The known: A good recovery and return home after surgery are important for both patients and the health care system. The number of days at home — reduced by complications, discharge to a care facility, re‐admission, and death — can be quantified as the “days alive and at home during the 30 days after surgery” (DAH30).
The new: DAH30 was lower for women and older people, and negatively associated with comorbidity, poor physical status, complexity of surgery, and hospital costs.
The implications: As a highly practical proxy measure of quality of care, DAH30 could be used to assess the overall improvement in patient outcomes after surgery.
Hospitals, governments, and private funders of health care strive to provide high value surgical care,1 reduce the number of avoidable complications, and maximise economic efficiency.2,3 Post‐surgical complications increase hospital length of stay and health care costs.4,5 Enhanced recovery after surgery (ERAS) programs aim to reduce complication rates and health care costs.6
Hospital length of stay is often used as a surgical outcome measure and as a proxy for hospital costs. However, length of stay does not take re‐admissions into account, and is misleading if cut short by in‐hospital death,7 premature discharge home,8 or early transfer to a residential care facility.9 Length of stay, despite being a driver of hospital costs,10 is consequently an unreliable measure of patient outcomes and quality of care.11
The number of days alive and at home during the 30 days following surgery (DAH30) has been validated as a surgical outcome measure in several countries.9,12,13 In a Swedish study, lower DAH30 was associated with patients at higher medical risk, more complex surgical procedures, and serious post‐surgical complications.9 The 12‐month risk of death was seven times as high for people with DAH30 of eight or fewer days as for other patients.9 DAH30 captures the impact of peri‐operative complications, including those not otherwise detected or reported in routine hospital data, such as complications that develop after discharge from hospital.
However, the relationship between DAH30 and total hospital costs has not been examined. We therefore evaluated DAH30 as a potential measure of the quality of surgical care and of hospital costs in the private health sector in Australia by assessing its relationship with patient and surgical factors (criterion validity) and hospital costs.
Methods
In our cohort study, we analysed de‐identified data provided by Medibank Private, the largest private health insurer in Australia. The original data were prospectively collected during private hospital admissions linked with health insurance benefits provided to Medibank Private policy holders. Our report complies with the Reporting of studies Conducted using Observational Routinely collected health Data (RECORD) guidelines.14 Our study protocol and programming codes are available on request.
Study population
We included data for all adult patients (18 years or older) with Medibank Private health insurance who underwent inpatient elective or emergency surgery with anaesthesia in Australian hospitals (private and public) during 1 January 2016 – 31 December 2017. We included only the first admission for a surgical procedure with anaesthesia for each patient. Patients who underwent day case procedures and those living in residential care prior to admission were not included.
Data source
A Medibank Private data analyst extracted and de‐identified the data,15 and password‐protected and encrypted datasets were transferred to a secure server at Monash University using managed file transfer software (MOVEit, Ipswitch). All data were anonymised and the identities of surgeons and hospitals not disclosed. Data were grouped by state and territory; because of the low volume of activity, Northern Territory data were combined with those for South Australia.
The dataset included hospital claims data, hospital casemix protocol information, and Medicare Benefits Schedule (MBS)16 data related to diagnoses and interventions. Data fields included patient demographic characteristics, American Society of Anesthesiologists (ASA) physical status (Supporting Information, part 1), location (state), admission and discharge details, re‐admissions (planned or unplanned) within 30 days of discharge, unplanned return to theatre, intensive care admission, mode of separation (home, rehabilitation, hospital transfer, nursing home, died), MBS item numbers, and hospital and hospital‐in‐the‐home charges.
We selected four high volume procedures, identified by MBS code, for subgroup analyses17 (primary knee arthroplasty, primary hip arthroplasty, hemicolectomy, and prostatectomy), as well as one low volume procedure (common femoral endarterectomy) (Supporting Information, part 2).
Assumptions
It was assumed that the date of surgery was the date of hospital admission and that the date difference between admission and discharge for inpatient surgery admissions was at least one day. Admission to hospital from a residential aged care facility was indicated by the hospital admission mode. Discharge to residential aged care facility was indicated by hospital separation mode; subsequent admission to a residential aged care facility after discharge could not be determined. Only in‐hospital deaths could be inferred from the dataset (hospital separation mode).
Variability
Unexplained variation in care was defined as a surgery‐specific median DAH30 difference of one day or more, after adjusting for all predictor variables, in any state or territory.
Criterion validity
Validity refers to whether DAH30 is an accurate measure of peri‐operative risk or outcome. DAH30 has face validity (ie, it is subjectively regarded as an appropriate measure of quality of care), and was associated with known patient risk and surgical risk factors and with post‐surgical complications in a Swedish population (criterion validity).9 We undertook criterion validity evaluations in the Australian dataset by measuring associations of DAH30 with patient age, ASA physical status, Charlson comorbidity index score, emergency surgery, duration of surgery, and need for post‐surgical mechanical ventilation or other intensive care.
Calculation of total hospital costs and DAH30
Total hospital and associated health care costs for both the index admission and re‐admissions were derived from Medibank Private data fields. This included all hospital charges (operating room, high dependency/intensive care unit care, prostheses, pharmacy, other), as well as additional charges for hospital‐in‐the‐home, coronary care unit, rehabilitation, and palliative care. Doctors’ fees were not included, nor were government reimbursements paid to private hospitals (daily accommodation fees and additional payments based on casemix).
DAH30 was calculated using the published method (Supporting Information, part 3).9 In brief, it is defined as 30 days less the numbers of inpatient hospital and nursing facility days and of re‐admission days. If a patient died within 30 days of surgery, DAH30 was scored as zero, irrespective of whether they had spent time at home.
Statistical analysis
The primary analysis included data for all eligible patients with complete case data. We assumed that at least 1000 patients per high volume surgical group would provide more than 90% power to identify clinically meaningful differences in DAH30.
DAH30 was analysed, by demographic and clinical characteristics, using quantile regression.18 Given the large sample size, we estimated confidence intervals using the asymptotic distribution. Adjusted models included the variables age category, sex, ASA physical status, location (state), Charlson comorbidity index score (part A only)19 derived from International Classification of Diseases, tenth revision, Australian modification (ICD‐10‐AM) hospital codes (Supporting Information, part 4), surgery time (quartiles), surgical urgency, surgical risk,20 and any requirement for higher dependency care (intensive care, coronary care, or mechanical ventilation).
Given the high proportion of incomplete data for ASA physical status, we undertook a second analysis in which we applied a general missing data approach combining imputation analysis and inverse probability weighting,21 using the R MultiRobust package.22 For this analysis, we were required to dichotomise ASA physical status scores (ie, 1 or 2 v 3 or 4). Patient age was treated in two different ways: first, grouped into deciles for people aged 40 years or more, with 18–40 years the reference category; second, B‐splines were used to avoid categorisation and to relax the assumption of linearity with age.23 A similar strategy was applied to duration of surgery.
The association between DAH30 and total hospital costs was quantified with the Spearman rank correlation coefficient (ρ). We also analysed the association after log‐transforming total hospital costs, using the geometric mean for DAH30.
All analyses were performed in R 4.1.1 (R Foundation for Statistical Computing). All tests were two‐sided (α = 0.05) and uncorrected for multiple comparisons.
Ethics approval
Medibank Private and the Alfred Hospital Ethics Committee (736/19) approved our study.
Results
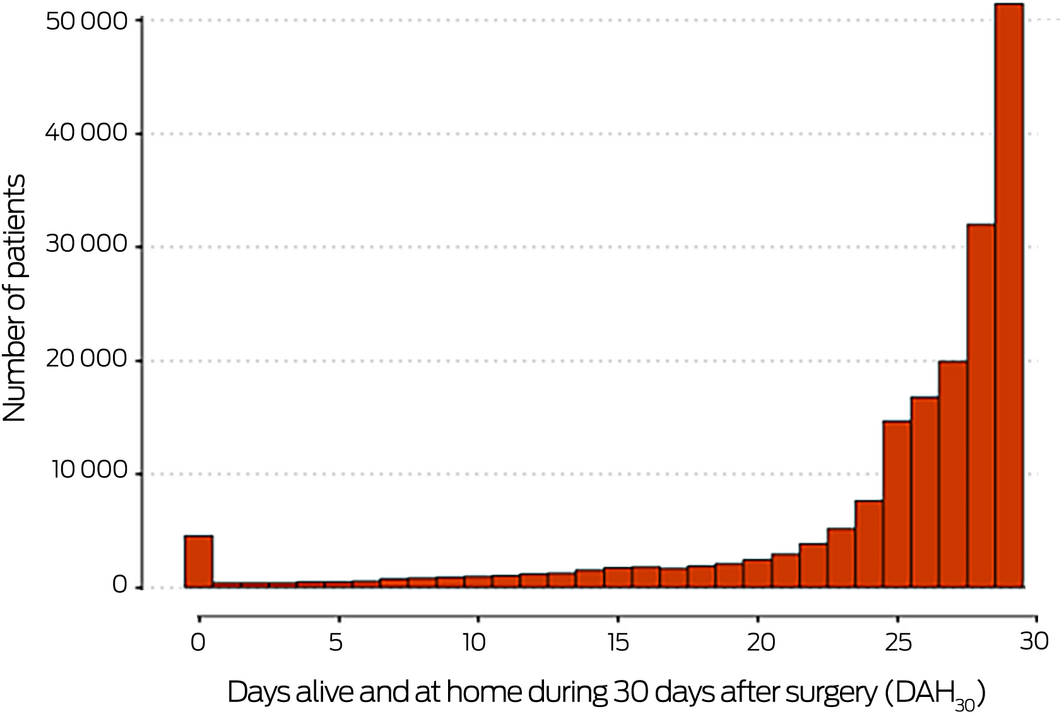
Of 2 213 760 Medibank Private records (1 January 2016 – 31 December 2017), 181 281 were eligible for our analysis (Supporting Information, figure 1). ASA physical status data were missing for 54 493 patients (30.1%); summary patient‐related and peri‐operative characteristics were similar for complete and incomplete cases (Box 1). The median age of the 126 788 patients included in our complete case analysis was 62 years (interquartile range [IQR], 47–73 years), 72 872 were women (57%), and 115 117 had undergone elective surgery (91%). The median hospital cost was $10 358 (IQR, $6624–20 174) per patient (Box 1); the median DAH30 was 27.1 days (IQR, 24.2–28.8 days) (Box 2).
DAH30 and patient and surgical factors
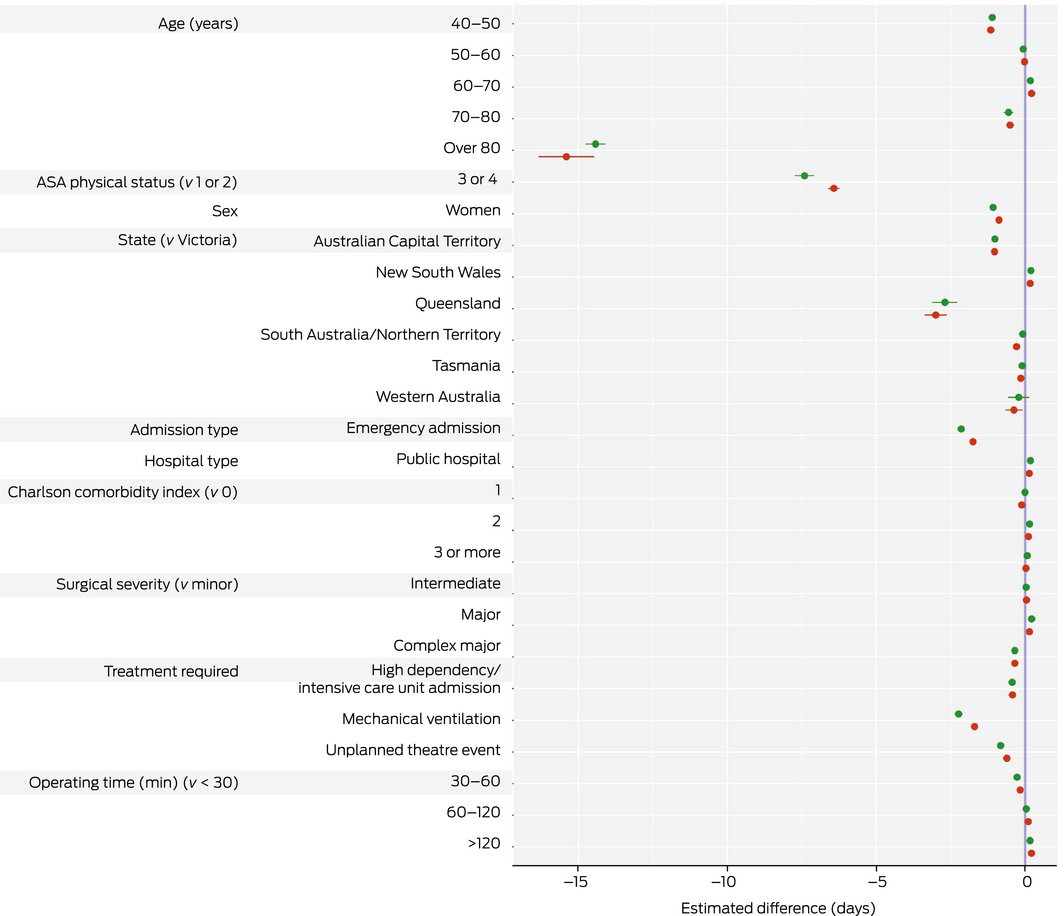
In the complete case analysis, median DAH30 declined with age, comorbidity score, ASA physical status score, and surgical severity, and was lower following emergency admissions, longer duration (greater than 120 minutes) or more extensive surgery, and was also lower for women. The median DAH30 varied between states, including after adjusting for casemix, but the differences were not clinically significant (ie, they were less than one day) (Box 3). In the multiple imputation analysis, median DAH30 was lowest for patients aged 80 years or more, who had ASA physical status scores of 3 or 4, who required emergency surgery or post‐surgical mechanical ventilation for more than 24 hours, or who were admitted to intensive care for more than 24 hours (Box 4).
The impact of most patient and surgical characteristics on median DAH30 was greatest at extreme values (Supporting Information, figure 2). Secondary analyses of median DAH30 using B‐spline regression for age or duration of surgery yielded similar results to the primary analysis, but with evidence of non‐linearity (Supporting Information, figures 3 and 4, table 1).
The missing ASA physical status (dichotomised) data analysis also yielded similar findings (Supporting Information, table 2). Multivariate quantile regression analysis of the 25th centile and 75th centile identified similar effects for most variables (Supporting Information, table 3). The relationships between patient and peri‐operative risk factors and DAH30 differed between the one low volume and four high volume procedures assessed (Supporting Information, table 5, figures 5–9).
Association of DAH30 with hospital costs
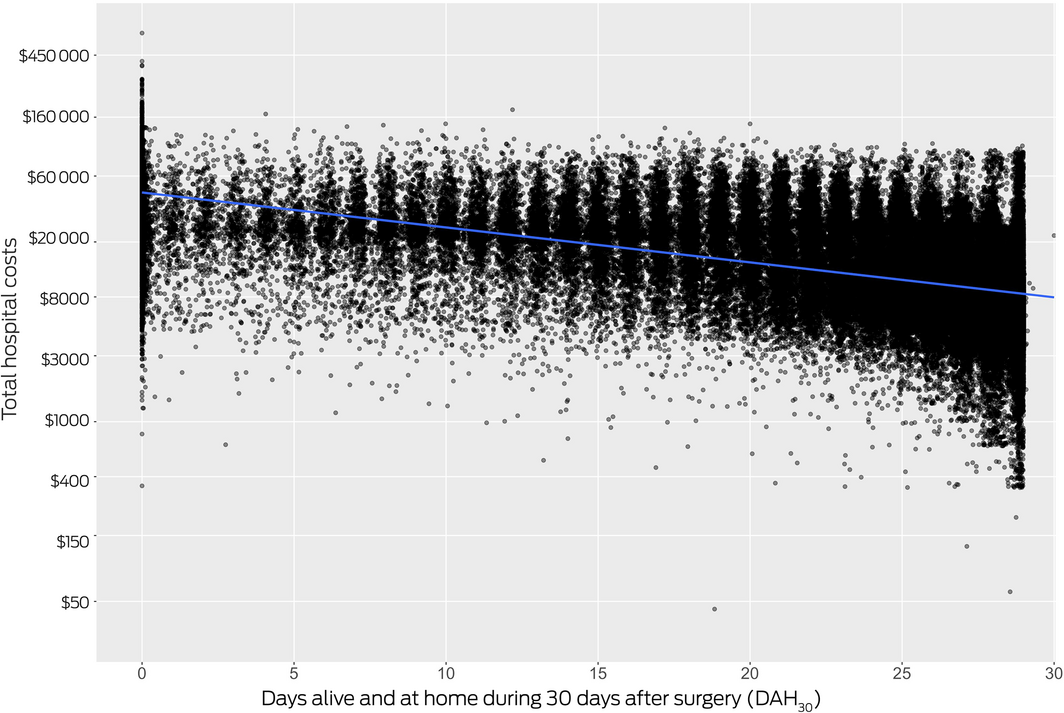
We detected a moderate association between DAH30 and total hospital cost (ρ = –0.60; P < 0.001) (Box 5). Using the geometric mean estimated in the multivariate model of DAH30 and total hospital costs, the coefficient for DAH30 was 0.958 (95% CI, 0.957–0.959) (Supporting Information, table 6); that is, a one‐day increase in time at home was associated with a mean reduction in total hospital cost of 4.2% (95% CI, 4.1–4.3%).
Discussion
In our analysis of Medibank Private data for private sector inpatient surgical care in Australia, the median DAH30 for patients was 27.1 days, and lower DAH30 was associated with higher total hospital costs. Our study is the first to examine the association of DAH30 with health economic outcomes and to examine the potential of DAH30 as a quality‐of‐care metric. The strong associations with patient and surgical risk factors in our study affirmed in Australian private hospital care the criterion and predictive validity of the DAH30 reported overseas.9
About two‐thirds of elective surgery in Australia is undertaken in the private hospital system.24 The dataset we analysed includes hospitals from across Australia, and analysing patient‐ rather than hospital‐level data means that a diverse range of private hospitals were represented, differing in size, location, and ownership. On the other hand, we could not assess between‐hospital variability in costs that could influence the relationship between DAH30 and costs.
Unexplained variation in DAH30 as a quality indicator for surgical care requires further investigation, including the influence of surgical specialty and local practice patterns. Without hospital‐level data, we could analyse risk‐adjusted variation in DAH30 only at the state/territory level. We plan to re‐examine in a future analysis of Medibank Private data our definition of unexplained variation at the hospital and surgeon levels. The private health insurance dataset is ideal for this purpose because it includes patient‐level data from across the Australian private healthcare system, obviating the need for data linkage to obtain a similar dataset for the public hospital system, capturing multiple admissions at different hospitals. The performance of individual surgeons can vary between hospitals, and future analyses need to take this interaction into account.
As our quantile regression analysis indicates, the decline in DAH30 with patient age is not linear, but accelerates markedly for people older than 70 years of age. The effect of surgery duration for patients in the highest DAH30 quartile was similarly non‐linear. Median DAH30 declined with patient age, the number and severity of comorbid conditions, and declining functional status, and was also lower for women. Surgical factors associated with lower DAH30 included more extensive or emergency surgery, surgery duration longer than two hours, unplanned return to the operating theatre, high dependency or intensive care unit admission for 24 hours or more, and post‐surgical mechanical ventilation for 24 hours or more. The directions of the covariate effects were consistent with clinical experience, providing compelling evidence for face and construct validity, and consistent with other studies.9,13,25 The differences in DAH30 highlight the cumulative physical deconditioning and economic cost associated with post‐surgical complications in people at high medical risk.
Limitations
Analysing a dataset from the largest Australian private health insurer, one major purpose of which is to capture potentially recoverable costs, was appropriate for our research question. Its national coverage, our use of quantile regression to model the full range of DAH30 values, and assessing the differential effects of covariates across this range rather than modelling the mean or median values alone, were strengths of our study.
However, the Medibank Private dataset was not designed for research purposes, and may overestimate the total costs of surgical care. Shortcomings of administrative datasets include the possibility of misclassification (eg, comorbidity scores were derived from hospital coding information), unmeasured confounding (factors not covered by the dataset could not be included in our analyses), and missing data. We could impute missing values for ASA physical status, but other factors were omitted altogether, including physician fees and the costs of re‐admissions to public hospitals not reimbursed by Medibank Private. The minimum private health insurance benefit for hospital accommodation is $420 per bed‐day for surgical patients, or $453 per bed‐day for advanced surgery (for the first 14 days).26 Other costs not captured by our methodology are thus likely to at least double our calculated cost of each additional day in hospital attributed to complications, strengthening the association between DAH30 and total cost of care. Hospital costs are front‐weighted (ie, are highest on the day of surgery) because of operating theatre costs, complexity of surgery, and need for intensive care, and will consequently vary widely.
In Australia, 44% of people have private hospital insurance, and insurance status varies with age, residential location, and socio‐economic status.27 As private health insurance datasets do not capture re‐admissions to public hospitals (unless the patient makes a claim on their private health insurance), re‐admissions may be under‐reported, leading to overestimation of DAH30.
Conclusion
DAH30 is a validated, patient‐centred outcome measure of post‐surgical outcomes; higher values reflect shorter hospital stays and fewer serious complications, re‐admissions, and deaths. DAH30 is a practical proxy measure of cost‐effective quality of care and unexplained variation, supporting its utility as an indicator of the quality of surgical care in Australia.
Open access
Open access publishing facilitated by Monash University, as part of the Wiley – Monash University agreement via the Council of Australian University Librarians.
Box 1 – Patient‐related and peri‐operative characteristics for 181 281 people who underwent inpatient surgery with anaesthesia in Australia as private patients, 1 January 2016 – 31 December 2017
|
Characteristic |
Complete dataset |
Incomplete dataset* |
|||||||||||||
|
|
|||||||||||||||
|
Number of patients |
126 788 |
54 493 |
|||||||||||||
|
Age (years), median (IQR) |
62 (47–73) |
62 (42–73) |
|||||||||||||
|
Sex |
|
|
|||||||||||||
|
Men |
53 916 (43%) |
22 616 (42%) |
|||||||||||||
|
Women |
72 872 (57%) |
31 877 (58%) |
|||||||||||||
|
State/territory |
|
|
|||||||||||||
|
Victoria |
37 784 (30%) |
18 888 (35%) |
|||||||||||||
|
Queensland |
34 210 (27%) |
13 595 (25%) |
|||||||||||||
|
New South Wales |
26 122 (21%) |
12 454 (23%) |
|||||||||||||
|
South Australia/Northern Territory |
8314 (6.6%) |
3098 (5.7%) |
|||||||||||||
|
Western Australia |
14 273 (11%) |
4196 (7.7%) |
|||||||||||||
|
Tasmania |
3538 (2.8%) |
1341 (2.5%) |
|||||||||||||
|
Australian Capital Territory |
2547 (2.0%) |
921 (1.7%) |
|||||||||||||
|
Admission type |
|
|
|||||||||||||
|
Elective |
115 117 (91%) |
36 880 (85%) |
|||||||||||||
|
Emergency |
11 671 (9.2%) |
6488 (15%) |
|||||||||||||
|
Unknown |
0 |
11 125 |
|||||||||||||
|
Hospital type |
|
|
|||||||||||||
|
Private |
125 899 (99%) |
48 706 (94%) |
|||||||||||||
|
Public |
889 (0.7%) |
3088 (6.0%) |
|||||||||||||
|
Unknown |
0 |
2699 |
|||||||||||||
|
DAH30 (days), median (IQR) |
27.1 (24.2–28.8) |
26.9 (24.4–28.7) |
|||||||||||||
|
Charlson Comorbidity Index score (part A) |
|
|
|||||||||||||
|
0 |
98 933 (78%) |
43 627 (80%) |
|||||||||||||
|
1 |
17 714 (14%) |
6669 (12%) |
|||||||||||||
|
2 |
7525 (5.9%) |
2938 (5.4%) |
|||||||||||||
|
3 or more |
2616 (2.1%) |
1259 (2.3%) |
|||||||||||||
|
ASA physical status |
|
|
|||||||||||||
|
1 |
27 118 (21%) |
3568 (27%) |
|||||||||||||
|
2 |
54 119 (43%) |
4851 (37%) |
|||||||||||||
|
3 |
39 632 (31%) |
4090 (31%) |
|||||||||||||
|
4 or more |
5919 (4.7%) |
723 (5.5%) |
|||||||||||||
|
Unknown |
0 |
41 261 |
|||||||||||||
|
Surgical severity |
|
|
|||||||||||||
|
Complex major |
29 195 (23%) |
13 760 (28%) |
|||||||||||||
|
Major |
59 874 (47%) |
21 529 (44%) |
|||||||||||||
|
Intermediate |
36 508 (29%) |
13 307 (27%) |
|||||||||||||
|
Minor |
1211 (1.0%) |
417 (0.9%) |
|||||||||||||
|
Unknown |
0 |
5480 |
|||||||||||||
|
High dependency or intensive care unit admission |
|
|
|||||||||||||
|
No |
122 120 (96%) |
52 328 (96%) |
|||||||||||||
|
Yes |
4668 (3.7%) |
2165 (4.0%) |
|||||||||||||
|
Mechanical ventilation |
|
|
|||||||||||||
|
No |
126 388 (100%) |
54 345 (100%) |
|||||||||||||
|
Yes |
400 (0.3%) |
143 (0.3%) |
|||||||||||||
|
Unknown |
0 |
5 |
|||||||||||||
|
Unplanned theatre event |
2573 (2.0%) |
913 (1.7%) |
|||||||||||||
|
Unknown |
0 |
5 |
|||||||||||||
|
Operating time (minutes) |
|
|
|||||||||||||
|
< 30 |
11 498 (9.1%) |
10 622 (19%) |
|||||||||||||
|
30–60 |
23 698 (19%) |
13 387 (25%) |
|||||||||||||
|
60–120 |
50 948 (40%) |
19 536 (36%) |
|||||||||||||
|
> 120 |
40 644 (32%) |
10 948 (20%) |
|||||||||||||
|
Hospital cost, median (IQR) |
$10 358 ($6624–20 174) |
10 059 ($6147–15 511) |
|||||||||||||
|
|
|||||||||||||||
|
* ASA physical status data not available. ASA = American Society of Anesthesiologists; DAH30 = number of days alive and at home during 30 days after surgery; IQR = interquartile range. |
|||||||||||||||
Box 2 – Distribution of numbers of days alive and at home during the 30 days following surgery (DAH30) for 181 281 people who underwent inpatient surgery with anaesthesia in Australia as private patients, 1 January 2016 – 31 December 2017
Box 3 – Estimated median days alive and at home during the 30 days following surgery (DAH30): univariate (unadjusted) and multivariate (adjusted) quantile regression analysis
|
|
Estimated difference (days) (95% CI) |
||||||||||||||
|
Variable/term |
Univariate analysis |
Multivariate analysis* |
|||||||||||||
|
|
|||||||||||||||
|
Intercept |
— |
28.46 (28.36–28.55) |
|||||||||||||
|
Patient age (continuous) per year (v 59 years) |
–0.045 (–0.046 to –0.044) |
–0.023 (–0.024 to –0.022) |
|||||||||||||
|
ASA physical status (v 1) |
|
|
|||||||||||||
|
2 |
–0.65 (–0.72 to –0.58) |
0.002 (–0.02 to 0.03) |
|||||||||||||
|
3 |
–1.86 (–1.95 to –1.77) |
–0.47 (–0.52 to –0.42) |
|||||||||||||
|
4 or more |
–7.16 (–7.46 to –6.86) |
–1.93 (–2.16 to –1.70) |
|||||||||||||
|
Sex (women) |
–0.76 (–0.82 to –0.69) |
–0.44 (–0.46 to –0.41) |
|||||||||||||
|
State (v Victoria) |
|
|
|||||||||||||
|
Australian Capital Territory |
0.41 (0.05 to 0.76) |
0.22 (0.13 to 0.30) |
|||||||||||||
|
New South Wales |
0.05 (0.01 to 0.08) |
0.06 (0.02 to 0.10) |
|||||||||||||
|
Queensland |
0.24 (0.17 to 0.32) |
0.268 (0.23 to 0.29) |
|||||||||||||
|
South Australia/Northern Territory |
0.12 (0.07 to 0.18) |
0.12 (0.06 to 0.17) |
|||||||||||||
|
Tasmania |
–0.05 (–0.10 to –0.01) |
0.01 (–0.05 to 0.08) |
|||||||||||||
|
Western Australia |
0.89 (0.86 to 0.92) |
0.22 (0.19 to 0.25) |
|||||||||||||
|
Emergency admission |
–2.83 (–3.02 to –2.65) |
–2.19 (–2.32 to –2.06) |
|||||||||||||
|
Public hospital |
–1.28 (–1.67 to –0.88) |
–0.16 (–0.52 to 0.19) |
|||||||||||||
|
Charlson comorbidity score (part A) (v 0) |
|
|
|||||||||||||
|
1 |
–0.58 (–0.64 to –0.52) |
–0.14 (–0.18 to –0.10) |
|||||||||||||
|
2 |
–1.39 (–1.48 to –1.29) |
–0.14 (–0.23 to –0.05) |
|||||||||||||
|
3 or more |
–6.33 (–7.16 to –5.49) |
–2.81 (–3.25 to –2.36) |
|||||||||||||
|
Surgical severity (v minor) |
|
|
|||||||||||||
|
Intermediate |
0.750 (0.68 to 0.82) |
0.18 (0.10 to 0.25) |
|||||||||||||
|
Major |
–1.24 (–1.33 to –1.15) |
–1.07 (–1.15 to –0.99) |
|||||||||||||
|
Complex major |
–2.17 (–2.26 to –2.07) |
–1.10 (–1.19 to –1.02) |
|||||||||||||
|
High dependency or intensive care unit admission |
–9.85 (–10.19 to –9.51) |
–6.79 (–7.10 to –6.48) |
|||||||||||||
|
Mechanical ventilation |
–27.1 (–27.1 to –27.0) |
–14.5 (–14.8 to –14.1) |
|||||||||||||
|
Unplanned theatre event |
–0.88 (–1.20 to –0.55) |
–0.63 (–0.82 to –0.44) |
|||||||||||||
|
Operating time (minutes) (v < 30) |
|
|
|||||||||||||
|
30–60 |
0.94 (0.83 to 1.04) |
0.27 (0.22 to 0.32) |
|||||||||||||
|
60–120 |
0.61 (0.50 to 0.71) |
0.005 (–0.04 to 0.05) |
|||||||||||||
|
> 120 |
–1.33 (–1.43 to –1.22) |
–1.00 (–1.06 to –0.94) |
|||||||||||||
|
|
|||||||||||||||
|
* Adjusted for all other variables in table. ASA = American Society of Anesthesiologists; CI = confidence interval. |
|||||||||||||||
Received 5 November 2021, accepted 4 April 2022
- Jennifer R Reilly1,2
- Paul S Myles1,2
- Darren Wong3
- Stephane R Heritier4,5
- Wendy A Brown1,2
- Toby Richards6
- Max Bell7
- 1 Alfred Hospital, Melbourne, VIC
- 2 Monash University, Melbourne, VIC
- 3 Austin Health, Melbourne, VIC
- 4 Royal Prince Alfred Hospital, Sydney, NSW
- 5 The George Institute for International Health, Sydney, NSW
- 6 University of Western Australia, Perth, WA
- 7 Karolinska Institutet, Stockholm, Sweden
We thank David Rankin (clinical director/governance, research, innovation and product healthcare and strategy) and Alex Hickey (strategy analyst/healthcare and strategy) at Medibank Private Limited for their interest and providing the data for this study. The investigation was funded by Monash University. Paul Myles is supported by an National Health and Medical Research Council Practitioner Fellowship (1135937).
No relevant disclosures.
- 1. Porter ME, Lee TH. From volume to value in health care: the work begins. JAMA 2016; 316: 1047‐1048.
- 2. Pryor D, Hendrich A, Henkel RJ, et al. The quality “journey” at Ascension Health: how we’ve prevented at least 1500 avoidable deaths a year: and aim to do even better. Health Aff (Millwood) 2011; 30: 604‐611.
- 3. Crawford B, Skeath M, Whippy A. Multifocal clinical performance improvement across 21 hospitals. J Healthc Qual 2015; 37: 117‐125.
- 4. Staiger RD, Cimino M, Javed A, et al. The Comprehensive Complication Index (CCI®) is a novel cost assessment tool for surgical procedures. Ann Surg 2018; 268: 784‐791.
- 5. Birkmeyer JD, Gust C, Dimick JB, et al. Hospital quality and the cost of inpatient surgery in the United States. Ann Surg 2012; 255: 1‐5.
- 6. Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg 2017; 152: 292‐298.
- 7. Merkow RP, Ju MH, Chung JW, et al. Underlying reasons associated with hospital readmission following surgery in the United States. JAMA 2015; 313: 483‐495.
- 8. Graham LA, Mull HJ, Wagner TH, et al. Comparison of a potential hospital quality metric with existing metrics for surgical quality‐associated readmission. JAMA Netw Open 2019; 2: e191313.
- 9. Bell M, Eriksson LI, Svensson T, et al. Days at home after surgery: an integrated outcome measure for clinical trials and quality assurance. EClinicalMedicine 2019; 11: 18‐26.
- 10. Rotter T, Kinsman L, James E, et al. Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev 2010; CD006632.
- 11. Barnett ML, Grabowski DC, Mehrotra A. Home‐to‐home time: measuring what matters to patients and payers. N Engl J Med 2017; 377: 4‐6.
- 12. Myles PS, Shulman M, Heritier S, et al. Validation of days at home as an outcome measure after surgery: a prospective cohort study in Australia. BMJ Open 2017; 7: e015828.
- 13. Jerath A, Austin PC, Wijeysundera DN. Days alive and out of hospital: validation of a patient‐centered outcome for perioperative medicine. Anesthesiology 2019; 131: 84‐93.
- 14. Benchimol EI, Smeeth L, Guttmann A, et al; RECORD Working Committee. The REporting of studies Conducted using Observational Routinely‐collected health Data (RECORD) Statement. PLoS Med 2015; 12: e1001885.
- 15. Royal Australasian College of Surgeons; Medibank. Surgical variance report 2017. General surgery. https://www.surgeons.org/‐/media/Project/RACS/surgeons‐org/files/reports‐guidelines‐publications/surgical‐variance‐reports/surgical‐variance‐report‐2017‐general‐surgery.pdf?rev=3ae25da22f90449ab4b8700f9c4a5ca6&hash=505B9DC205B7C5514F66C6284412862D (viewed Mar 2022).
- 16. Department of Health. Medicare Benefits Schedule Review Taskforce: interim report to the Minister for Health. 4 Oct 2016. https://apo.org.au/node/69738 (viewed Mar 2022).
- 17. Australian Institute of Health and Welfare. Australia’s hospitals at a glance 2016–17 (Cat. no. HSE 204). 27 June 2018. https://www.aihw.gov.au/reports/hospitals/ahs‐2016‐17‐at‐a‐glance/formats (viewed Mar 2022).
- 18. Koenker R, Bassett G. Regression quantiles. Econometrica 1978; 46: 33‐50.
- 19. Charlson ME, Pompei P, Ales KL, Mackenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40: 373‐383.
- 20. Campbell D, Boyle L, Soakell‐Ho M, et al. National risk prediction model for perioperative mortality in non‐cardiac surgery. Br J Surg 2019; 106: 1549‐1557.
- 21. Han P, Kong L, Zhao J, Zhou Z. A general framework for quantile estimation with incomplete data. J R Stat Soc Series B Stat Methodol 2019; 81: 305‐333.
- 22. Zhang S, Han P. MultiRobust: multiply robust methods for missing data problems; version 1.0.5. June 2019. https://cran.r‐project.org/package=MultiRobust (viewed Mar 2022).
- 23. Marrie RA, Dawson NV, Garland A. Quantile regression and restricted cubic splines are useful for exploring relationships between continuous variables. J Clin Epidemiol 2009; 62: 511‐517.
- 24. Australian Institute of Health and Welfare. Australia's hospitals at a glance. Updated 23 June 2022. https://www.aihw.gov.au/reports/hospitals/australias‐hospitals‐at‐a‐glance/contents/summary (viewed June 2022).
- 25. Myles PS, Shulman M, Heritier S, et al. Validation of days at home as an outcome measure after surgery: a prospective cohort study in Australia. BMJ Open 2017; 7 :e015828.
- 26. Australian Government. Private Health Insurance (Benefit Requirements) Rules 2011. Compilation No. 71. 1 July 2021. https://www.legislation.gov.au/Details/F2021C00777 (viewed Mar 2022).
- 27. Australian Institute of Health and Welfare. Private health insurance. 23 July 2020. https://www.aihw.gov.au/reports/australias‐health/private‐health‐insurance (viewed Mar 2022).






Summary
Objective: To assess the relationships of patient and surgical factors and hospital costs with the number of days alive and at home during the 30 days following surgery (DAH30).
Design: Retrospective cohort study; analysis of Medibank Private health insurance hospital claims data, Australia, 1 January 2016 – 31 December 2017.
Setting, participants: Admissions of adults (18 years or older) to hospitals for elective or emergency inpatient surgery with anaesthesia covered by private health insurance, Australia, 1 January 2016 – 31 December 2017.
Main outcome measures: Associations between DAH30 and total hospital costs, and between DAH30 and surgery risk factors.
Results: Complete data were available for 126 788 of 181 281 eligible patients (69.9%); their median age was 62 years (IQR, 47–73 years), 72 872 were women (57%), and 115 117 had undergone elective surgery (91%). The median DAH30 was 27.1 days (IQR, 24.2–28.8 days), the median hospital cost per patient was $10 358 (IQR, $6624–20 174). The association between DAH30 and total hospital costs was moderate (Spearman ρ = –0.60; P < 0.001). Median DAH30 declined with age, comorbidity score, ASA physical status score, and surgical severity and duration, and was also lower for women.
Conclusions: DAH30 is a validated, patient-centred outcome measure of post-surgical outcomes; higher values reflect shorter hospital stays and fewer serious complications, re-admissions, and deaths. DAH30 can be used to benchmark quality of surgical care and to monitor quality improvement programs for reducing the costs of surgical and other peri-operative care.