The known: By 12 March 2020, 140 cases of COVID‐19 (the illness caused by SARS‐CoV‐2) had been confirmed in Australia; three patients had died. At the end of January, the sequence of the virus had been shared but no laboratory outside China had grown the virus or had access to live virus.
The new: We describe the clinical course and laboratory features of the first reported case of COVID‐19 in Australia, as well as the isolation, sequencing, imaging, and rapid global sharing of virus isolated from the patient.
The implications: Rapid identification, propagation and international sharing of SARS‐CoV‐2 is an important step in collaborative scientific efforts and diagnostic test validation in response to this public health emergency.
The recognition in 2019 of the first outbreak in Wuhan, China, of a respiratory disease (COVID‐19) associated with a novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) triggered an international response unparalleled in its scale and tempo.1,2,3,4,5,6 In particular, the rapid sharing and integration of clinical and epidemiological data has facilitated understanding of the spectrum of clinical disease caused by SARS‐CoV‐2 and the extent of its global spread, although there are still many unanswered questions. Further, rapid genomic analyses have corroborated epidemiological investigations, suggesting a global point source outbreak of a novel betacoronavirus originating in Wuhan.2,5
The fundamental pillars of the control of any infectious disease are effective prevention, diagnostic, and treatment strategies. For viral pathogens, the propagation of live virus and the timely dissemination of the viral isolate to domestic and international scientific and public health agencies are critical. Rapid sharing of material has allowed laboratories to validate their diagnostic assays and to confirm their ability to detect SARS‐CoV‐2. In this report we describe the clinical course and laboratory features of the first reported case of COVID‐19 in Australia, together with the isolation, sequencing, and imaging of the virus.
Case report and clinical course
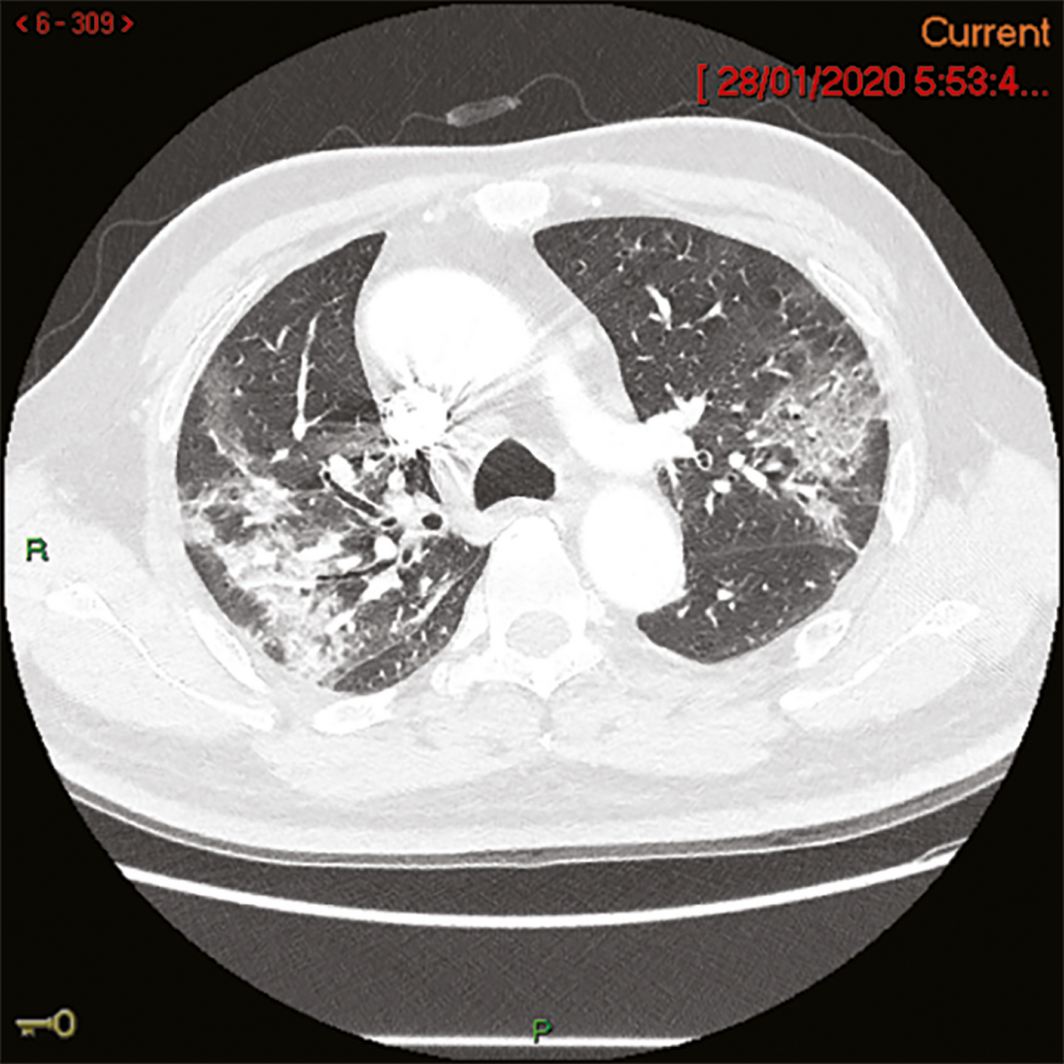
A 58‐year‐old man from Wuhan, China, felt unwell on the day of his arrival in Melbourne (19 January 2020). In China, he had had no contact with live food markets, people known to have COVID‐19, or hospitals. His medical history included type 2 diabetes mellitus, and he had ceased smoking four years previously. He developed fever on 20 January and a cough with sputum production on 23 January; on 24 January, he was admitted to the Monash Medical Centre, Melbourne, from its emergency department with progressive dyspnoea. His temperature was 38.1°C, his heart rate 95 beats/min, and O2 saturation 94% on room air. A chest x‐ray showed subtle ill‐defined opacities in the middle zones bilaterally and in the left lower zone. A thoracic computed tomography scan on admission day four identified extensive ground glass opacities with a peribronchovascular and peripheral distribution in the middle to upper zones of the lungs (Box 1). Full blood examination results included a lymphocyte count of 0.80 × 109/L (reference range, 1.0–4.0 × 109/L). C‐reactive protein concentration peaked on admission day 6 at 182 mg/L (reference range, 0–5 mg/L). Liver function test abnormalities peaked on admission day 12 — alkaline phosphatase, 210 U/L (reference range, 30–110 U/L); γ‐glutamyltransferase, 416 U/L (reference range, 30–110 U/L); alanine aminotransferase, 183 U/L (reference range, 5–40 U/L) — and hepatic steatosis was evident on liver ultrasound. Intravenous ceftriaxone (2 g/day) and azithromycin (500 mg/day) were commenced on admission day 4 to treat potential secondary bacterial pneumonia, although no bacterial pathogen was identified. Low‐flow oxygen (maximum 3 L/min via nasal prongs) was administered until admission day 10. The patient gradually improved; fever, productive cough and dyspnoea resolved by admission day 12, and he was discharged from hospital on 7 February (admission day 15).
Methods
Diagnostic testing for SARS‐CoV‐2
Real time reverse transcription (RT) polymerase chain reaction (PCR) testing for SARS‐CoV‐2 was performed on material from an initial nasopharyngeal swab in 200 μL viral transport medium, and separately for sputum, urine, faeces, and serum samples. Briefly, an in‐house real time RT‐PCR assay was developed, and all positive tests confirmed by nested RT‐PCR, using previously described methods.7 PCR products underwent in‐house Sanger sequencing, which confirmed the presence of SARS‐CoV‐2 (online Supporting Information, 1.1–1.3).
Virus culture and electron microscopy
Material from the initial nasopharyngeal swab was used to inoculate a Vero/hSLAM cell line (European Collection of Authenticated Cell Cultures [ECACC] #04091501). Flasks were monitored for the development of viral cytopathic effect and 140 μL aliquots removed every 48 hours to assess virus burden by real time RT‐PCR.
For electron microscopy, a 4 mL aliquot of supernatant from cell cultures grown in the presence of 4 μg/mL trypsin was inactivated with 0.5% glutaraldehyde for 12 h and clarified by centrifugation at 1000 g for 3 min. Supernatant was negatively stained with 3% phosphotungstic acid (pH 7.0) and examined with an FEI Tecnai T12 electron microscope at 80kV. The remaining pellet was stained en bloc and embedded in resin; 70 nm sections were examined with an FEI Tecnai F30 electron microscope at 200kV (Supporting Information, 2.1–2.2).
Whole genome sequencing of SARS‐CoV‐2 and bioinformatic analysis
We extracted RNA for whole genome sequencing of the viral isolate. Briefly, RNA was extracted from clarified cell culture supernatant and randomly amplified cDNA prepared by sequence‐independent single‐primer amplification (SISPA).8,9 Sequencing was performed with a combination of Oxford Nanopore Technologies and Illumina short‐read sequencing. Genomic assembly of the BetaCoV/Australia/VIC/01/2020 genome was confirmed by parallel de novo and reference‐guided methods (Supporting Information, 3.1–3.4).
Results
Detection of SARS‐CoV‐2 in clinical samples
A nasopharyngeal swab and sputum collected on presentation were positive for SARS‐CoV‐2 on real time RT‐PCR assay. Serial daily RT‐PCR testing of nasopharyngeal swabs and sputum from the patient indicated a gradual decline in viral load in sputum between admission days 1 and 8, and a decline in viral load and disappearance from nasopharyngeal swabs by admission day 7. No virus was detected in urine samples, nor in single faecal (admission day 3) or plasma samples (admission day 1) (Box 2).
Growth, visualisation, and global sharing of SARS‐CoV‐2 virus
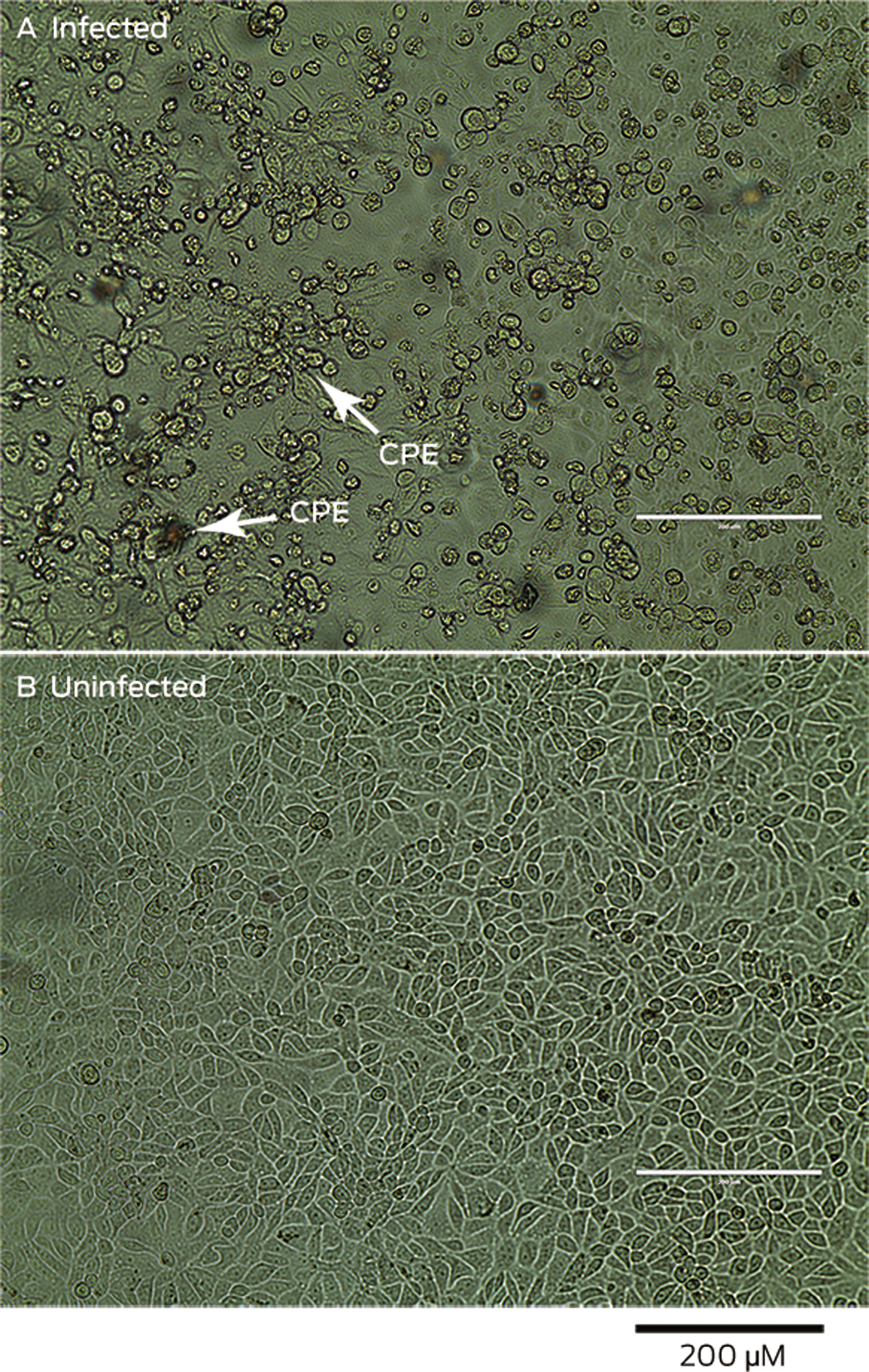
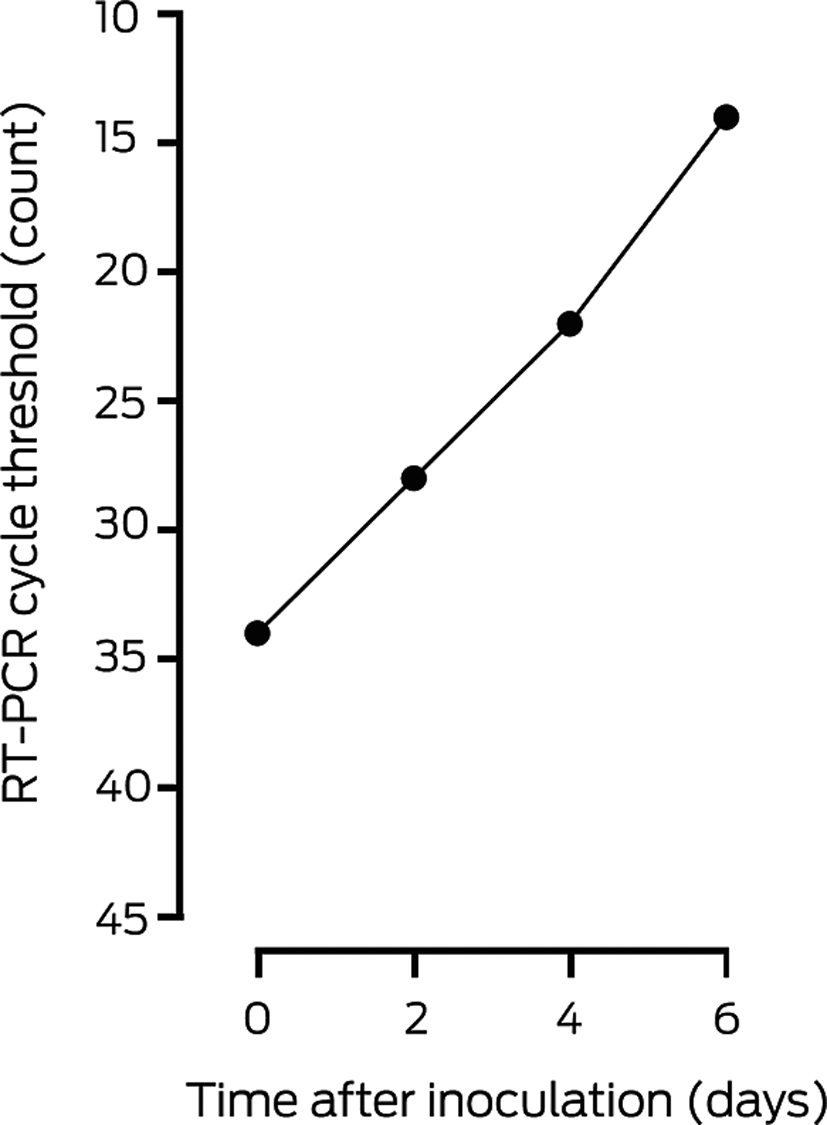
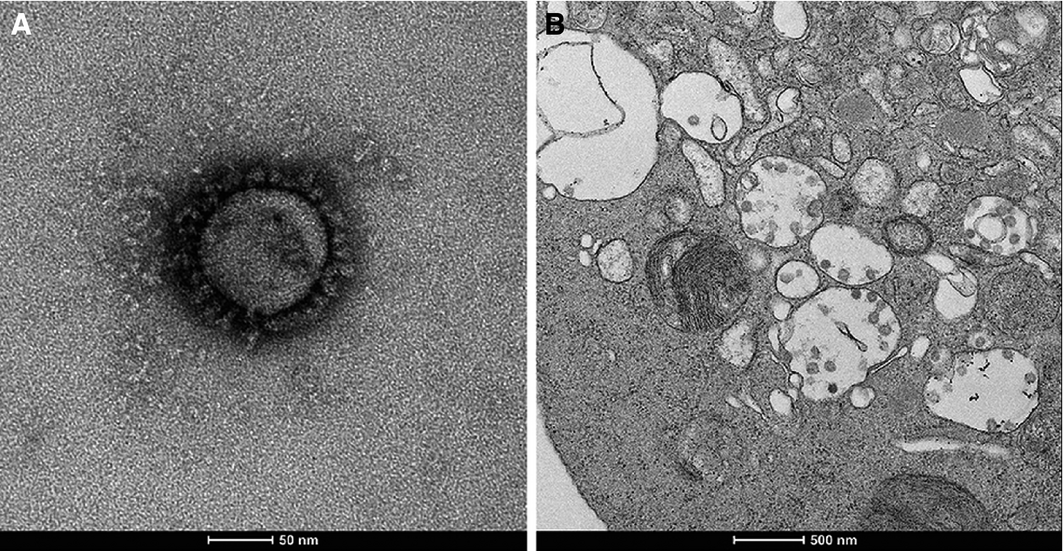
Two days after inoculation of the VERO/hSLAM cell line, a subtle viral cytopathic effect was observed, and was distinct at day 6 compared with an uninfected control cell line (Box 3). RT‐PCR testing of the cell line supernatant confirmed a high viral load, suggesting productive viral infection (Box 4). Electron micrographs of the negatively stained supernatant showed spherical and pleomorphic virus‐like particles of 90–110 nm diameter; the particles displayed prominent spikes (9–12 nm), characteristic of viruses from the family Coronaviridae (Box 5, A). Electron micrographs of sectioned VERO/hSLAM cells showed cytoplasmic membrane‐bound vesicles containing coronavirus particles (Box 5, B) Following several failures to recover virions with the characteristic fringe of surface spike proteins, it was found that adding trypsin to the cell culture medium immediately improved virion morphology.
In consultation with the World Health Organization, the viral isolate was shared with domestic and international reference laboratories within 24 hours, and lodgement with major North American and European culture collections for further distribution is underway.
Phylogenetic analysis
Phylogenetic analysis indicated that the genome sequence of our isolate (BetaCoV/Australia/VIC01/2020) exhibited greater than 99.99% sequence identity with other publicly available SARS‐CoV‐2 genomes (online Supporting Information, 3.4), consistent with the epidemiological features of this case originating in Wuhan.10 Compared with the National Center for Biotechnology Information (NCBI) SARS‐CoV‐2 reference sequence (NC_045512.3), there were three previously described single nucleotide polymorphisms and a 10 base pair deletion in the 3’ untranslated region (3'UTR) (Supporting Information, 3.4). Our sequences are available at GenBank (accession number, MT007544.1), and the genome was rapidly uploaded to the Global Initiative of Sharing All Influenza (GISAID) (accession number, EPI_ISL_406844).
Discussion
We have described the first reported case of COVID‐19 in Australia, with rapid diagnosis, and isolation, imaging, and sharing of the causative agent, SARS‐CoV‐2. By 12 March 2020, there had been 140 confirmed cases in Australia; three patients had died.11 Although 65% of confirmed cases around the world have been reported from China, an increasing number are being reported in South Korea, Italy, and Iran, and limited human‐to‐human transmission has been described.6,10,11,12 Although the number of cases in Australia is relatively small, the political and societal effects (as in other countries) have already been considerable, including travel restrictions to and from mainland China, the Republic of Korea, Italy, and Iran.13 The sustainability of these measures and their effects on local and global control remain to be established, but the consequences of the outbreak will probably be felt for many months, if not longer.
The clinical features in our case were consistent with other recent reports, including the initial presentation of fever, cough, and progressive dyspnoea.3,6 It is notable that the viral burden was greatest in sputum specimens, which remained positive for SARS‐CoV‐2 for eight days after initial presentation, compared with four days for nasopharyngeal swabs (Box 2). The decline in viral load was correlated with the resolution of fever and, ultimately, clinical improvement. One unresolved question is whether patients who are clinically stable and deemed fit to be discharged from hospital but have PCR‐detectable virus are infectious, or whether this indicates only the persistence of non‐infectious, residual viral RNA.
We applied standard techniques to isolate the virus, but we were the first group to isolate it outside China during the early stages of the epidemic. Potential reasons for our success could be the viral burden of the collected specimens and the extensive clinical experience in our reference laboratory.
An important aspect of the scientific response to the COVID‐19 outbreak has been the rapid sharing of information about diagnostic assays and genomic data, enabling rapid elucidation of the emergence and spread of the novel virus. In addition, a major principle of our laboratory response in Australia was to immediately share the viral isolate with the WHO and other laboratories to facilitate rapid validation of diagnostic testing. We continue to share live virus with other agencies, both locally and overseas, involved in the development and testing of therapeutic agents and vaccines. This is an essential function of public health reference and research laboratories, and we strongly encourage others to apply a similarly collaborative approach to streamlining efforts to diagnose, prevent, and treat COVID‐19 during this public health emergency.
Box 2 – Clinical course of first Australian case of COVID‐19, including laboratory investigations
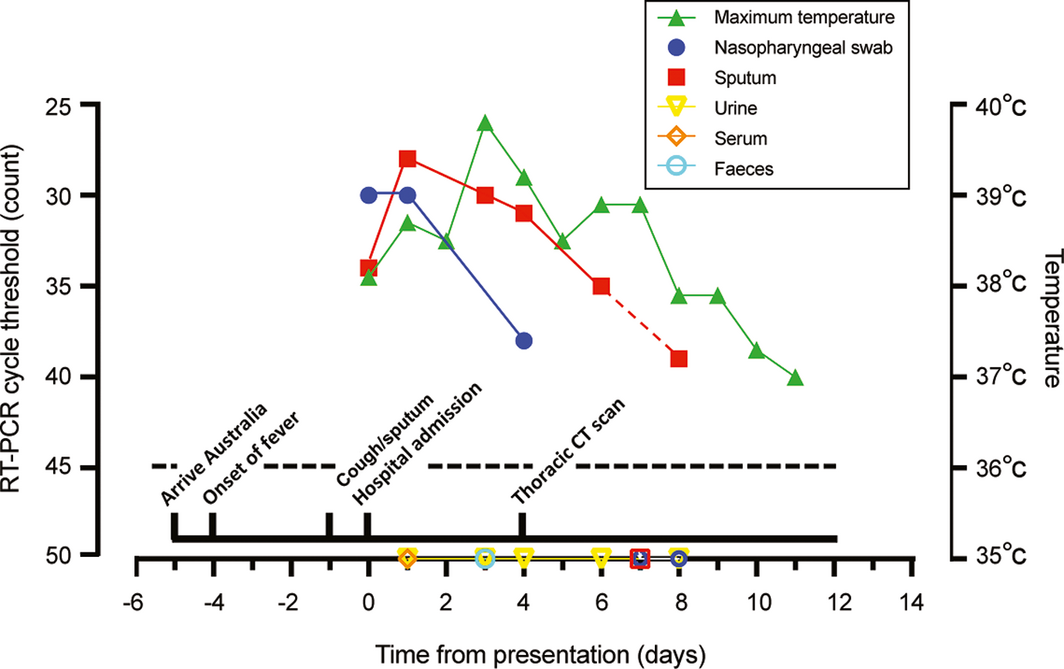
SARS‐CoV‐2 was quantified by real time RT‐PCR. The cycle threshold count is shown for each specimen type; an increase in count value is consistent with reduced viral load. The assay limit of detection (dashed line) threshold is a count of 45; open symbols beneath the threshold indicate null detection of virus.
Box 3 – Light microscopy of Vero/hSLAM cells. A. Cells infected with material from patient, with viral cytopathic effect (CPE) evident six days after inoculation. B. Uninfected (control) cells
Received 25 February 2020, accepted 5 March 2020
- Leon Caly1
- Julian Druce1
- Jason Roberts1
- Katherine Bond1
- Thomas Tran1
- Renata Kostecki1
- Yano Yoga1
- William Naughton2
- George Taiaroa3
- Torsten Seemann5
- Mark B Schultz5
- Benjamin P Howden5
- Tony M Korman2
- Sharon R Lewin3,4
- Deborah A Williamson5,6
- Mike G Catton1
- 1 Victorian Infectious Diseases Reference Laboratory, Melbourne Health at the Peter Doherty Institute for Infection and Immunity, Melbourne, VIC
- 2 Monash Medical Centre, Melbourne, VIC
- 3 Peter Doherty Institute for Infection and Immunity, University of Melbourne and Royal Melbourne Hospital, Melbourne, VIC
- 4 The Alfred, Melbourne, VIC
- 5 Microbiological Diagnostic Unit Public Health Laboratory, University of Melbourne at the Peter Doherty Institute for Infection and Immunity, Melbourne, VIC
- 6 Melbourne Health, Melbourne, VIC
Sharon Lewin receives research support from the National Health and Medical Research Council (NHMRC). Sharon Lewin and Benjamin Howden are NHMRC Practitioner Fellows; Deborah Williamson holds an NHMRC Investigator grant. Sharon Lewin and Mike Catton are supported by the Australian Partnership for Preparedness Research in Infectious Diseases Emergencies (APPRISE), an NHMRC‐funded Centre for Research Excellence.We acknowledge our public health partners and the Victorian Department of Health and Human Services, the major funder of the Victorian Infectious Diseases Reference Laboratory, without whom this work would not have been possible. We also thank the clinical and laboratory staff involved in the care of our patient. We thank Andrew Leis and Eric Hanssen (Melbourne Advanced Microscopy Facility, Bio21 Institute) for their assistance with the FEI L120C (during preliminary screening) and FEI F30 microscopes, and Susan Ballard and Michelle Sait (Microbiological Diagnostic Unit Public Health Laboratory, University of Melbourne) for assistance with Illumina sequencing. We also thank Dave O'Connor (University of Wisconsin Medical Foundation) for his support and assistance in establishing the SISPA protocol at the Doherty Institute. We thank Jasminka Sterjovski for assisting with preparation and submission of the manuscript.
No relevant disclosures.
- 1. Wu F, Zhao S, Yu B, et al. A new coronavirus associated with human respiratory disease in China. Nature 2020; 579: 265–269.
- 2. Zhou P, Yang XL, Wang XG, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020; 579: 270–273.
- 3. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395: 497–506.
- 4. Wang C, Horby PW, Hayden FG, Gao GF. A novel coronavirus outbreak of global health concern. Lancet 2020; 395: 470–473.
- 5. Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020; 382: 727–733.
- 6. Holshue ML, DeBolt C, Lindquist S, et al. First case of 2019 novel coronavirus in the United States. N Engl J Med 2020; 382: 929–936.
- 7. Chu DK, Leung CY, Gilbert M, et al. Avian coronavirus in wild aquatic birds. J Virol 2011; 85: 12815–12820.
- 8. Lewandowski K, Xu Y, Pullan ST, et al. Metagenomic nanopore sequencing of influenza virus direct from clinical respiratory samples. J Clin Microbiol 2019; 58: e00963–19.
- 9. Kafetzopoulou LE, Efthymiadis K, Lewandowski K, et al. Assessment of metagenomic Nanopore and Illumina sequencing for recovering whole genome sequences of chikungunya and dengue viruses directly from clinical samples. Euro Surveill 2018; 23: 1800228.
- 10. Lu R, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet 2020; 395: 565–574.
- 11. World Health Organization. Coronavirus disease 2019 (COVID‐19). Situation report 53. 13 Mar 2020. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200313-sitrep-53-covid-19.pdf?sfvrsn=adb3f72_2 (viewed 14 Mar 2020).
- 12. Phan LT, Nguyen TV, Luong QC, et al. Importation and human‐to‐human transmission of a novel coronavirus in Vietnam. N Engl J Med 2020; 382: 872–874.
- 13. Australian Department of Health. Coronavirus (COVID‐19) health alert. https://www.health.gov.au/news/health-alerts/novel-coronavirus-2019-ncov-health-alert#travellers-and-visitors (viewed 13 Mar 2020).





Abstract
Objectives: To describe the first isolation and sequencing of SARS‐CoV‐2 in Australia and rapid sharing of the isolate.
Setting: SARS‐CoV‐2 was isolated from a 58‐year‐old man from Wuhan, China who arrived in Melbourne on 19 January 2020 and was admitted to the Monash Medical Centre, Melbourne from the emergency department on 24 January 2020 with fever, cough, and progressive dyspnoea.
Major outcomes: Clinical course and laboratory features of the first reported case of COVID‐19 (the illness caused by SARS‐CoV‐2) in Australia; isolation, whole genome sequencing, imaging, and rapid sharing of virus from the patient.
Results: A nasopharyngeal swab and sputum collected when the patient presented to hospital were each positive for SARS‐CoV‐2 (reverse transcription polymerase chain reaction). Inoculation of Vero/hSLAM cells with material from the nasopharyngeal swab led to the isolation of SARS‐CoV‐2 virus in culture. Electron microscopy of the supernatant confirmed the presence of virus particles with morphology characteristic of viruses of the family Coronaviridae. Whole genome sequencing of the viral isolate and phylogenetic analysis indicated the isolate exhibited greater than 99.99% sequence identity with other publicly available SARS‐CoV‐2 genomes. Within 24 hours of isolation, the first Australian SARS‐CoV‐2 isolate was shared with local and overseas reference laboratories and major North American and European culture collections.
Conclusions: The ability to rapidly identify, propagate, and internationally share our SARS‐CoV‐2 isolate is an important step in collaborative scientific efforts to deal effectively with this international public health emergency by developing better diagnostic procedures, vaccine candidates, and antiviral agents.