Following the recommendation for coronavirus disease 2019 (COVID‐19) vaccination in all children and adolescents aged 12 years and older, the Australian Technical Advisory Group on Immunisation (ATAGI) and the New Zealand Ministry of Health have extended the group to include all children from 5 years of age. This includes children and adolescents with immunocompromising conditions such as cancer, haematopoietic stem cell transplant (HSCT) recipients, certain non‐malignant haematological conditions, and survivors of childhood cancer.1,2,3
Initial reports of severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) infection in children and adolescents with cancer were suggestive of mild COVID‐19,4,5,6 similar to children without significant comorbidities, where COVID‐19‐related mortality is estimated between 0.001% and 0.01%, rising to 0.7% in children with pre‐existing medical conditions.7,8 A large international cohort study subsequently reported severe COVID‐19 in 19.9% of children with SARS‐CoV‐2 undergoing treatment for cancer or following HSCT, with a 3.8% mortality rate.9 Notably, a large proportion of reported cases were from low income and lower middle income countries, and reporting bias may have occurred as cases were self‐reported. Nonetheless, considered alongside adult data demonstrating a more than threefold increased risk of severe COVID‐19 disease in immunocompromised people,10 the study suggests children receiving treatment for cancer or following HSCT are at increased risk of severe or fatal COVID‐19.
There remains a paucity of data regarding the immune response to COVID‐19 vaccine in children and adolescents receiving immunosuppressive therapy. In severely immunocompromised adults, COVID‐19 vaccine immune responses are less pronounced and likely result in reduced effectiveness compared with the broader population.11,12 Considering the safety profile of mRNA COVID‐19 vaccines and the potential to offer significant, albeit potentially reduced, protection against severe or fatal COVID‐19 in children and adolescents undergoing immunosuppressive treatment for cancer and non‐malignant haematological conditions, the Australian and New Zealand Children’s Haematology/Oncology Group (ANZCHOG) recommends vaccination in this vulnerable population. A summary of the key messages is provided in Box 1.
Methods
The ANZCHOG COVID‐19 Clinical and Research Reference Group was established to provide COVID‐19‐related recommendations, including vaccination guidelines, based on review of publications and current international and national health advisory committee guidelines, and where evidence is lacking, to provide consensus recommendations. The following disciplines from different states and hospitals across Australia and New Zealand are represented: medical oncology, transplant and cellular therapy, haematology, infectious diseases, microbiology, and immunology. Input was sought from the Australian and New Zealand Cardio‐Oncology Registry steering committee members.13
We reviewed the following health organisations and advisory group position statements in detail: ATAGI; NZ Ministry of Health COVID‐19 program; Canadian National Advisory Committee on Immunisation; Australia and New Zealand Transplant and Cellular Therapies; British Society of Blood and Marrow Transplantation and Cellular Therapy; European Society for Blood and Marrow Transplantation; Children’s Cancer and Leukaemia Group; Haematology Society of Australia and New Zealand; American Society of Hematology; Australasian Society of Clinical Immunology and Allergy; Cardiac Society of Australia and New Zealand; Centers for Disease Control and Prevention (CDC); European Medicines Agency; and United Kingdom Joint Committee on Vaccination and Immunisation.
Most data on COVID‐19 vaccination in populations without notable comorbidities was supported by high quality evidence according to the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) criteria,14 and included large international randomised controlled trials. Data on COVID‐19 vaccination in people who are immunocompromised are sparse, based predominantly on observational studies of adults, and of moderate to poor quality. Some COVID‐19 vaccination adverse effects data are supported by large observational cohort studies, surveillance programs and case studies (eg, myocarditis and pericarditis), whereas others have only been assessed in small cohort studies (eg, polyethylene glycol–asparaginase allergy) and for some, evidence is yet to be obtained (eg, COVID‐19 vaccination following anthracyclines).
Recommendations
Cohort of patients
Recommendation
All children and adolescents in Australia and New Zealand with cancer and non‐malignant haematological conditions, and all survivors of childhood cancer aged ≥ 5 years should receive at least two doses of an mRNA COVID‐19 vaccine.
Children and adolescents with the following conditions are considered to be at the greatest risk of severe COVID‐19 due to a high degree of immunosuppression, and should receive an augmented three‐dose primary schedule (Box 2):
- ▪ any type of cancer including haematological, solid or brain cancer undergoing active treatment;
- ▪ recipients of autologous or allogeneic HSCT or chimeric antigen receptor (CAR) T‐cell therapy within 2 years of treatment;
- ▪ those receiving immunosuppressive therapies including:
- ► high dose corticosteroids;
- ► biological and targeted therapies including anti‐CD20 antibodies, anti‐CD52 antibodies, anti‐thymocyte globulin, Janus kinase inhibitors and anti‐complement antibodies;
- ► conventional synthetic disease‐modifying anti‐rheumatic drugs including mycophenolate mofetil, methotrexate, cyclosporine, tacrolimus and cyclophosphamide;
- ▪ those receiving multiple immunosuppressants with an expected cumulative effect.
Comments
Children and adolescents with confirmed COVID‐19 who have not yet received vaccination should proceed with vaccination as soon as they recover to boost their natural, albeit likely reduced, immune response.
This is in accordance with ATAGI and NZ Ministry of Health statements addressing COVID‐19 vaccination in children aged 5–11 years1,3 and 12–15 years,2,3 and severely immunocompromised people,15 and is supported by recommendations set by the CDC and European Medicines Agency.16,17
Type of vaccine
Recommendation
The recommended vaccine is Comirnaty (Pfizer), available in both Australia and New Zealand, or alternatively, Spikevax (Moderna), available for use in Australian adolescents ≥ 12 years of age.
Comments
Currently, Comirnaty is the only COVID‐19 vaccine approved for use in children and adolescents aged ≥ 5 years in both Australia and New Zealand.18 Spikevax has been approved, in addition to Comirnaty, in the ≥ 12 years age group in Australia only.19 Vaxzevria (AstraZeneca), Ad26.COV2.S (Janssen) and Nuvaxovid (Novavax) are registered in both Australia and New Zealand for use in people aged ≥ 18 years, with either Comirnaty or Spikevax being preferred in younger adults.20,21
The safety of both Comirnaty and Spikevax vaccines is supported by a number of large clinical trials conducted in people aged ≥ 16 years, in addition to real world data on hundreds of millions of people vaccinated worldwide to date.22 The two largest placebo‐controlled trials conducted on 43 548 and 30 420 participants studying safety and efficacy of Comirnaty and Spikevax demonstrated 95% and 94% efficacy in preventing COVID‐19, respectively, and an excellent safety profile for both.23,24 The safety of these vaccines is also supported by local post‐marketing safety surveillance data.25,26
Both vaccines are mRNA‐based lipid nanoparticle‐encapsulated vaccines that encode the full‐length spike glycoprotein of SARS‐CoV‐2. They are non‐live vaccines and therefore safe for use in immunocompromised people.16
Safety, immunogenicity and efficacy of both Comirnaty and Spikevax were studied in placebo‐controlled trials of 2260 and 3732 adolescents aged 12–15 and 12–17 years, respectively. Favourable vaccine safety profiles were demonstrated, with mainly transient mild to moderate reactions, the most common after the first and second doses being injection site pain (in 86% and 79% of participants after Comirnaty, and 93% and 92% after Spikevax), fatigue (in 60% and 66% after Comirnaty, and 48% and 68% after Spikevax), and headache (in 55% and 65% after Comirnaty, and 45% and 70% after Spikevax). No vaccine‐related serious adverse events were observed in either study. Both Comirnaty and Spikevax were efficacious, with no breakthrough infections in vaccine recipients. For Comirnaty, immunogenicity was greater than previously observed in young adults aged 16–25 years, and for Spikevax, immunogenicity was similar to that observed in young adults.27,28
Safety, immunogenicity and efficacy of COVID‐19 vaccine in children aged 6 months to 11 years are being studied in an ongoing placebo‐controlled trial. Results for the 5–11 years group have recently been published, showing efficacy of 90.7% after two paediatric doses of Comirnaty, with a similar safety profile to that observed in older children and adolescents among 2186 study participants.29
An association between COVID‐19 vaccination and myocarditis or pericarditis had been reported30 but not observed in the initial Comirnaty and Spikevax clinical trials in adults, or in the corresponding adolescent trials, likely due to inadequate participant numbers to detect such rare events.23,24 Since licensure, a number of cases of myocarditis or pericarditis linked to mRNA COVID‐19 vaccines have been reported in the literature, and investigated by national vaccine safety programs.16,31,32,33 Symptoms typically appear 1–5 days after vaccination and are more frequent after the second dose. The condition seems to occur more frequently in male adolescents and young adults at an estimated incidence of 1 to 5 per 100 000 individuals,34,35,36 and appears to be extremely rare in the 5–11 years age group.37 Most cases have required hospitalisation; however, the majority responded well to standard treatment with a mild and self‐limiting course and no associated deaths.32 Notably, in comparison, SARS‐CoV‐2 infection‐associated myocarditis occurs at an incidence of 11 per 100 000 individuals.36
Vaccination schedule
Recommendation
COVID‐19 vaccination schedule should vary depending on a child’s age, the level of immunosuppression, and vaccine availability (Box 2).
Comments
Increasing evidence suggests reduced vaccine response in immunocompromised patients compared with the broader population. Lower post‐vaccination antibody geometric mean titres have been found in recipients of solid organ transplants and adult patients with haematological malignancies.38,39,40,41 A study identified reduced cellular immunity in patients immunosuppressed after solid organ transplants;42 however, the majority of studies did not evaluate T‐cell vaccine responses. Although immunogenicity data cannot directly predict protection from infection, observational studies finding a high proportion of immunocompromised individuals among fully vaccinated hospitalised patients with severe COVID‐19 support the hypothesis.43,44
Based on the available data, ATAGI15 and the NZ Ministry of Health45 have recently recommended a three‐dose primary schedule of either Comirnaty or Spikevax in Australia, or Comirnaty in NZ, in severely immunocompromised people. This is in keeping with previously released recommendations by the CDC and the UK Joint Committee on Vaccination and Immunisation.16,46
An extended dosing interval in adult populations has resulted in improved vaccine effectiveness and potentially a longer duration of protection compared with the standard interval.47 Based on this, ATAGI and the NZ Ministry of Health recommend an 8‐week interval between doses in non‐immunocompromised and moderately immunocompromised children aged 5–11 years where optimisation of immune response can be prioritised over a need for rapidly gained protection against COVID‐19.1,48
Vaccine contraindications and precautions
Recommendation
Children and adolescents who have developed an anaphylactic reaction to an mRNA COVID‐19 vaccine should not receive further mRNA vaccines.
Comments
Anaphylaxis to mRNA vaccination is a rare event, reported at a rate of 4.7 cases per million doses.49
Generalised allergic reactions (without anaphylaxis) are not necessarily a contraindication to subsequent vaccination but should involve consultation with an allergy/immunology specialist for further management.50
A history of reaction to previous non‐COVID‐19 vaccinations is not a contraindication for receipt of an mRNA COVID‐19 vaccine.50
Recommendation
Children and adolescents who have previously developed an allergic reaction (including anaphylaxis) to polyethylene glycol (PEG)–asparaginase, except for those with a documented anaphylaxis to PEG, may still receive an mRNA vaccine, although this should take place in a hospital setting. In the absence of hospital setting vaccination, children and adolescents in New Zealand with a history of PEG–asparaginase allergy (including anaphylaxis) may receive Comirnaty in large medically attended vaccination centres within a close proximity to hospital, with increased post vaccination observation time.51
Comments
Both Comirnaty and Spikevax contain PEG, an excipient used in a number of drugs to improve water solubility. The vast majority of children who develop anaphylaxis following PEG–asparaginase are likely to be allergic to the asparaginase and not PEG, although this is difficult to elucidate and justifies caution when administering the vaccine.52 Furthermore, the type of PEG used in COVID‐19 vaccines differs from those commonly used in other medicines and the risk of cross‐reactivity appears to be low. In a cohort study of children and young adults with a history of PEG–asparaginase allergy, none of the 32 participants developed an allergic reaction to the Comirnaty vaccine.53 Evidence‐based algorithms have been developed internationally, and have been endorsed by the Australasian Society of Clinical Immunology and Allergy.50
Recommendation
Children and adolescents with myocarditis attributed to a previous dose of an mRNA COVID‐19 vaccine, and those with pericarditis associated with a previous mRNA COVID‐19 vaccine and abnormal investigations, should not receive further mRNA vaccines.
Comments
This recommendation is in keeping with Cardiac Society of Australia and New Zealand guidance regarding mRNA COVID‐19 vaccination‐associated myocarditis and pericarditis in individuals aged ≥ 12 years.54
Recommendation
According to the Cardiac Society of Australia and New Zealand guideline:54
- ▪ Children and adolescents who have developed pericarditis possibly associated with a previous dose of an mRNA COVID‐19 vaccine but with normal investigation results (electrocardiography, troponin and inflammatory markers), and who have been symptom‐free for at least 6 weeks, may receive further doses of an mRNA COVID‐19 vaccine.
- ▪ Children and adolescents with current or recent (within the past 3 months) myocarditis or pericarditis due to causes other than the mRNA vaccination, including rheumatic heart disease and acute rheumatic fever, can receive an mRNA COVID‐19 vaccine after consultation with a general practitioner, immunisation specialist or cardiologist.
Comments
ANZCHOG recommends that all children and adolescents with cancer and non‐malignant haematological conditions with myocarditis or pericarditis considered for COVID‐19 vaccination should receive specialist consultation.
Recommendation
Children and adolescents who have received anthracyclines as part of their treatment are not considered at higher risk of developing side effects from mRNA COVID‐19 vaccination, and should therefore receive either of the mRNA vaccines.
Comments
Survivors of childhood cancer treated with anthracyclines, such as doxorubicin, daunorubicin, idarubicin and mitoxantrone, are at an increased risk of developing cardiomyopathy and congestive heart failure.55,56
Currently, there is no evidence to suggest long term sequelae in people who have developed mRNA vaccine‐related myocarditis or pericarditis. Similarly, there is no evidence to suggest poor cardiac outcomes in patients who have received anthracyclines and mRNA vaccination.
Further support for mRNA COVID‐19 vaccination in children and adolescents with cancer who have been exposed to anthracyclines can be garnered from Cardiac Society of Australia and New Zealand guidance recommending mRNA COVID‐19 vaccination without specific precautions in individuals aged ≥ 12 years with most pre‐existing cardiac conditions, including dilated cardiomyopathy and stable heart failure.54
Vaccine timing
Recommendation
mRNA COVID‐19 vaccines can be given during treatment, providing that scheduling of the vaccine has been discussed with the treating haematology/oncology team.
Comments
It has been shown that some drugs, such as the antimetabolites (ie, methotrexate, mycophenolate mofetil) and corticosteroids, have a negative impact on humoral vaccine response in adult patients.38,57,58 In a study of Comirnaty and Vaxzevria in adult patients with multiple myeloma, patients on treatment showed a significantly lower vaccine response compared with those off treatment;59 and a blunted antibody response was found after two doses of Comirnaty in another study of adults with haematological malignancies.60 Immunogenicity was significantly higher in patients with solid tumours compared with haematological malignancies in a study of Comirnaty in adult patients undergoing treatment for cancer.61
Despite emerging evidence of a poorer vaccine response in the setting of immunosuppressive therapy, there are currently no guidelines regarding ideal timing of vaccination during treatment for cancer and other conditions requiring immunosuppressive therapy. Given the demonstrated safety of vaccination and the higher risk of severe disease in the setting of immunosuppression, vaccination should proceed during cancer therapy as soon as practicable.
Recommendation
Children and adolescents who have received anthracyclines, high dose cytarabine (> 6000 mg/m2) or immune checkpoint inhibitors should receive mRNA COVID‐19 vaccination at the following time points: at least 1 week after anthracycline administration; at least 4 weeks after high dose cytarabine administration; and at least 6 weeks after the first dose of immune checkpoint inhibitors, and only if they did not develop myocarditis or pericarditis.
Comments
Anthracyclines, high dose cytarabine and immune checkpoint inhibitors have all very rarely been associated with acute pericarditis.62,63,64,65,66,67 Acute pericarditis following anthracyclines occurs within the first week after administration.68 Conversely, high dose cytarabine has been shown to cause pericarditis up to 28 days after exposure, supporting a hypothesis of this adverse event being a type IV delayed hypersensitivity reaction.62 For immune checkpoint inhibitors, the median time for development of pericarditis is longer (usually 6–8 weeks) with wide inter‐patient variability.69,70,71,72
Recommendation
Children and adolescents who are being treated with rituximab, CD19‐targeted CAR T‐cells or HSCT should receive mRNA COVID‐19 vaccination before the treatment begins or, if the treatment has already been administered, at least 3 months after completion of the treatment.
Comments
Rituximab, a humanised chimeric anti‐CD20 monoclonal antibody, is widely used for the treatment of B‐cell lymphoproliferative disease and refractory auto‐immune conditions. The profound effect on B‐cell depletion following rituximab has been found to have a significantly negative effect on immune response to other vaccines, and it is therefore expected that the mRNA COVID‐19 vaccine response would be reduced in a similar manner.73
CAR T‐cells, currently licensed in Australia for the treatment of refractory, relapsed post‐HSCT, or second or greater relapse CD19‐positive B‐cell acute lymphoblastic leukaemia in children and young adults ≤ 25 years of age,74 induce a long‐lasting and profound hypogammaglobinaemia, which is likely to affect the serological response to mRNA COVID‐19 vaccines.75,76
Patients who have received HSCT experience a state of profound immune dysfunction for weeks to months, with a gradual recovery to full immune function about 2 years after HSCT.77 Current international guidelines for vaccination of HSCT recipients recommend administration of inactivated vaccines from 6 months following HSCT, and from 3 months in community outbreaks.78
Although it is acknowledged that optimal response to mRNA COVID‐19 vaccines is more likely to develop ≥ 6 months after HSCT, introducing vaccination as early as 3 months after HSCT in the context of the COVID‐19 pandemic has been endorsed by the British Society of Blood and Marrow Transplantation and Cellular Therapy, European Society for Blood and Marrow Transplantation, and Australia and New Zealand Transplant and Cellular Therapies in their COVID‐19 vaccination consensus statements.79,80,81 A recent report revealed a good humoral response to COVID‐19 vaccination in allogeneic recipients < 12 months after HSCT; however, the report did not specify if a good response was observed in those who received COVID‐19 vaccination 3 months after HSCT.82
Infection prevention and control
Recommendation
COVID‐19 vaccination should not replace public health measures reducing the risk of SARS‐CoV‐2 infection.
Comments
Measures such as wearing masks, social distancing, good hand hygiene and optimal indoor ventilation should continue.83 These are of particular importance in children and adolescents undergoing treatment for cancer and those on immunosuppressive therapies, given the likelihood of reduced immunogenicity to COVID‐19 vaccination.
All household contacts, including parents, siblings aged ≥ 5 years, and grandparents with no contraindication to COVID‐19 vaccination, should receive COVID‐19 vaccination.
Conclusion
In this consensus statement, ANZCHOG provides current recommendations for COVID‐19 vaccination in children and adolescents aged ≥ 5 years with cancer and immunocompromising non‐malignant haematological conditions (Box 3).
The recommendations are based on evidence‐based knowledge of the safety, immunogenicity and efficacy of the vaccines in the general population, in addition to emerging data regarding COVID‐19 vaccination in immunocompromised individuals.
Large international trials and collection of real world data are essential to provide best evidence to identify the ideal timing, schedule, efficacy and safety of COVID‐19 vaccines in this vulnerable population.84
Endorsement
These recommendations have been endorsed by ANZCHOG, and are regularly updated by the ANZCHOG COVID‐19 Clinical and Research Reference Group. A lay summary, produced in consultation with consumers, can be found on the ANZCHOG organisational webpage: https://anzchog.org/covid‐19‐vaccination‐guidance‐for‐children‐5‐years‐and‐older‐undergoing‐cancer‐treatment‐and‐children‐with‐non‐cancerous‐blood‐disorders‐updated/.
Box 1 – Key messages
- ▪ The Australian Technical Advisory Group on Immunisation (ATAGI) and New Zealand Ministry of Health have extended their recommendation for COVID‐19 vaccination to include children aged 5 years and older.1,3
- ▪ Children and adolescents undergoing treatment for cancer and immunosuppressive therapy for non‐malignant haematological conditions are among the most vulnerable.
- ▪ Comirnaty (Pfizer) and Spikevax (Moderna) are mRNA‐based non‐live vaccines suitable for use in immunocompromised individuals. Both vaccines have an excellent safety profile and high efficacy in the general population, including children and adolescents.
- ▪ Immunogenicity of COVID‐19 vaccines in the immunocompromised is reduced,11,12 leading to recommendations for an augmented primary schedule in severely immunocompromised people.
- ▪ Considering the safety profile of mRNA COVID‐19 vaccines and the increased risk of severe COVID‐19, the Australian and New Zealand Children’s Haematology/Oncology Group strongly recommends COVID‐19 vaccination.
- ▪ All children and adolescents with cancer and non‐malignant haematological conditions, and all survivors of childhood cancer aged ≥ 5 years, except for those in whom vaccination is contraindicated, should receive COVID‐19 vaccination.
- ▪ All household contacts, including parents, siblings ≥ 5 years of age, and grandparents, should be encouraged to receive a COVID‐19 vaccine.
Box 2 – Primary vaccination schedule for children and adolescents, based on age and level of immunosuppression*
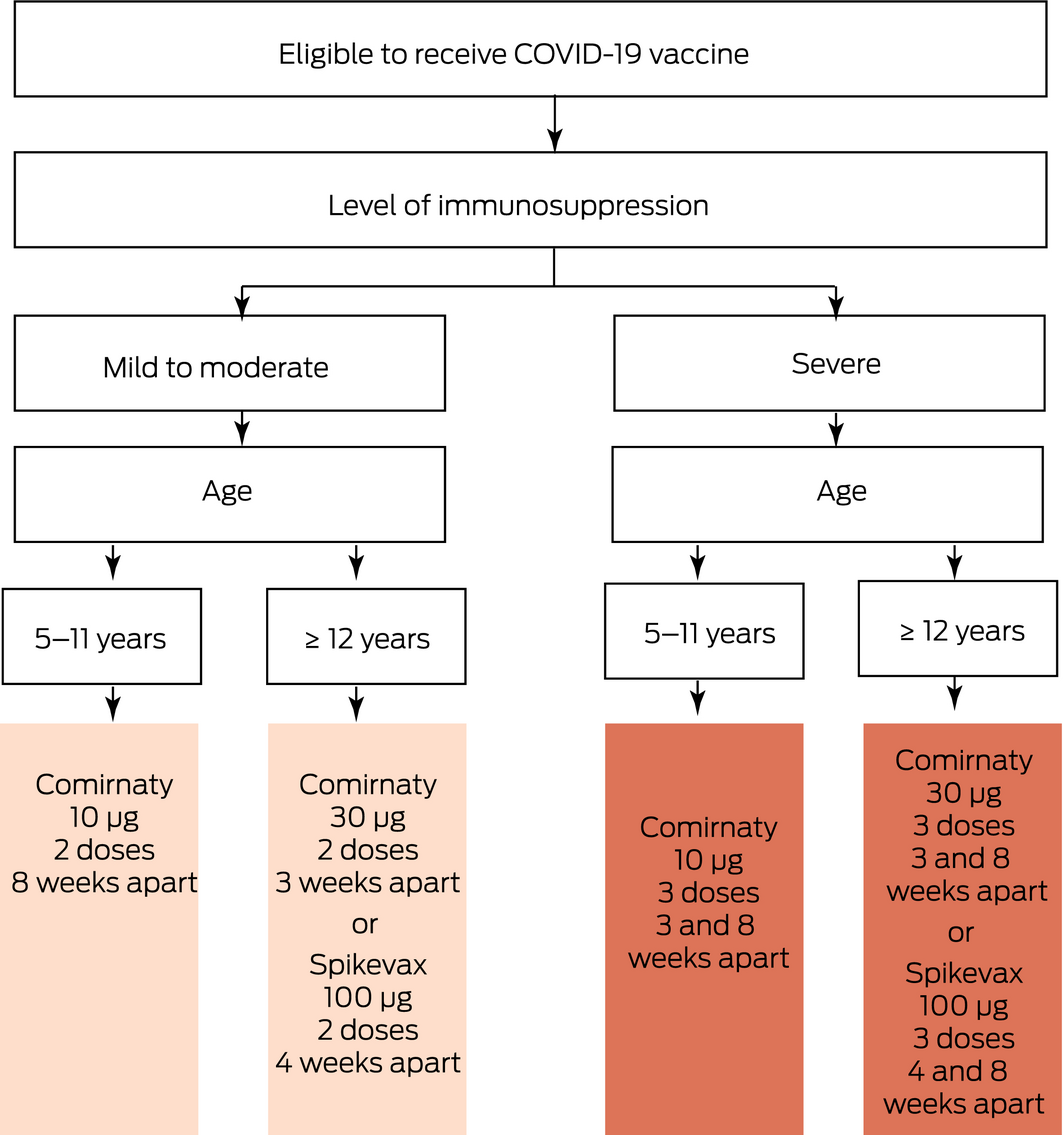
* Comirnaty (Pfizer) is available in Australia and New Zealand; Spikevax (Moderna) is available in Australia only. Note the primary vaccination schedule is regularly updated to reflect latest changes.
Box 3 – Key recommendations
- ▪ All children in Australia and New Zealand with cancer and non‐malignant haematological conditions, and all survivors of childhood cancer aged ≥ 5 years, should receive COVID‐19 vaccination.
- ▪ Children and adolescents with the following conditions are considered to be at the highest risk of severe COVID‐19, and should receive an augmented primary vaccine schedule:
- ► any type of cancer including haematological, solid or brain cancer undergoing active treatment;
- ► recipients of autologous or allogeneic haematopoietic stem cell transplantation (HSCT) or chimeric antigen receptor (CAR) T‐cell therapy within 2 years of treatment;
- ► those receiving immunosuppressive therapies including high dose corticosteroids, biological and targeted therapies such as rituximab, or conventional synthetic disease‐modifying anti‐rheumatic drugs;
- ► those receiving multiple immunosuppressants.
- ▪ Children and adolescents with confirmed COVID‐19 should proceed with vaccination after they have recovered from COVID‐19.
- ▪ Recommended COVID‐19 vaccines are:
- ► Comirnaty (Pfizer), available in both Australia and New Zealand; and
- ► Spikevax (Moderna), available in Australian adolescents ≥ 12 years old.
- ▪ COVID‐19 vaccination schedule varies depending on child’s age, level of immunosuppression, and vaccine availability.
- ▪ Vaccine contraindications:
- ► anaphylaxis to an mRNA COVID‐19 vaccine;
- ► anaphylaxis to polyethylene glycol (PEG), but not PEG–asparaginase;
- ► myocarditis attributed to an mRNA COVID‐19 vaccine;
- ► pericarditis associated with an mRNA COVID‐19 vaccine and abnormal investigations.
- ▪ Vaccine precautions:
- ► generalised allergic reaction other than anaphylaxis to an mRNA COVID‐19 vaccine;
- ► anaphylaxis to PEG‐asparaginase;
- ► current or recent (within the past 3 months) myocarditis or pericarditis due to causes other than an mRNA COVID‐19 vaccine;
- ► acute decompensated heart failure.
- ▪ Treatment with anthracyclines is not a contraindication to COVID‐19 vaccination.
- ▪ COVID‐19 vaccines can be administered during treatment as soon as practicable, with the exception of the following:
- ► at least 1 week after anthracycline administration;
- ► at least 4 weeks after high dose cytarabine administration;
- ► at least 6 weeks after the first dose of immune checkpoint inhibitors, and only if there has been no development of myocarditis or pericarditis;
- ► before the treatment, or at least 3 months after the completion of treatment in those receiving rituximab, CAR T‐cell therapy or HSCT.
- ▪ COVID‐19 vaccination should not replace public health measures reducing the risk of SARS‐CoV‐2 infection.
- ▪ All household contacts ≥ 5 years of age with no COVID‐19 vaccine contraindication should receive COVID‐19 vaccination.
Provenance: Not commissioned; externally peer reviewed.
- Eliska Furlong1,2
- Rishi S Kotecha1,2,3
- Rachel Conyers4,5,6
- Tracey A O'Brien7,8
- Jordan R Hansford4,5,6
- Leanne Super5,9
- Peter Downie9,10,11
- David D Eisenstat4,5,6
- Gabrielle Haeusler4,12,13
- Brendan McMullan8,13,14
- Marianne B Phillips1
- Bhavna Padhye15
- Luciano Dalla‐Pozza15
- Frank Alvaro16,17
- Christopher J Fraser18
- Wayne Nicholls18,19
- Julia E Clark18,19
- Matthew O'Connor20
- Benjamin R Saxon20
- Heather Tapp20
- John Heath21
- Sarah E Hunter22
- Karen Tsui22
- Mark Winstanley22
- Amanda Lyver23
- Emma J Best24,25,26
- Ushma Wadia1,27,28
- Daniel Yeoh1,13
- Christopher C Blyth1,28,29
- Nicholas G Gottardo1,2,29
- 1 Perth Children's Hospital, Perth, WA
- 2 Telethon Kids Cancer Centre, Perth, WA
- 3 Curtin University, Perth, WA
- 4 Murdoch Children’s Research Institute, Melbourne, VIC
- 5 Children’s Cancer Centre, Royal Children’s Hospital, Melbourne, VIC
- 6 University of Melbourne, Melbourne, VIC
- 7 Kids Cancer Centre, Sydney Children’s Hospital, Sydney, NSW
- 8 University of New South Wales, Sydney, NSW
- 9 Monash University, Melbourne, VIC
- 10 Monash Children’s Hospital, Melbourne, VIC
- 11 Hudson Institute of Medical Research, Melbourne, VIC
- 12 Paediatric Integrated Cancer Service, Melbourne, VIC
- 13 National Centre for Infections in Cancer, Melbourne, VIC
- 14 Sydney Children’s Hospital, Sydney, NSW
- 15 Cancer Centre for Children, The Children's Hospital at Westmead, Sydney, NSW
- 16 John Hunter Children’s Hospital, Newcastle, NSW
- 17 University of Newcastle, Newcastle, NSW
- 18 Queensland Children’s Hospital, Brisbane, QLD
- 19 University of Queensland, Brisbane, QLD
- 20 Michael Rice Centre for Haematology and Oncology, Women’s and Children’s Hospital, Adelaide, SA
- 21 Children’s and Adolescent/Young Adult Cancer Centre, Royal Hobart Hospital, Hobart, TAS
- 22 Blood and Cancer Centre, Starship Children’s Hospital, Auckland, NZ
- 23 Children’s Haematology Oncology Centre, Christchurch Hospital, Christchurch, NZ
- 24 Starship Children’s Hospital, Auckland, NZ
- 25 Immunisation Advisory Centre, University of Auckland, Auckland, NZ
- 26 University of Auckland, Auckland, NZ
- 27 Fiona Stanley Hospital, Perth, WA
- 28 Wesfarmers Centre of Vaccines and Infectious Diseases, Telethon Kids Institute, Perth, WA
- 29 University of Western Australia, Perth, WA
No relevant disclosures.
- 1. Australian Government Department of Health. ATAGI recommendations on Pfizer COVID‐19 vaccine use in children aged 5 to 11 years. 9 Dec 2021. https://www.health.gov.au/resources/publications/atagi‐recommendations‐on‐pfizer‐covid‐19‐vaccine‐use‐in‐children‐aged‐5‐to‐11‐years (viewed Feb 2022).
- 2. Australian Government Department of Health. ATAGI statement regarding vaccination of adolescents aged 12–15 years. 2 Aug 2021. https://www.health.gov.au/news/atagi‐statement‐regarding‐vaccination‐of‐adolescents‐aged‐12‐15‐years (viewed Feb 2022).
- 3. New Zealand Government. Unite Against COVID‐19. Vaccination for children and young people. https://covid19.govt.nz/covid‐19‐vaccines/get‐the‐facts‐about‐covid‐19‐vaccination/covid‐19‐vaccination‐and‐children/ (viewed Feb 2022).
- 4. Liguoro I, Pilotto C, Bonanni M, et al. SARS‐COV‐2 infection in children and newborns: a systematic review. Eur J Pediatr 2020; 179: 1029–1046.
- 5. Ibrahim LF, Tham D, Chong V, et al. The characteristics of SARS‐CoV‐2‐positive children who presented to Australian hospitals during 2020: a PREDICT network study. Med J Aust 2021; 215: 217–221. https://www.mja.com.au/journal/2021/215/5/characteristics‐sars‐cov‐2‐positive‐children‐who‐presented‐australian‐hospitals .
- 6. Millen GC, Arnold R, Cazier J‐B, et al. Severity of COVID‐19 in children with cancer: report from the United Kingdom Paediatric Coronavirus Cancer Monitoring Project. Br J Cancer 2021; 124: 754–759.
- 7. O’Driscoll M, Ribeiro Dos Santos G, Wang L, et al. Age‐specific mortality and immunity patterns of SARS‐CoV‐2. Nature 2021; 590: 140–145.
- 8. Götzinger F, Santiago‐García B, Noguera‐Julián A, et al. COVID‐19 in children and adolescents in Europe: a multinational, multicentre cohort study. Lancet Child Adolesc Health 2020; 4: 653–661.
- 9. Mukkada S, Bhakta N, Chantada GL, et al. Global characteristics and outcomes of SARS‐CoV‐2 infection in children and adolescents with cancer (GRCCC): a cohort study. Lancet Oncol 2021; 22: 1416–1426.
- 10. Gao Y, Chen Y, Liu M, Shi S, Tian J. Impacts of immunosuppression and immunodeficiency on COVID‐19: a systematic review and meta‐analysis. J Infect 2020; 81: e93–e95.
- 11. Chodick G, Tene L, Rotem RS, et al. The effectiveness of the two‐dose BNT162b2 vaccine: analysis of real‐world data. Clin Infect Dis 2022; 74: 472–478.
- 12. Dagan N, Barda N, Kepten E, et al. BNT162b2 mRNA Covid‐19 vaccine in a nationwide mass vaccination setting. N Engl J Med 2021; 384: 1412–1423.
- 13. Lapirow D, La Gerche A, Toro C, et al. The Australia and New Zealand Cardio‐Oncology Registry: evaluation of chemotherapy‐related cardiotoxicity in a national cohort of paediatric cancer patients. Intern Med J 2021; 51: 229–234.
- 14. Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008; 336: 924–926.
- 15. Australian Government Department of Health. ATAGI recommendations on the use of a third primary dose of COVID‐19 vaccine in individuals who are severely immunocompromised. Last updated: 11 Feb 2022. https://www.health.gov.au/resources/publications/atagi‐recommendations‐on‐the‐use‐of‐a‐third‐primary‐dose‐of‐covid‐19‐vaccine‐in‐individuals‐who‐are‐severely‐immunocompromised (viewed Feb 2022).
- 16. Centers for Disease Control and Prevention. COVID‐19. https://www.cdc.gov/coronavirus/2019‐ncov/index.html (viewed Feb 2022).
- 17. European Medicines Agency. COVID‐19 vaccines. https://www.ema.europa.eu/en/human‐regulatory/overview/public‐health‐threats/coronavirus‐disease‐covid‐19/treatments‐vaccines/covid‐19‐vaccines (viewed Feb 2022).
- 18. Therapeutic Goods Administration. Australian product information – Comirnaty (BNT162b2 [mRNA]) COVID‐19 vaccine. https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP‐2021‐PI‐01092‐1&d=20220216172310101 (viewed Feb 2022).
- 19. Therapeutic Goods Administration. Australian product information – Spikevax (elasomeran) COVID‐19 vaccine. https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP‐2021‐PI‐01968‐1 (viewed Feb 2022).
- 20. Australian Government Department of Health. ATAGI statement on revised recommendations on the use of COVID‐19 vaccine AstraZeneca, 17 June 2021. https://www.health.gov.au/news/atagi‐statement‐on‐revised‐recommendations‐on‐the‐use‐of‐covid‐19‐vaccine‐astrazeneca‐17‐june‐2021 (viewed Feb 2022).
- 21. New Zealand Ministry of Health. COVID‐19: AstraZeneca vaccines. https://www.health.govt.nz/our‐work/diseases‐and‐conditions/covid‐19‐novel‐coronavirus/covid‐19‐vaccines/covid‐19‐vaccine‐health‐advice/covid‐19‐getting‐other‐vaccines/covid‐19‐astrazeneca‐vaccines (viewed Feb 2022).
- 22. Centers for Disease Control and Prevention. Coronavirus disease 2019 (COVID‐19) vaccines. Advisory Committee on Immunization Practices (ACIP) presentation slides, 30 Aug 2021. https://www.cdc.gov/vaccines/acip/meetings/slides‐2021‐08‐30.html (viewed Feb 2022).
- 23. Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid‐19 vaccine. N Engl J Med 2020; 383: 2603–2615.
- 24. Baden LR, El Sahly HM, Essink B, et al. Efficacy and safety of the mRNA‐1273 SARS‐CoV‐2 vaccine. N Engl J Med 2021; 384: 403–416.
- 25. AusVaxSafety. COVID‐19 vaccine safety surveillance. https://ausvaxsafety.org.au/our‐work/covid‐19‐vaccine‐safety‐surveillance (viewed Feb 2022).
- 26. Therapeutic Goods Administration. COVID‐19 vaccine weekly safety monitoring and reporting. 16 Dec 2021. https://www.tga.gov.au/periodic/covid‐19‐vaccine‐weekly‐safety‐report‐16‐12‐2021 (viewed Feb 2022).
- 27. Frenck RW, Klein NP, Kitchin N, et al. Safety, Immunogenicity, and Efficacy of the BNT162b2 Covid‐19 Vaccine in Adolescents. N Engl J Med 2021; 385: 239–250.
- 28. Ali K, Berman G, Zhou H, et al. Evaluation of mRNA‐1273 SARS‐CoV‐2 vaccine in adolescents. N Engl J Med 2021; 385: 2241–2251.
- 29. Walter EB, Talaat KR, Sabharwal C, et al. Evaluation of the BNT162b2 Covid‐19 vaccine in children 5 to 11 years of age. N Engl J Med 2022; 386: 35–46.
- 30. Castiello T, Georgiopoulos G, Finocchiaro G, et al. COVID‐19 and myocarditis: a systematic review and overview of current challenges. Heart Fail Rev 2022; 27: 251–261.
- 31. Abu Mouch S, Roguin A, Hellou E, et al. Myocarditis following COVID‐19 mRNA vaccination. Vaccine 2021; 39: 3790–3793.
- 32. Montgomery J, Ryan M, Engler R, et al. Myocarditis following immunization with mRNA COVID‐19 vaccines in members of the US Military. JAMA Cardiol 2021; 6: 1202–1206.
- 33. Rosenblum H. Benefits and risks of Pfizer‐BioNTech COVID‐19 vaccine and myocarditis by age and sex in individuals aged 16–29 years. ACIP meeting, 30 Aug 2021. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides‐2021‐08‐30/06‐COVID‐Rosenblum‐508.pdf (viewed Feb 2022).
- 34. Marshall M, Ferguson ID, Lewis P, et al. Symptomatic acute myocarditis in 7 adolescents after Pfizer‐BioNTech COVID‐19 vaccination. Pediatrics 2021; 148: e2021052478.
- 35. Snapiri O, Rosenberg Danziger C, Shirman N, et al. Transient cardiac injury in adolescents receiving the BNT162b2 mRNA COVID‐19 vaccine. Pediatr Infect Dis J 2021; 40: e360–e363.
- 36. Barda N, Dagan N, Ben‐Shlomo Y, et al. Safety of the BNT162b2 mRNA Covid‐19 vaccine in a nationwide setting. N Engl J Med 2021; 385: 1078–1090.
- 37. Su JR. Adverse events among children ages 5‐11 years after COVID‐19 vaccination: updates from v‐safe and the Vaccine Adverse Event Reporting System (VAERS). Centers for Disease Control and Prevention, 13 Dec 2021. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides‐2021‐12‐16/05‐COVID‐Su‐508.pdf (viewed Feb 2022).
- 38. Boyarsky BJ, Werbel WA, Avery RK, et al. Antibody Response to 2‐Dose SARS‐CoV‐2 mRNA Vaccine Series in Solid Organ Transplant Recipients. JAMA 2021; 325: 2204–2206.
- 39. Grupper A, Rabinowich L, Schwartz D, et al. Reduced humoral response to mRNA SARS‐CoV‐2 BNT162b2 vaccine in kidney transplant recipients without prior exposure to the virus. Am J Transplant 2021; 21: 2719–2726.
- 40. Herishanu Y, Avivi I, Aharon A, et al. Efficacy of the BNT162b2 mRNA COVID‐19 vaccine in patients with chronic lymphocytic leukemia. Blood 2021; 137: 3165–3173.
- 41. Parry H, McIlroy G, Bruton R, et al. Antibody responses after first and second Covid‐19 vaccination in patients with chronic lymphocytic leukaemia. Blood Cancer J 2021; 11: 136.
- 42. Sattler A, Schrezenmeier E, Weber UA, et al. Impaired humoral and cellular immunity after SARS‐CoV‐2 BNT162b2 (tozinameran) prime‐boost vaccination in kidney transplant recipients. J Clin Invest 2021; 131: e150175.
- 43. Tenforde MW, Patel MM, Ginde AA, et al. Effectiveness of SARS‐CoV‐2 mRNA vaccines for preventing Covid‐19 hospitalizations in the United States. Clin Infect Dis 2021; https://doi.org/10.1093/cid/ciab687 (online ahead of print).
- 44. Brosh‐Nissimov T, Orenbuch‐Harroch E, Chowers M, et al. BNT162b2 vaccine breakthrough: clinical characteristics of 152 fully vaccinated hospitalized COVID‐19 patients in Israel. Clin Microbiol Infect 2021; 27: 1652–1657.
- 45. New Zealand Government Ministry of Health. COVID‐19 vaccine: severely immunocompromised people. https://www.health.govt.nz/covid‐19‐novel‐coronavirus/covid‐19‐vaccines/covid‐19‐vaccine‐severely‐immunocompromised‐people (viewed Feb 2022).
- 46. Department of Health and Social Care. Joint Committee on Vaccination and Immunisation (JCVI) advice on third primary dose vaccination. Published 1 Sept 2021. https://www.gov.uk/government/publications/third‐primary‐covid‐19‐vaccine‐dose‐for‐people‐who‐are‐immunosuppressed‐jcvi‐advice/joint‐committee‐on‐vaccination‐and‐immunisation‐jcvi‐advice‐on‐third‐primary‐dose‐vaccination (viewed Feb 2022).
- 47. Payne RP, Longet S, Austin JA, et al. Immunogenicity of standard and extended dosing intervals of BNT162b2 mRNA vaccine. Cell 2021; 184: 5699–5714.e11.
- 48. Government of Canada National Advisory Committee on Immunization, 25 January 2022. https://www.canada.ca/en/public‐health/services/immunization/national‐advisory‐committee‐on‐immunization‐naci/updated‐recommendations‐use‐covid‐19‐vaccines‐children‐5‐11‐years‐age.html (viewed Feb 2022).
- 49. Shimabukuro TT, Cole M, Su JR. Reports of anaphylaxis after receipt of mRNA COVID‐19 vaccines in the US — December 14, 2020‐January 18, 2021. JAMA 2021; 325: 1101–1102.
- 50. Australasian Society of Clinical Immunology and Allergy. Allergy, immunodeficiency, autoimmunity and COVID‐19 vaccination position statement. Update 20 Sept 2021. https://www.allergy.org.au/images/stories/pospapers/ASCIA_HP_Position_Statement_COVID‐19_Vaccination_2021‐09‐20.pdf (viewed Feb 2022).
- 51. The Immunisation Advisory Centre. Update around management of those with allergic reaction to their first dose of Comirnaty or history of PEG allergy. 17 Sept 2021. https://covid.immune.org.nz/sites/default/files/2021‐09/Update%20around%20management%20of%20those%20with%20allergic%20reaction%20to%20their%20first%20dose%20of%20Comirnaty%20or%20history%20of%20PEG%20allergy.pdf (viewed Feb 2022).
- 52. Tong WH, Pieters R, Kaspers GJL, et al. A prospective study on drug monitoring of PEG asparaginase and Erwinia asparaginase and asparaginase antibodies in pediatric acute lymphoblastic leukemia. Blood 2014; 123: 2026–2033.
- 53. Banerji A, Wickner PG, Saff R, et al. mRNA Vaccines to Prevent COVID‐19 Disease and Reported Allergic Reactions: Current Evidence and Suggested Approach. J Allergy Clin Immunol Pract 2021; 9: 1423–1437.
- 54. Australian Government Department of Health. COVID‐19 vaccination – guidance on myocarditis and pericarditis after mRNA COVID‐19 vaccines. Last updated: 2 Dec 2021. https://www.health.gov.au/resources/publications/covid‐19‐vaccination‐guidance‐on‐myocarditis‐and‐pericarditis‐after‐mrna‐covid‐19‐vaccines (viewed Feb 2022).
- 55. Volkova M, Russell R 3rd. Anthracycline cardiotoxicity: prevalence, pathogenesis and treatment. Curr Cardiol Rev 2011; 7: 214–220.
- 56. Conyers R, Costello B, La Gerche A, et al. Chemotherapy‐related cardiotoxicity: are Australian practitioners missing the point? Intern Med J 2017; 47: 1166–1172.
- 57. Boyarsky BJ, Werbel WA, Avery RK, et al. Immunogenicity of a single dose of SARS‐CoV‐2 messenger RNA vaccine in solid organ transplant recipients. JAMA 2021; 325: 1784–1786.
- 58. Geisen UM, Berner DK, Tran F, et al. Immunogenicity and safety of anti‐SARS‐CoV‐2 mRNA vaccines in patients with chronic inflammatory conditions and immunosuppressive therapy in a monocentric cohort. Ann Rheum Dis 2021; 80: 1306–1311.
- 59. Bird S, Panopoulou A, Shea RL, et al. Response to first vaccination against SARS‐CoV‐2 in patients with multiple myeloma. Lancet Haematol 2021; 8: e389–e392.
- 60. Maneikis K, Šablauskas K, Ringelevičiūtė U, et al. Immunogenicity of the BNT162b2 COVID‐19 mRNA vaccine and early clinical outcomes in patients with haematological malignancies in Lithuania: a national prospective cohort study. Lancet Haematol 2021; 8: e583–e592.
- 61. Monin L, Laing AG, Muñoz‐Ruiz M, et al. Safety and immunogenicity of one versus two doses of the COVID‐19 vaccine BNT162b2 for patients with cancer: interim analysis of a prospective observational study. Lancet Oncol 2021; 22: 765–778.
- 62. Pai VB, Nahata MC. Cardiotoxicity of chemotherapeutic agents: incidence, treatment and prevention. Drug Saf 2000; 22: 263–302.
- 63. Woods T, Vidarsson B, Mosher D, Stein JH. Transient effusive‐constrictive pericarditis due to chemotherapy. Clin Cardiol 1999; 22: 316–318.
- 64. Reykdal S, Sham R, Kouides P. Cytarabine‐induced pericarditis: a case report and review of the literature of the cardio‐pulmonary complications of cytarabine therapy. Leuk Res 1995; 19: 141–144.
- 65. Yamada T, Tsurumi H, Hara T, et al. Cytarabine‐induced pericarditis. Rinsho Ketsueki 1998; 39: 1115–1120.
- 66. Gähler A, Hitz F, Hess U, Cerny T. Acute pericarditis and pleural effusion complicating cytarabine chemotherapy. Onkologie 2003; 26: 348–350.
- 67. Hermans C, Straetmans N, Michaux JL, Ferrant A. Pericarditis induced by high‐dose cytosine arabinoside chemotherapy. Ann Hematol 1997; 75: 55–57.
- 68. Hengel CL, Russell PA, Gould PA, Kaye DM. Subacute anthracycline cardiotoxicity. Heart Lung Circ 2006; 15: 59–61.
- 69. Waissengrin B, Agbarya A, Safadi E, Padova H, Wolf I. Short‐term safety of the BNT162b2 mRNA COVID‐19 vaccine in patients with cancer treated with immune checkpoint inhibitors. Lancet Oncol 2021; 22: 581–583.
- 70. Baldassarre LA, Yang EH, Cheng RK, et al. Cardiovascular Care of the Oncology Patient During COVID‐19: An Expert Consensus Document From the ACC Cardio‐Oncology and Imaging Councils. J Natl Cancer Inst 2021; 113: 513–522.
- 71. Lazarus G, Budiman RA, Rinaldi I. Does immune checkpoint inhibitor increase the risks of poor outcomes in COVID‐19‐infected cancer patients? A systematic review and meta‐analysis. Cancer Immunol Immunother 2021; 71: 373–386.
- 72. Lyon AR, Yousaf N, Battisti NML, Moslehi J, Larkin J. Immune checkpoint inhibitors and cardiovascular toxicity. Lancet Oncol 2018; 19: e447–e458.
- 73. Yri OE, Torfoss D, Hungnes O, et al. Rituximab blocks protective serologic response to influenza A (H1N1) 2009 vaccination in lymphoma patients during or within 6 months after treatment. Blood 2011; 118: 6769–6771.
- 74. Therapeutic Goods Administration. Australian product information – Kymriah (Tisagenlecleutel) suspension. https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP‐2021‐PI‐02122‐1&d=20210916172310101 (viewed Feb 2022).
- 75. Dhakal B, Abedin S, Fenske T, et al. Response to SARS‐CoV‐2 vaccination in patients after hematopoietic cell transplantation and CAR‐T cell therapy. Blood 2021; 138: 1278–1281.
- 76. Gastinne T, Le Bourgeois A, Coste‐Burel M et al. Safety and antibody response after one and/or two doses of BNT162b2 Anti‐SARS‐CoV‐2 mRNA vaccine in patients treated by CAR T cells therapy. Br J Haematol 2022; 196: 360–362.
- 77. Tomblyn M, Chiller T, Einsele H, et al. Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow Transplant 2009; 15: 1143–1238.
- 78. Cordonnier C, Einarsdottir S, Cesaro S, et al. Vaccination of haemopoietic stem cell transplant recipients: guidelines of the 2017 European Conference on Infections in Leukaemia (ECIL 7). Lancet Infect Dis 2019; 19: e200–e212.
- 79. British Society for Haematology. COVID‐19 vaccination in patients with haematological disorders. 21 Dec 2020. https://b‐s‐h.org.uk/media/19195/haematology‐covid‐19‐v10‐vaccination‐statement‐231220.pdf (viewed Feb 2022).
- 80. European Society for Blood and Marrow Transplantation. Coronavirus disease COVID‐19: EBMT recommendations. Version 17, 26 Jan 2022. https://www.ebmt.org/sites/default/files/2022‐02/EBMT%20COVID‐19%20guidelines%20v.17.2.pdf (viewed Feb 2022).
- 81. Australia and New Zealand Transplant and Cellular Therapies. COVID‐19 vaccination consensus position statement. https://anztct.org.au/wp‐content/uploads/2021/02/FINAL‐ANZTCT‐COVID19‐Vaccination‐Position‐Statement‐Version2‐2‐Feb‐2021.pdf (viewed Feb 2022).
- 82. Le Bourgeois A, Coste‐Burel M, Guillaume T, et al. Safety and antibody response after 1 and 2 doses of BNT162b2 mRNA vaccine in recipients of allogeneic hematopoietic stem cell transplant. JAMA Netw Open 2021; 4: e2126344.
- 83. Kotecha RS. Challenges posed by COVID‐19 to children with cancer. Lancet Oncol 2020; 21: e235.
- 84. Kotecha RS. COVID‐19 vaccination for children with cancer. Pediatr Blood Cancer 2021; 69: e29340.





Abstract
Introduction: The Australian Technical Advisory Group on Immunisation and New Zealand Ministry of Health recommend all children aged ≥ 5 years receive either of the two mRNA COVID‐19 vaccines: Comirnaty (Pfizer), available in both Australia and New Zealand, or Spikevax (Moderna), available in Australia only. Both vaccines are efficacious and safe in the general population, including children. Children and adolescents undergoing treatment for cancer and immunosuppressive therapy for non‐malignant haematological conditions are particularly vulnerable, with an increased risk of severe or fatal COVID‐19. There remains a paucity of data regarding the immune response to COVID‐19 vaccines in immunosuppressed paediatric populations, with data suggestive of reduced immunogenicity of the vaccine in immunocompromised adults.
Recommendations: Considering the safety profile of mRNA COVID‐19 vaccines and the increased risk of severe COVID‐19 in immunocompromised children and adolescents, COVID‐19 vaccination is strongly recommended for this at‐risk population. We provide a number of recommendations regarding COVID‐19 vaccination in this population where immunosuppressive, chemotherapeutic and/or targeted biological agents are used. These include the timing of vaccination in patients undergoing active treatment, management of specific situations where vaccination is contraindicated or recommended under special precautions, and additional vaccination recommendations for severely immunocompromised patients. Finally, we stress the importance of upcoming clinical trials to identify the safest and most efficacious vaccination regimen for this population.
Changes in management as a result of this statement: This consensus statement provides recommendations for COVID‐19 vaccination in children and adolescents aged ≥ 5 years with cancer and immunocompromising non‐malignant haematological conditions, based on evidence, national and international guidelines and expert opinion.
Endorsed by: The Australian and New Zealand Children’s Haematology/Oncology Group.