The known Rheumatic heart disease (RHD) is a disease of poverty that is highly prevalent in resource-limited settings and among Indigenous Australians and New Zealand Māori. The prevalence of RHD in Timor-Leste has not been described.
The new The prevalence of echocardiography-detected RHD in Timor-Leste is comparable with the highest rates in the world. All cases of RHD had previously been undiagnosed.
The implications A public health response for preventing and managing RHD in Timor-Leste is urgently needed, including improving the recognition of acute rheumatic fever. Further investigation is required to determine the best approaches for managing RHD detected by echocardiography.
Rheumatic heart disease (RHD) is a preventable condition that is now extremely rare in high income countries, although its prevalence in indigenous, migrant and refugee populations remains high.1-4 The prevalence of RHD is also high in low and middle income countries in sub-Saharan Africa, South America, Asia, and the Pacific, where high rates of Streptococcus pyogenes (group A streptococcus [GAS]) transmission have been documented.5,6 Group A streptococcal infections are associated with poverty, and cause a spectrum of disease ranging from skin infections and pharyngitis to invasive disease, including bacteraemia.5 Acute rheumatic fever (ARF) is a multisystem immune-mediated condition that can follow infection with GAS; GAS pharyngitis is associated with ARF, whereas a link between GAS pyoderma and ARF has been suggested but not confirmed.7 In RHD, chronic valvular damage results from carditis associated with repeated episodes of ARF. ARF recurrences and the associated progression of valvular disease in established RHD can be effectively prevented by 4-weekly injections of long-acting benzathine penicillin G (BPG).8,9
After 24 years of occupation, the people of Timor-Leste voted for independence from Indonesia in 1999; following 3 years of transitional administration by the United Nations, it achieved independence in 2002. During and immediately following the referendum and the subsequent withdrawal of the Indonesian army, 70% of the national infrastructure was destroyed, including 35% of health facilities; the vast majority of clinicians and health managers fled the country.10 Timor-Leste has been reconstructing its health system ever since. In a setting of limited resources such as Timor-Leste, the importance of research for identifying needs and driving improvements in health service delivery is clear.11
ARF is not notifiable in Timor-Leste, and its incidence is unknown. The limited capacity of the country for diagnostic microbiology means that the incidence of GAS infections is also unknown, although skin infections (including impetigo) are common.12 Anecdotal reports from clinicians in Timor-Leste suggest that the number of patients presenting with ARF to heath care facilities is small, but that the burden of RHD among children and young people is significant. Many patients present to local health services with end-stage heart disease, but access to surgery is limited. Some East Timorese can undergo cardiac surgery in other countries, funded by charitable organisations such as the East Timor Hearts Fund (ETHF) and Rotary Oceania Medical Aid for Children (ROMAC), but many succumb to their disease while still young.13
No published studies have described the burden of RHD in Timor-Leste. We therefore conducted an echocardiography screening study with the aims of describing the prevalence of RHD in school-aged children and young people in Timor-Leste, and of establishing an RHD register for coordinating secondary prophylaxis for East Timorese people with RHD.
Methods
Schools were recruited in the capital city of Dili and the inland district of Ermera to obtain a sample population representative of the different geographic regions and a spectrum of socio-economic status in Timor-Leste. A consultation visit to the schools was made 4 months prior to the commencement of screening. A plain language information sheet in Tetum was distributed to parents and families, giving them the opportunity to decline screening of their children. An opt-out approach to consent was adopted because it was strongly preferred by local school principals and community members; this method has been applied successfully in other studies.14
All students aged 5–20 years who attended school on the screening days were eligible to participate in the study. Demographic data for all students were collected on a standard form to reduce measurement bias; to maximise accuracy, only study staff fluent in the local language recorded the data. Date of birth, age, number of people in their household, and number of rooms in their home were based on student self-report. All participants were examined for evidence of impetigo or scabies; these results will be reported in a separate article. As the capacity for diagnostic microbiology in Timor-Leste is limited, samples for microbiological testing were not collected.
An echocardiogram was performed by one of five cardiologists (including two paediatric specialists) with a Vivid I or Q machine (GE Healthcare). All children had full screening echocardiograms, including parasternal long axis, parasternal short axis, apical four-chamber, and apical five-chamber views (2D and Doppler).
RHD was classified as borderline or definite according to World Heart Federation criteria (Box 1).15 Abnormal echocardiograms were flagged during the screening process and immediately reviewed by a paediatric cardiologist, as well as by all five cardiologists at the end of the screening week; a diagnosis of borderline or definite RHD was confirmed only after consensus was reached by at least three of the cardiologists involved in the study. Normal echocardiograms were not reviewed.
Statistical analysis
Data were entered into an Access 2016 database (Microsoft), and statistical analysis was conducted in Stata 13 (StataCorp). Descriptive statistical analysis was undertaken and the prevalence of RHD estimated with 95% confidence intervals (CIs). Continuous variables that were not normally distributed were compared in Mann–Whitney rank sum tests; differences in binary variables were assessed in χ2 tests (univariate analysis). Relative risks (RRs) and 95% CIs were calculated. For multivariate analyses, logistic regression was employed to estimate adjusted odds ratios (aORs) with 95% CIs. Sex, age, and location of screening were included in the logistic regression model because these variables could plausibly influence the prevalence of RHD. P < 0.05 was deemed statistically significant.
Patients with RHD were entered into a register to facilitate monitoring of adherence to subsequent secondary prophylaxis, with the first dose of BPG administered on the day of the study. Patients with heart disease amenable to surgery were referred to ROMAC for possible surgery in Australia.
Ethics approval
Ethics approval for the study was obtained from the Human Research Ethics Committee of the Northern Territory Department of Health and Menzies School of Health Research (reference, 2016-2546) and the Instituto Nacional de Saúde in Timor-Leste (reference, MS-INS/DF/DP/V/2016/220). Permission to undertake screening was granted by the Ministério da Educação in Timor-Leste and by the principals of the schools involved.
Results
A total of 1365 participants aged 5–20 years were screened by echocardiography. Their median age was 11 years (interquartile range [IQR], 9–14 years) and 725 (53%) were girls; the sex distribution was similar for all age groups (data not shown). Five hundred and five children (37%) were screened in Dili, 860 (63%) in the district of Ermera. All students knew their age, but fewer than half were able to report their date of birth. None knew whether they were allergic to penicillin. The median number of residents per household was 7 (IQR, 6–9) (Box 2).
A total of 25 definite and 23 borderline cases of RHD were detected by echocardiography. The estimated prevalence of definite RHD was 18.3 cases per 1000 population (95% CI, 12.3–27.0 per 1000); that of definite or borderline RHD was 35.2 cases per 1000 population (95% CI, 26.5–46.4 per 1000). None of the children in whom RHD was detected had previously been diagnosed with RHD; one had a history of ARF without evidence of carditis and of poor adherence to penicillin prophylaxis. All cases were classified as either mild or moderate, none as severe (Box 3). All borderline and definite cases included mild or moderate mitral regurgitation. No mitral stenosis was detected. Three children also had mild or moderate aortic regurgitation. Most of the children in whom definite or borderline RHD was detected lived in Ermera, where the estimated prevalence was 43.0 cases per 1000 population (95% CI, 31.2–58.9) (Box 3).
Eleven children (0.8%) had congenital heart disease. Two were referred for surgery (one case of severe aortic stenosis, one of Ebstein’s anomaly); nine children had relatively minor cardiac anomalies, including three with haemodynamically insignificant atrial septal defects, two with mitral valve prolapse, two with patent ductus arteriosus, one with dextrocardia, and one with mild pulmonary stenosis.
There was a clear sex difference in prevalence of RHD. There were 27.6 cases of definite RHD per 1000 girls (95% CI, 17.7–42.5) and 7.8 cases per 1000 boys (95% CI, 2.8–18.7) (aOR, 3.6; 95% CI, 1.3–9.5; P = 0.012); there were 49.7 cases of definite or borderline RHD per 1000 girls (95% CI, 35.9–68.2) and 18.8 cases per 1000 boys (95% CI, 10.4–32.9) (aOR, 2.7; 95% CI, 1.4–5.2; P = 0.004).
Students screened in Ermera appeared more likely to have definite or borderline RHD in the univariate analysis (RR, 2.0; 95% CI, 1.0–3.8; P = 0.040), but the multivariate analysis (adjusted for age and sex) detected no significant association between RHD and age or location (Box 4).
Twenty-one children with definite RHD (84%) received BPG and education on the day of screening, and all children were entered into the RHD register for follow-up. Children whose parents were not available on the day of screening did not initially receive BPG, but all children and families have subsequently received education about RHD and the importance of secondary prophylaxis, and have commenced treatment with BPG.
Discussion
Our echocardiography-based screening study identified a very high prevalence of RHD among young people in Timor-Leste, comparable with the highest documented rates of RHD elsewhere (Box 5).1,6,16-24 The prevalence of definite RHD in our study (18 cases per 1000 population) is higher than the rate for Fiji (7 cases per 1000), and similar to that in other regional neighbours, including the Top End of Australia (15 cases per 1000 for Indigenous Australians at high risk).1,18 The prevalence in Timor-Leste may be higher than estimated; we did not detect any cases of mitral stenosis or other forms of severe RHD, and it is possible that students with severe RHD were too unwell to attend school, and therefore not screened. It is also possible that children and young people from poorer families, at greater risk of RHD, do not regularly attend school. The absence of severe RHD in our study contrasts with anecdotal reports of fatal cases managed by Timor-Leste health services, and with the experience of visiting cardiology services,13 but the morbidity and mortality of severe RHD in Timor-Leste has not been formally documented.
Further investigation of the morbidity and mortality of RHD in Timor-Leste are needed, and, as RHD had not previously been diagnosed in any of the young people we screened, active case detection and close follow-up is warranted. The World Health Organization has recently renewed its resolution to tackle RHD as an important international health problem requiring a multisectoral response. The WHO recognises that RHD affects a substantial proportion of the world’s most vulnerable populations, and that antibiotic treatment of group A streptococcal infections and secondary prophylaxis for preventing ARF are cost-effective strategies that should be implemented in high burden settings.25
Efforts to reduce the prevalence of RHD in Timor-Leste should focus on primordial (reducing risk factors for GAS infection), primary (treating GAS infections to prevent ARF), and secondary prevention (penicillin prophylaxis for people with a history of ARF or established RHD). Access to cardiac surgery (tertiary prevention) is limited to cases amenable to international referral, usually to Australia, Indonesia or Singapore, as cardiac surgery is not currently performed in Timor-Leste.
Household sizes in this cohort were large, consistent with census data from Timor-Leste, and domestic crowding is a known risk factor for RHD.26 Access to health care services is limited, particularly in regional areas. Ermera, like much of Timor-Leste, is a mountainous area with very poor roads in some parts. Delivering BPG regularly is challenging, but has been successfully undertaken since our screening study. Our study has shown that it is possible to screen for and diagnose RHD, immediately enter patients into a register, provide education, and commence BPG prophylaxis on the same day. However, we relied on volunteer cardiologists with specialist skills in the echocardiographic diagnosis of RHD, and it would be difficult to reproduce this approach to screening on a large scale.
Registries for the surveillance and follow-up of patients with established RHD have been shown to be effective in reducing morbidity and mortality, especially when incorporated into national disease surveillance networks.25 Increasing the available information on disease burden by active surveillance could provide the impetus for providing more resources to the Timorese health care system, leading to long term improvement of local cardiac services, as well as to improving the chances of those with established RHD of undergoing surgery overseas. Most importantly, an effective register facilitates the delivery of penicillin prophylaxis that can halt the progression of RHD and prevent the sequelae of heart failure and death.8,9
We found that that risk of RHD in Timor-Leste is greater for girls. This is similar to findings in Australia and other parts of the world,1,27 although a significant sex difference was not detected by a recent meta-analysis.6 The cause of the sex difference is yet to be determined. The risk of morbidity and mortality during pregnancy and childbirth is very high in women with RHD, and increases with successive pregnancies.5 Catholicism is common in Timor-Leste, a country with a median household size of 5.7 people;28 access to contraception is limited, and societal opposition to its use significant.29 The prevalence of RHD among pregnant women in Timor-Leste is unknown, but multiple pregnancies in a setting of high prevalence RHD may place women and babies at increased risk of adverse outcomes and death.
There were several limitations to our study. Screening was only conducted in two districts of Timor-Leste. The prevalence of RHD in other districts may be different, but we expect that enrolling subjects from both an urban and a rural district resulted in a sufficiently representative sample for producing meaningful results. We enrolled only children and young people attending school, and those from poorer families or too sick to go to school will not have been included in our sample. The children screened in Ermera were older than those screened in Dili, but neither age nor location were significant factors in the multivariate analysis.
The cardiologists performing the echocardiograms provided a diagnosis during screening, and there was no external review of the images by a blinded cardiologist. However, a diagnosis was only provided after consensus among three of the five cardiologists was reached; it is very unlikely that the burden of RHD was overestimated. On the other hand, unremarkable echocardiograms were not reviewed, so it is possible that some cases were missed and the prevalence therefore underestimated. Some uncertainty regarding the clinical significance of borderline RHD remains, but recent studies have found that children with borderline RHD are more likely to have ARF, progression of their valvular lesions, and later development of definite RHD.30 Nevertheless, echocardiography screening for RHD case detection remains controversial, as there is no evidence that screening leads to improved outcomes.
Conclusion
We found a significant burden of undetected RHD in our sample of young people, particularly among girls and young women. Our finding of a large burden of undetected disease indicates that active case detection is needed in Timor-Leste. The health system of Timor-Leste needs to be improved to increase the capacity of health workers to recognise and manage ARF and RHD. The nascent RHD register needs to be expanded, and further investigations in Timor-Leste and elsewhere are required to better define the role and practice of echocardiography screening, and to guide secondary prophylaxis for patients in whom RHD is detected by screening.
Box 1 – World Heart Federation criteria for echocardiographic diagnosis of rheumatic heart disease (RHD) in people under 21 years of age (2012)15
|
|
|||||||||||||||
|
Definite rheumatic heart disease
|
|||||||||||||||
|
Borderline rheumatic heart disease
|
|||||||||||||||
|
Normal echocardiographic findings
|
|||||||||||||||
|
|
|||||||||||||||
|
* Congenital mitral valve anomalies must be excluded. † Bicuspid aortic valve, dilated aortic root, and hypertension must be excluded. ‡ Combined aortic and mitral regurgitation in high prevalence regions and in the absence of congenital heart disease is regarded as rheumatic. |
|||||||||||||||
Box 2 – Demographic data for people under 21 years of age screened for rheumatic heart disease in Dili or Ermera, Timor-Leste
|
|
Dili |
Ermera |
Total |
||||||||||||
|
|
|||||||||||||||
|
Total number |
505 |
860 |
1365 |
||||||||||||
|
Sex |
|
|
|
||||||||||||
|
Girls |
255 (50.5%) |
470 (54.7%) |
725 (53.1%) |
||||||||||||
|
Boys |
250 (49.5%) |
390 (45.3%) |
640 (46.9%) |
||||||||||||
|
Age (years) |
|
|
|
||||||||||||
|
5–9 |
233 (46.1%) |
180 (20.9%) |
413 (30.3%) |
||||||||||||
|
10–14 |
269 (53.3%) |
351 (40.8%) |
620 (45.4%) |
||||||||||||
|
15–20 |
3 (0.6%) |
329 (38.3%) |
332 (24.3%) |
||||||||||||
|
People per household, median (interquartile range) |
6 (4–8) |
8 (6–9) |
7 (6–9) |
||||||||||||
|
|
|||||||||||||||
|
|
|||||||||||||||
Box 3 – Estimated prevalence of rheumatic heart disease in people under 21 years of age in Dili or Ermera, Timor-Leste
|
|
Dili |
Ermera |
Total |
||||||||||||
|
Cases |
Prevalence, per 1000 (95% CI) |
Cases |
Prevalence, per 1000 (95% CI) |
Cases |
Prevalence, per 1000 (95% CI) |
||||||||||
|
|
|||||||||||||||
|
People with rheumatic heart disease |
|||||||||||||||
|
Definite |
7 |
13.9 (0.6–28.9) |
18 |
20.9 (13.1–33.1) |
25 |
18.3 (12.3–27.0) |
|||||||||
|
Borderline |
4 |
7.9 (0.2–21.0) |
19 |
22.1 (14.0–34.5) |
23 |
16.8 (11.1–25.3) |
|||||||||
|
Definite or borderline |
11 |
21.8 (11.7–39.0) |
37 |
43.0 (31.2–58.9) |
48 |
35.2 (26.5–46.4) |
|||||||||
|
Severity of definite or borderline rheumatic heart disease |
|||||||||||||||
|
Mild |
10 |
|
32 |
|
42 |
|
|||||||||
|
Moderate |
1 |
|
5 |
|
6 |
|
|||||||||
|
Severe |
0 |
|
0 |
|
0 |
|
|||||||||
|
|
|||||||||||||||
|
CI = confidence interval. |
|||||||||||||||
Box 4 – Univariate and multivariate analysis of prevalence of rheumatic heart disease in people under 21 years of age in Dili and Ermera, Timor-Leste
|
|
Definite rheumatic heart disease |
Definite and borderline rheumatic heart disease |
|||||||||||||
|
Cases |
Relative risk |
P |
Adjusted odds ratio* (95% CI) |
P |
Cases |
Relative risk (95% CI) |
P |
Adjusted odds ratio* (95% CI) |
P |
||||||
|
|
|||||||||||||||
|
Sex |
|
|
0.007 |
|
0.012 |
|
|
0.002 |
|
0.004 |
|||||
|
Boys |
5 (0.8%) |
1 |
|
1 |
|
12 (1.9%) |
1 |
|
1 |
|
|||||
|
Girls |
20 (2.8%) |
3.5 (1.3–9.4) |
|
3.6 (1.3–9.5) |
|
36 (5.0%) |
2.6 (1.4–5.0) |
|
2.7 (1.4–5.2) |
|
|||||
|
Age (years) |
|
|
|
|
|
|
|
|
|
|
|||||
|
5–9 |
5 (1%) |
|
|
1 |
|
9 (2%) |
|
|
1 |
|
|||||
|
10–14 |
15 (2.4%) |
|
|
1.9 (0.7–5.3) |
0.22 |
24 (3.9%) |
|
|
1.7 (0.8–3.7) |
0.20 |
|||||
|
15–20 |
5 (2%) |
|
|
1.0 (0.3–3.6) |
0.97 |
15 (4.5%) |
|
|
1.6 (0.7–3.9) |
0.31 |
|||||
|
Location |
|
|
|
|
|
|
|
0.040 |
|
|
|||||
|
Dili |
7 (1%) |
1 |
0.35 |
1 |
0.34 |
11 (2.2%) |
1 |
|
1 |
0.13 |
|||||
|
Ermera |
18 (2.1%) |
1.5 (0.6–3.6) |
|
1.6 (0.6–4.0) |
|
37 (4.3%) |
2.0 (1.0–3.8) |
|
1.8 (0.8.–3.7) |
|
|||||
|
|
|||||||||||||||
|
CI = confidence interval. * Adjusted for other two factors in table. |
|||||||||||||||
Box 5 – Prevalence in recent studies of definite and borderline rheumatic heart disease in young people detected by echocardiography according to World Heart Federation criteria
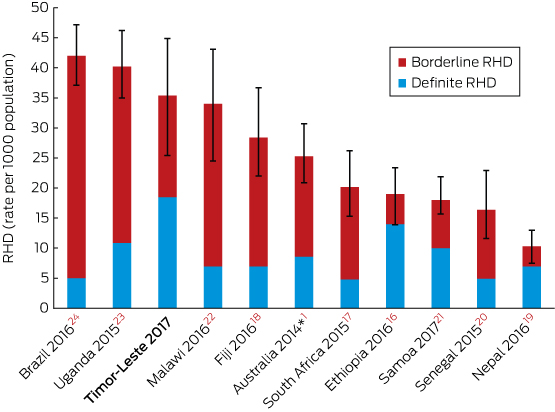
* Indigenous Australian population at high risk of rheumatic heart disease. All studies reported prevalence for people under 21 years of age, except the South African study (24 years or younger).
Received 10 July 2017, accepted 23 November 2017
- Kimberly Davis1,2
- Bo Remenyi1
- Anthony DK Draper3
- Januario Dos Santos4
- Noel Bayley5
- Elizabeth Paratz6
- Benjamin Reeves7
- Alan Appelbe8
- Andrew Cochrane9
- Timothy D Johnson10
- Laura M Korte11
- Ivonia M Do Rosario2
- Inez T Da Silva Almeida2
- Kathryn V Roberts1
- Jonathan R Carapetis10
- Joshua R Francis1,12
- 1 Royal Darwin Hospital, Darwin, NT
- 2 Hospital Nacional Guido Valadares, Dili, Timor-Leste
- 3 Centre for Disease Control, Northern Territory Department of Health, Darwin, NT
- 4 Maluk Timor, Dili, Timor-Leste
- 5 East Timor Hearts Fund, Melbourne, VIC
- 6 St Vincent's Hospital, Melbourne, VIC
- 7 Cairns Hospital, Cairns, QLD
- 8 Geelong Cardiology Group, Barwon Health, Geelong, VIC
- 9 Monash Medical Centre Clayton, Melbourne, VIC
- 10 Telethon Kids Institute, University of Western Australia, Perth, WA
- 11 Paediatric WA Country Health Service – Kimberley, Broome, WA
- 12 Global and Tropical Health Division Menzies School of Health Research, Darwin, NT
This study was funded and supported by the East Timor Hearts Fund (ETHF). Pfizer donated benzathine penicillin for treating children with rheumatic heart disease. Neither the ETHF nor Pfizer had any role in the design, implementation or analysis of the study. The investigation was supported by staff and volunteers from the ETHF (Annette Appelbe, Nicki Mock, Liam Callaghan, Julie Kean, Ken Dusting, Ana Saldanha), St John of God Healthcare Timor-Leste (Benjamin Dingle, Nuno Soares), the Northern Territory Centre for Disease Control (Desley Williams, Christian James), the Telethon Kids Institute (Rosemary Wyber) and the Bairo Pite Clinic. We thank Laura Francis, Jack Francis, Dinorah Caeiro Alves and Patricia “Kuri” Xana Caiero Alves for their contributions. This study would not have been possible without the invaluable assistance of Eddie De Pina and the Bakhita Centre. We also thank the late Andrew Metcalfe of St John of God Healthcare for his contribution.
No relevant disclosures.
- 1. Roberts KV, Maguire GP, Brown A, et al. Rheumatic heart disease in Indigenous children in northern Australia: differences in prevalence and the challenges of screening. Med J Aust 2015; 203: 221. <MJA full text>
- 2. Lawrence JG, Carapetis JR, Griffiths K, et al. Acute rheumatic fever and rheumatic heart disease: incidence and progression in the Northern Territory of Australia, 1997 to 2010. Circulation 2013; 128: 492-501.
- 3. He VY, Condon JR, Ralph AP, et al. Long-term outcomes from acute rheumatic fever and rheumatic heart disease: a data-linkage and survival analysis approach. Circulation 2016; 134: 222-232.
- 4. Milne RJ, Lennon D, Stewart JM, et al. Mortality and hospitalisation costs of rheumatic fever and rheumatic heart disease in New Zealand. J Paediatr Child Health 2012; 48: 692-697.
- 5. Carapetis JR, Steer AC, Mulholland EK, Weber M. The global burden of group A streptococcal diseases. Lancet Infect Dis 2005; 5: 685-694.
- 6. Rothenbuhler M, O’Sullivan CJ, Stortecky S, et al. Active surveillance for rheumatic heart disease in endemic regions: a systematic review and meta-analysis of prevalence among children and adolescents. Lancet Glob Health 2014; 2: e717-e726.
- 7. McDonald MI, Towers RJ, Andrews RM, et al. Low rates of streptococcal pharyngitis and high rates of pyoderma in Australian aboriginal communities where acute rheumatic fever is hyperendemic. Clin Infect Dis 2006; 43: 683-689.
- 8. Manyemba J, Mayosi BM. Penicillin for secondary prevention of rheumatic fever. Cochrane Database Syst Rev 2002; (3): CD002227.
- 9. Remenyi B, Carapetis J, Wyber R, et al; World Heart Federation. Position statement of the World Heart Federation on the prevention and control of rheumatic heart disease. Nat Rev Cardiol 2013; 10: 284-292.
- 10. Alonso A, Brugha R. Rehabilitating the health system after conflict in East Timor: a shift from NGO to government leadership. Health Policy Plan 2006; 21: 206-216.
- 11. Martins N, Hawkins Z. Striving for better health through health research in post-conflict Timor-Leste. Health Res Policy Syst 2012; 10: 13.
- 12. dos Santos MM, Amaral S, Harmen SP, et al. The prevalence of common skin infections in four districts in Timor-Leste: a cross sectional survey. BMC Infect Dis 2010; 10: 61.
- 13. Paratz ED, Bayley N. Heart disease in East Timor: cross-sectional analysis of 474 patients attending Timor-Leste’s first cardiology service. Intern Med J 2017; 47: 423-428.
- 14. Beaton A, Okello E, Lwabi P, et al. Echocardiography screening for rheumatic heart disease in Ugandan schoolchildren. Circulation 2012; 125: 3127-3132.
- 15. Remenyi B, Wilson N, Steer A, et al. World Heart Federation criteria for echocardiographic diagnosis of rheumatic heart disease — an evidence-based guideline. Nat Rev Cardiol 2012; 9: 297-309.
- 16. Yadeta D, Hailu A, Haileamlak A, et al. Prevalence of rheumatic heart disease among school children in Ethiopia: a multisite echocardiography-based screening. Int J Cardiol 2016; 221: 260-263.
- 17. Engel ME, Haileamlak A, Zuhlke L, et al. Prevalence of rheumatic heart disease in 4720 asymptomatic scholars from South Africa and Ethiopia. Heart 2015; 101: 1389-1394.
- 18. Engelman D, Kado JH, Remenyi B, et al. Focused cardiac ultrasound screening for rheumatic heart disease by briefly trained health workers: a study of diagnostic accuracy. Lancet Glob Health 2016; 4: e386-e394.
- 19. Shrestha NR, Karki P, Mahto R, et al. Prevalence of subclinical rheumatic heart disease in Eastern Nepal: a school-based cross-sectional study. JAMA Cardiol 2016; 1: 89-96.
- 20. Ngaide AA, Mbaye A, Kane A, et al. Prevalence of rheumatic heart disease in Senegalese school children: a clinical and echocardiographic screening. Heart Asia 2015; 7: 40-45.
- 21. Allen M, Allen J, Naseri T, et al. A rapid echocardiographic screening protocol for rheumatic heart disease in Samoa: a high prevalence of advanced disease. Cardiol Young 2017; 27: 1599-1605.
- 22. Sims Sanyahumbi A, Sable CA, Beaton A, et al. School and community screening shows Malawi, Africa, to have a high prevalence of latent rheumatic heart disease. Congenit Heart Dis 2016; 11: 615-621.
- 23. Beaton A, Lu JC, Aliku T, et al. The utility of handheld echocardiography for early rheumatic heart disease diagnosis: a field study. Eur Heart J Cardiovasc Imaging 2015; 16: 475-482.
- 24. Nascimento BR, Beaton AZ, Nunes MC, et al. Echocardiographic prevalence of rheumatic heart disease in Brazilian schoolchildren: data from the PROVAR study. Int J Cardiol 2016; 219: 439-445.
- 25. World Health Organization. Rheumatic fever and rheumatic heart disease (Document no. EB141.R11) [recommendation]. June 2017. http://apps.who.int/gb/ebwha/pdf_files/EB141/B141_R1-en.pdf (viewed Oct 2017).
- 26. Jaine R, Baker M, Venugopal K. Acute rheumatic fever associated with household crowding in a developed country. Pediatr Infect Dis J 2011; 30: 315-319.
- 27. Zuhlke L, Engel ME, Karthikeyan G, et al. Characteristics, complications, and gaps in evidence-based interventions in rheumatic heart disease: the Global Rheumatic Heart Disease Registry (the REMEDY study). Eur Heart J 2015; 36: 1115-1122a.
- 28. Timor-Leste Government. Population and housing census: preliminary results. 2015. http://www.statistics.gov.tl/wp-content/uploads/2015/10/1-Preliminary-Results-4-Printing-Company-19102015.pdf (viewed Oct 2017).
- 29. Wayte K, Zwi AB, Belton S, et al. Conflict and development: challenges in responding to sexual and reproductive health needs in Timor-Leste. Reprod Health Matters 2008; 16: 83-92.
- 30. Remond M, Atkinson D, White A, et al. Are minor echocardiographic changes associated with an increased risk of acute rheumatic fever or progression to rheumatic heart disease? Int J Cardiol 2015; 198: 117-122.





Abstract
Objectives: To determine the prevalence of rheumatic heart disease (RHD) in school-aged children and young people in Timor-Leste.
Design: Prospective cross-sectional survey. Echocardiography was performed by Australian cardiologists to determine the presence of RHD. Demographic data were also collected. Patients in whom RHD was detected were entered into a register to allow monitoring of adherence to secondary prophylaxis; the first dose of benzathine penicillin G (BPG) was administered on the day of screening.
Setting: Schools in urban (Dili) and rural (Ermera) Timor-Leste.
Participants: School students aged 5–20 years.
Outcome measures: Definite and borderline RHD, as defined by World Heart Federation echocardiographic criteria.
Results: 1365 participants were screened; their median age was 11 years (IQR, 9–14 years), and 53% were girls. The estimated prevalence of definite RHD was 18.3 cases per 1000 population (95% CI, 12.3–27.0 per 1000), and of definite or borderline RHD 35.2 per 1000 (95% CI, 26.5–46.4 per 1000). Definite (adjusted odds ratio [aOR], 3.5; 95% CI, 1.3–9.4) and definite or borderline RHD (aOR, 2.7; 95% CI, 1.4–5.2) were more prevalent among girls than boys. Eleven children (0.8%) had congenital heart disease. Of the 25 children in whom definite RHD was identified, 21 (84%) received education and a first dose of BPG on the day of screening; all 25 have since received education about primary care for RHD and have commenced penicillin prophylaxis.
Conclusions: The rates of RHD in Timor-Leste are among the highest in the world, and prevalence is higher among girls than boys. Community engagement is essential for ensuring follow-up and the effective delivery of secondary prophylaxis.