart H, Savulescu J, Mand C, et al. “Small cost to pay for peace of mind”: women’s experiences with non‐invasive prenatal testing. Aust N Z J Obstet Gynaecol 2019; 59: 649‐655.
- 16. Biggs MA, Upadhyay UD, McCulloch CE, Foster DG. Women’s mental health and well‐being 5 years after receiving or being denied an abortion: a prospective, longitudinal cohort study. JAMA Psychiatry 2017; 7: 169‐178.
- 17. Cashman C, Downing SG, Russell D. Women’s experiences of accessing a medical termination of pregnancy through a Queensland regional sexual health service: a qualitative study. Sex Health 2021; 18: 232‐238.
- 18. Black KI, Douglas H, de Costa C. Women’s access to abortion after 20 weeks’ gestation for fetal chromosomal abnormalities: views and experiences of doctors in New South Wales and Queensland. Aust N Z J Obstet Gynaecol 2015; 55: 144‐148.
- 19. Doran FM, Hornibrook J. Barriers around access to abortion experienced by rural women in New South Wales, Australia Rural Remote Health 2016; 16: 3538.
- 20. Habiba M, Da Fre M, Taylor DJ, et al; EUROBS Study Group. Late termination of pregnancy: a comparison of obstetricians’ experience in eight European countries. BJOG 2009; 116: 1340‐1349.
- 21. Cations M, Ripper M, Dwyer J. Majority support for access to abortion care including later abortion in South Australia. Aust N Z J Public Health 2020; 44: 349‐352.
- 22. Corbacıoğlu A, Aslan H, Aydın S, et al. Trends in fetal indications for termination of pregnancy between 2002 and 2010 at a tertiary referral centre. J Turk Ger Gynecol Assoc 2012; 13: 85‐90.
- 23. Bayefsky MJ, Bartz D, Watson KL. Abortion during the Covid‐19 pandemic: ensuring access to an essential health service. N Engl J Med 2020; 382: e47.
- 24. Bojovic N, Stanisljevic J, Giunti G. The impact of COVID‐19 on abortion access: insights from the European Union and the United Kingdom. Health Policy 2021; 125: 841‐858.
- 25. Sifris R, Penovic T. Barriers to abortion access in Australia before and during the COVID‐19 pandemic. Women’s Studies International Forum 2021; 86: 102470.
- 26. Flowers P. Late termination of pregnancy: an internationally comparative study of public health policy, the law, and the experiences of providers (report for the Catherine Helen Spence Scholarship). Nov 2020. https://www.sa.gov.au/__data/assets/pdf_file/0004/713290/Late‐Termination‐of‐Pregnancy‐CHS‐Report.pdf (viewed May 2022).





Abstract
Objective: To review rates of and indications for late pregnancy feticide at a major Queensland tertiary perinatal centre over the past decade.
Design: Retrospective cohort study.
Setting, participants: The Centre for Advanced Prenatal Care at the Royal Brisbane and Women’s Hospital, a tertiary perinatal centre; feticides of singleton pregnancies of at least 22 weeks’ gestation, 1 January 2010 – 31 December 2020.
Main outcome measures: Indications for feticide; median gestational age at feticide; referral source; time between referral, maternal–fetal medicine review, and feticide.
Results: During 2010–2020, 305 feticides were undertaken at 22 weeks’ gestation or later. The annual number of feticides increased from 20 in 2010 to 54 in 2020. The median gestational age at feticide was consistent across the decade (24+6 weeks; range, 17+0 to 37+1 weeks). The most frequent fetal indications for feticide were neurological abnormalities (110 of 305, 36%), aneuploidy or genetic syndromes (67, 22%), and cardiac malformations (59, 19%). Most women were seen for review within seven days of referral for feticide (154 of 197 for whom this information was available, 78%; median, five days; range, 0–34 days), and 136 of 197 feticides (69%) were undertaken within seven days of the initial maternal–fetal medicine review.
Conclusions: Most late pregnancy feticides were performed because of fetal indications, primarily structural malformations or genetic abnormalities. Despite advances in prenatal imaging and diagnosis, late termination of pregnancy remains a necessary option in some pregnancies with maternal or fetal indications, and equitable access to late termination of pregnancy services is a vital component of reproductive health care.