Obesity epidemics have developed concurrently with population ageing worldwide. More than 40% of adults who were aged 25–29 years in 2000 will be obese by the time they reach the age of 60–64 years.1 The increasing prevalence of type 2 diabetes has mirrored obesity epidemics. There are about one million people living with type 2 diabetes in Australia, and more than 90% of these individuals are aged 40 years or older.2 Worldwide, the highest age-specific prevalence of any diabetes (19%) is observed in those aged 60–79 years, and this age group will also have the greatest proportional increase in patients with any diabetes by 2035.3
A characteristic of ageing that has been under-investigated as a potential contributor to the risk of type 2 diabetes, and functional deficits common to this condition, is sarcopenia. We performed a non-systematic search of the MEDLINE and Embase databases using search terms including (but not limited to) “sarcopenia”, “muscle mass”, “physical performance”, “diabetes” and “insulin resistance”, with additional review of our personal reference libraries, to identify recent scientific literature investigating the effects of sarcopenia on the risk of type 2 diabetes, the progression of sarcopenia in older adults with existing type 2 diabetes, and potential therapies beneficial for both conditions.
Defining and diagnosing sarcopenia
The term sarcopenia, from the Greek for “poverty of flesh”, was first proposed in 1989 as a descriptor for age-related muscle wasting by Irwin Rosenberg, who commented that “no decline with age is more dramatic or potentially more functionally significant than the decline in lean body mass”.4 Although this decline in muscle mass with age has consequences for health, subsequent research has found that loss of muscle strength during ageing outpaces loss of muscle mass by up to five times,5 and that low muscle strength is more consistently associated with functional decline than low muscle mass.6 Accordingly, experts now describe sarcopenia as a multidimensional condition requiring assessment of muscle mass, muscle strength and physical performance.
The development of clinically relevant operational definitions for sarcopenia and the recent establishment of an International Classification of Diseases, 10th revision, clinical modification (ICD-10-CM) code for the condition7 have provided the first real impetus for clinicians to diagnose sarcopenia in a systematic fashion.8 Box 1 summarises three current operational definitions and their appropriate measurement techniques and thresholds. Although the lack of consensus on a single operational definition of sarcopenia is a barrier to its clinical assessment, the condition can be diagnosed using relatively inexpensive equipment requiring minimal time and expertise. For example, the European Working Group on Sarcopenia in Older People (EWGSOP) defines sarcopenia as the presence of low appendicular lean mass (generally assessed by dual-energy x-ray absorptiometry, but can also be assessed by portable bioelectrical impedance analysis equipment) in addition to low hand grip strength (measured by hydraulic hand grip dynamometer) or gait speed9 over a short (4 m) walkway. The International Working Group on Sarcopenia10 and the Foundation for the National Institutes of Health (FNIH) Biomarkers Consortium Sarcopenia Project11 state that sarcopenia can be assessed using the same equipment, but with different thresholds.
There is justification for sarcopenia case finding in health care. Although sarcopenia prevalence estimates are influenced by the operational definition applied, as many as 30% of community-dwelling older adults may have the condition, depending on demographic characteristics including age and ethnicity.12 Sarcopenia is consistently a predictor of poor quality of life, difficulties with activities of daily living, mobility disability, falls, fractures, institutionalisation and mortality, independent of other comorbidities.8,9 The costs of sarcopenia to health services in Australia are likely to be substantial, given that annual health-related costs for older Dutch adults were about three times higher for individuals with sarcopenia than for those without the condition.13
Sarcopenia in the pathogenesis of type 2 diabetes
The metabolic outcomes of sarcopenia have received less attention than the functional consequences in the research literature, but they are no less relevant clinically. There are several pathways by which age-related changes in skeletal muscle may contribute to insulin resistance (Box 2). Skeletal muscle is the largest insulin-sensitive tissue in the body and accounts for 80% of glucose uptake under euglycaemic hyperinsulinaemic conditions. Skeletal muscle insulin resistance is a key process in the development of type 2 diabetes, which may be observed decades before β-cell failure and hyperglycaemia develop.14 It is likely that significantly lower skeletal muscle mass results in reduced capacity for glucose disposal in older adults with sarcopenia.
In addition to a loss of mass during ageing, muscle undergoes numerous composition changes that are often described as declines in muscle quality. These declines in quality partly explain the faster rate of loss of muscle strength compared with loss of mass, and may also increase the risk of insulin resistance. Ageing skeletal muscle has reduced oxidative capacity, resulting in increased production of reactive oxygen species, which contributes to oxidative mitochondrial DNA mutagenesis and pro-inflammatory processes.15 Both mitochondrial dysfunction and chronic low-grade inflammation are associated with insulin resistance.15 Also, during ageing, there is an increase in infiltration of skeletal muscle by ectopic fat, including intramyocellular lipids (IMCL) and adipocytes located between muscle groups (intermuscular) and between muscle fascicles (intramuscular). Both IMCL and intramuscular and intermuscular adipose tissue (IMAT) have been implicated in insulin resistance.16 Paradoxically, high levels of IMCL are reported in endurance athletes, suggesting that high levels are beneficial for some individuals. IMAT-derived adipocytes may deleteriously affect muscle metabolism and insulin sensitivity through increased local secretion of pro-inflammatory adipokines, and intermuscular fat may also impair insulin action through reducing blood flow to muscle.16
Using peripheral quantitative computed tomography imaging of calf muscles, we have observed that overweight and obese women aged 50–89 years with type 2 diabetes have a 70% larger IMAT cross-sectional area and 4% lower muscle density (indicating higher levels of intramuscular adipose tissue) than women without type 2 diabetes matched by age and body mass index (both P ≤ 0.05, our unpublished data) (Box 3). In the Look AHEAD trial of middle-aged to older adults in the United States, participants with type 2 diabetes had 0.5 kg more IMAT than did controls without diabetes.17 IMAT, but not subcutaneous adipose tissue, is positively correlated with insulin resistance in type 2 diabetes, despite constituting a much smaller proportion of total body fat.18
Leg muscle mass, strength and functional performance are significantly lower in older patients with type 2 diabetes compared with healthy controls,19 but few prospective studies have investigated the risk of incident type 2 diabetes in older adults with sarcopenia. Among obese participants in the English Longitudinal Study of Ageing, there was a more than threefold increased risk of self-reported incident type 2 diabetes over 6 years for those whose baseline hand grip strength was in the sarcopenic range according to the FNIH definition.20 In the Osteoporotic Fractures in Men study, older men in the highest quartile for insulin resistance (among those without type 2 diabetes), defined by the homoeostasis model assessment of insulin resistance, had a twofold increased likelihood of a 5% decline in total lean body mass over almost 5 years.21 An 11-year follow-up of the Health, Aging and Body Composition (Health ABC) Study found a 40–60% decrease in the risk of incident type 2 diabetes among normal-weight women with greater abdominal and thigh muscle area; but greater muscle mass predicted an increased risk in overweight and obese women.22 It is possible that larger IMAT depots in the muscles of obese women explain this controversial finding.
Factors including inflammation, comorbidities and low levels of physical activity also predispose patients with type 2 diabetes to an increased risk of sarcopenia. In the Health ABC Study, thigh muscle size declined twice as fast over 6 years in older women with type 2 diabetes compared with women without diabetes,23 and strength declined by one-third more over 3 years in older patients with type 2 diabetes compared with those without diabetes.24 Patients in the US over the age of 60 years with type 2 diabetes were found to have poorer balance and increased likelihood of falls in the previous 12 months compared with patients without diabetes.25 In a prospective analysis of the Study of Osteoporotic Fractures, older women with insulin-treated type 2 diabetes at baseline had an almost threefold increased risk of falling more than once a year over an average of 7 years, compared with patients without diabetes.26 Conditions common to type 2 diabetes, such as hypoglycaemia, poor vision and peripheral neuropathy, undoubtedly contribute to the increased falls risk in older adults with diabetes, but poor physical function is also clearly important. In a secondary analysis of the North Carolina Established Populations for Epidemiologic Studies of the Elderly and Women’s Health Initiative trials, a one-third higher risk of incident fracture was observed for older women with type 2 diabetes, but this association was mediated by poor physical function.27 Thus, poor muscle function may partly explain why older patients with type 2 diabetes have more fractures than those without diabetes, despite generally having higher bone mineral density.28 Furthermore, the increased mortality risk for normal-weight compared with overweight patients with type 2 diabetes appears to be mediated by their smaller relative muscle size.29
Concurrent therapies for type 2 diabetes and sarcopenia
There is little evidence that common pharmacological therapies for type 2 diabetes are beneficial in preventing or reversing sarcopenia in older adults. On the contrary, metformin, the first-line pharmacological therapy for diabetes, is an AMP-activated protein kinase agonist and may cause autophagic muscle cell death, while insulin stimulates muscle protein synthesis in young but not older adults, suggesting it provides no protection from age-related muscle wasting.30
Lifestyle modification, particularly weight loss, is a key therapeutic component for type 2 diabetes, with modest weight loss (5–10% of bodyweight) contributing to improved glucose control.31 However, weight loss can include declines in muscle mass and may result in undesirable metabolic and functional consequences, particularly in patients with type 2 diabetes and sarcopenia. For this reason, exercise that promotes gains in muscle mass and function should be a component of lifestyle modification for older adults with type 2 diabetes. A 6-month randomised controlled trial of high-intensity progressive resistance training plus moderate weight loss versus moderate weight loss alone in 36 overweight older adults showed threefold greater decreases in glycated haemoglobin levels in the resistance training group.32 Furthermore, this group had significantly higher lean body mass and muscle strength at follow-up compared with the weight loss-alone group, despite similar reductions in fat mass. Similarly, in postmenopausal women with obesity, glucose infusion rates increased significantly after 16 weeks of aerobic plus resistance (involving weight machines) exercise, but not aerobic exercise alone.33
Clearly, resistance training requiring access to large equipment such as weight machines is not feasible in most clinical settings. Nevertheless, exercise programs requiring minimal equipment may improve physical performance in older adults with type 2 diabetes. In the US Lifestyle Interventions and Independence for Elders (LIFE) study, more than 1600 participants aged 70–89 years with poor physical performance were randomly assigned to a structured physical activity or a health education intervention. The exercise group, who completed moderate walking, ankle weights, balance and flexibility exercises, had about 30% reduced risk for 2.5-year mobility disability compared with those receiving health education, and similar benefits were reported for those with and without type 2 diabetes.34 A meta-analysis of resistance band training, which uses inexpensive elastic bands to progressively increase resistance, suggests that this type of training may result in significant improvements in leg strength but not in glycated haemoglobin levels.35 Thus, lower-intensity resistance training programs are likely to be effective in preventing functional decline in older patients with type 2 diabetes, but further research is required to determine whether they can also provide improvements in metabolic health.
An area of recent research focus that is important to the prescription of lifestyle modification programs for older patients with type 2 diabetes is resistance to the beneficial effects of exercise. As many as 15–20% of individuals with type 2 diabetes obtain no improvements in glucose homoeostasis, insulin sensitivity or muscle mitochondrial density after supervised exercise interventions, despite adequate adherence.36 Furthermore, in a study investigating the effects of 5 months of aerobic or resistance training on physical function in overweight and obese women aged 65–79 years, 13%, 30% and 30% showed no improvement in aerobic capacity, knee extension strength and physical performance, respectively.37 It has been hypothesised that poor exercise responsiveness within skeletal muscle occurs as a result of attenuated expression of key fuel metabolism genes, including peroxisome proliferator-activated receptor γ coactivator-1α, peroxisome proliferator-activated receptor β/δ and pyruvate dehydrogenase kinase. Studies investigating regulators of the transcription of these genes may therefore have success in enhancing adaptations to exercise.36
Inflammation, low 25-hydroxyvitamin D (25(OH)D) status and poor muscle quality are all common in people with type 2 diabetes and may contribute to poor exercise responsiveness. Almost 20% of sedentary adults with elevated plasma C-reactive protein (CRP) concentrations have no improvement in fasting insulin levels after an endurance training program.38 We have found that older adults with high baseline levels of both 25(OH)D (≥ 50 nmol/L) and physical activity (≥ 10 000 steps/day) gained 2 kg less body fat over 5 years compared with those who had low 25(OH)D levels but high levels of physical activity, suggesting that adequate 25(OH)D levels enhance the benefits of physical activity for body composition in older adults.39 In support of this, the greatest improvements in physical performance in frail Japanese older adults after 3 months of exercise were observed in those with higher baseline 25(OH)D levels (> 67.5 nmol/L).40 Older women with adequate vitamin D status also demonstrated greater fat oxidation during exercise.41
We have previously proposed that low vitamin D status promotes adipogenesis, leading to increased IMAT deposition.42 Given older adults with high baseline IMAT levels have blunted improvements in muscle function after exercise,43 it is possible that increased IMAT and associated skeletal muscle inflammation is a mechanism through which low vitamin D status contributes to poor exercise responsiveness. Vitamin D supplementation has, to date, shown few benefits for metabolic health and physical function, although studies have been limited by inadequate sample sizes, doses and durations, and by inclusion of vitamin D-replete participants.44 This therapy is only likely to be effective in those who achieve replete 25(OH)D levels from initial low levels. A 12-month weight loss intervention combined with 2000 IU/day of vitamin D showed no effect on body composition compared with placebo.45 However, participants whose 25(OH)D levels reached ≥ 75 nmol/L lost 3 kg more bodyweight and 2% more body fat than did those whose 25(OH)D levels were < 75 nmol/L.
Although Australian guidelines currently recommend dietary protein intakes of 1 g/kg/day for adults aged over 70 years, intakes of 1.2 to 1.6 g/kg/day may be most effective for enhancing exercise-induced muscle gains, and there is no evidence of renal disorders with these intakes.46 Managing weight loss in older patients with type 2 diabetes while increasing the proportion of energy from protein may be best accomplished by reducing carbohydrate intake.47 High protein intakes may also support weight loss by increasing satiety. A 4-month cluster randomised controlled trial of 100 female nursing home residents found that progressive resistance training combined with 1.3 g/kg/day of red meat resulted in greater gains in muscle mass and strength and decreases in fat mass, relative to resistance training alone.48 Muscle protein synthesis in response to protein supplementation in older adults may also be enhanced by adequate vitamin D status. Daily supplementation of 2 g β-hydroxy β-methylbutyrate (a metabolite of leucine), 5 g arginine and 1.5 g lysine for 12 months in older adults resulted in significant improvement in knee extension strength only for those whose baseline 25(OH)D levels were ≥ 75 nmol/L.49 Similarly, in older adults with sarcopenia, exercise plus daily whey protein (22 g), essential amino acids (11 g, including 4 g leucine) and vitamin D (100 IU) resulted in almost 2 kg greater gain in lean mass compared with exercise alone, as well as significant gains in hand grip strength and declines in CRP levels.50 Nevertheless, further research is required to confirm the effects of dietary supplementation in patients with type 2 diabetes and sarcopenia.
Conclusions
The prevalence and socio-economic burden of sarcopenia will increase in Australia in coming years, but sarcopenia presently receives little attention in clinical settings, likely due in large part to a lack of clarity about its definition and assessment. Expert groups have attempted to reduce this confusion by providing clinical guidelines and, while further work is required to achieve a consensus operational definition of sarcopenia, diagnosis can now be easily integrated into clinical practice. The establishment of the ICD-10-CM code will enable improved reporting of the condition.
Through integrating sarcopenia case finding into clinical practice, this previously under-appreciated risk factor for type 2 diabetes in older adults can be systematically monitored, and lifestyle modification for primary and secondary prevention much better targeted. The evidence presented here shows that older adults with sarcopenia are at risk of developing type 2 diabetes, and those with prevalent type 2 diabetes show an accelerated loss of muscle mass and function that may increase the risk of further metabolic and functional declines. Interventions that reverse or halt progression of sarcopenia in patients with type 2 diabetes are likely to have important health benefits, given that the evidence suggests poor muscle mass and function substantially mediate associations of type 2 diabetes with incident fractures and mortality.
Including progressive resistance training in lifestyle modification programs should be considered for older patients with sarcopenia, type 2 diabetes or both. Clinicians need to be cognisant that individual responses to exercise vary considerably in patients with type 2 diabetes, and beneficial metabolic and functional outcomes are more likely to be obtained when adherence and responsiveness to the therapy are closely monitored, as with pharmacotherapy. Exercise programs should also be regularly adapted to support ongoing improvements in muscle mass and function. Ensuring adequate vitamin D status and maintenance of dietary protein intakes during energy restriction may optimise the effects of exercise interventions targeting type 2 diabetes and sarcopenia in older adults, thereby delaying onset of morbidity and loss of independence related to both conditions.
Box 1 – Suggested measurement techniques and thresholds for components of sarcopenia according to current consensus definitions
Component |
Thresholds |
Method and equipment |
|||||||||||||
European Working Group on Sarcopenia in Older People9 | |||||||||||||||
Low muscle mass |
Appendicular lean mass adjusted for height (m2):Men: < 7.26 kg/m2Women: < 5.50 kg/m2 |
Whole-body DXA and stadiometer |
|||||||||||||
Skeletal muscle mass adjusted for height (m2):Men: < 8.87 kg/m2Women: < 6.42 kg/m2 |
BIA and stadiometer |
||||||||||||||
Low muscle strength |
Hand grip strength:Men: < 30 kgWomen: < 20 kg |
Hydraulic hand grip dynamometer |
|||||||||||||
Poor physical performance |
Gait speed: ≤ 0.8 m/s |
4 m walkway and stop watch |
|||||||||||||
International Working Group on Sarcopenia10 | |||||||||||||||
Low muscle mass |
Appendicular lean mass adjusted for height (m2):Men: ≤ 7.23 kg/m2Women: ≤ 5.67 kg/m2 |
Whole-body DXA and stadiometer |
|||||||||||||
Poor physical performance |
Gait speed: < 1.00 m/s |
4 m walkway and stop watch |
|||||||||||||
Foundation for the National Institutes of Health Biomarkers Consortium Sarcopenia Project11 | |||||||||||||||
Low muscle mass |
Appendicular lean mass adjusted for BMI (kg/m2):Men: < 0.789Women: < 0.512 |
Whole-body DXA, stadiometer and weight scales |
|||||||||||||
Low muscle strength |
Hand grip strength:Men: < 26 kgWomen: < 16 kg |
Hydraulic hand grip dynamometer |
|||||||||||||
BIA = bioelectrical impedance analysis. BMI = body mass index. DXA = dual-energy x-ray absorptiometry. | |||||||||||||||
Box 2 – Potential pathways by which sarcopenia contributes to insulin resistance in ageing*
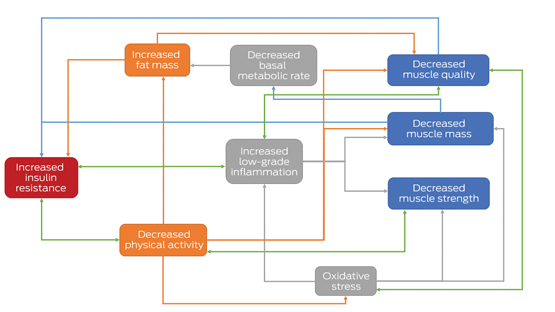
* Components of sarcopenia are shown in the blue boxes. Green arrows indicate possible bidirectional relationships, illustrating mechanisms by which sarcopenia may be accelerated in people with type 2 diabetes.
Box 3 – Transverse peripheral quantitative computed tomography images of the mid-calf highlighting IMAT in age- and BMI-matched obese older women (A) without and (B) with type 2 diabetes*
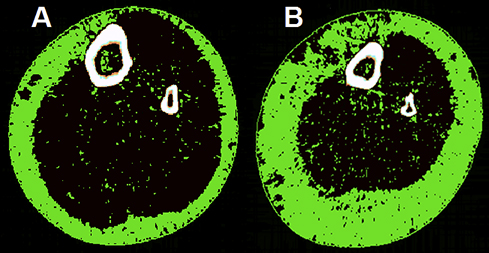
BMI = body mass index. IMAT = intramuscular and intermuscular adipose tissue. * IMAT is indicated by the green pixels located in the black muscle compartment. Both women had a BMI of 35 kg/m2 and were aged 75 years, but the woman with type 2 diabetes (B) had greater subcutaneous fat, a smaller muscle cross-sectional area (649 v 752 cm) and twice as much IMAT (28.5 v 14 cm) as the woman without type 2 diabetes (A). The woman with type 2 diabetes also had poor muscle function, meeting the European Working Group on Sarcopenia in Older People definition of sarcopenia (gait speed ≤ 0.8 m/s and hand grip strength < 20 kg).
Provenance: <p>Not commissioned; externally peer reviewed.</p>
- David Scott1
- Barbora de Courten2,3
- Peter R Ebeling1
- 1 Monash University, Melbourne, VIC
- 2 Monash Centre for Health Research and Implementation, Melbourne, VIC
- 3 Monash Medical Centre, Melbourne, VIC
Barbora de Courten is supported by the Royal Australasian College of Physicians, the National Health and Medical Research Council and a National Heart Foundation Future Leader Fellowship (100864).
No relevant disclosures.
- 1. Walls HL, Magliano DJ, Stevenson CE, et al. Projected progression of the prevalence of obesity in Australia. Obesity (Silver Spring) 2012; 20: 872-878.
- 2. Australian Institute of Health and Welfare. Australia’s health 2014. Canberra: AIHW, 2014.
- 3. Guariguata L, Whiting DR, Hambleton I, et al. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract 2014; 103: 137-149.
- 4. Rosenberg IH. Summary comments. Am J Clin Nutr 1989; 50: 1231-1233.
- 5. Hughes VA, Frontera WR, Wood M, et al. Longitudinal muscle strength changes in older adults: influence of muscle mass, physical activity, and health. J Gerontol A Biol Sci Med Sci 2001; 56: B209-B217.
- 6. Visser M, Schaap LA. Consequences of sarcopenia. Clin Geriatr Med 2011; 27: 387-399.
- 7. Lloyd N. AIM Coalition announces establishment of ICD-10-CM code for sarcopenia by the Centers for Disease Control and Prevention [media release]. Washington, DC: Aging in Motion Coalition, 28 Apr 2016. http://aginginmotion.org/news/2388-2 (accessed June 2016).
- 8. Landi F, Calvani R, Cesari M, et al. Sarcopenia as the biological substrate of physical frailty. Clin Geriatr Med 2015; 31: 367-374.
- 9. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010; 39: 412-423.
- 10. Fielding RA, Vellas B, Evans WJ, et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International Working Group on Sarcopenia. J Am Med Dir Assoc 2011; 12: 249-256.
- 11. Studenski SA, Peters KW, Alley DE, et al. The FNIH Sarcopenia Project: rationale, study description, conference recommendations, and final estimates. J Gerontol A Biol Sci Med Sci 2014; 69: 547-558.
- 12. Cruz-Jentoft AJ, Landi F, Schneider SM, et al. Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 2014; 43: 748-759.
- 13. Mijnarends DM, Schols JMGA, Halfens RJG, et al. Burden-of-illness of Dutch community-dwelling older adults with sarcopenia: health related outcomes and costs. Eur Geriatr Med 2016; 7: 276-284.
- 14. DeFronzo RA, Tripathy D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care 2009; 32 Suppl 2: S157-S163.
- 15. Kim JA, Wei Y, Sowers JR. Role of mitochondrial dysfunction in insulin resistance. Circ Res 2008; 102: 401-414.
- 16. Miljkovic I, Kuipers AL, Cauley JA, et al. Greater skeletal muscle fat infiltration is associated with higher all-cause and cardiovascular mortality in older men. J Gerontol A Biol Sci Med Sci 2015; 70: 1133-1140.
- 17. Gallagher D, Kelley DE, Yim J-E, et al. Adipose tissue distribution is different in type 2 diabetes. Am J Clin Nutr 2009; 89: 807-814.
- 18. Goodpaster BH, Thaete FL, Kelley DE. Thigh adipose tissue distribution is associated with insulin resistance in obesity and in type 2 diabetes mellitus. Am J Clin Nutr 2000; 71: 885-892.
- 19. Leenders M, Verdijk LB, van der Hoeven L, et al. Patients with type 2 diabetes show a greater decline in muscle mass, muscle strength, and functional capacity with aging. J Am Med Dir Assoc 2013; 14: 585-592.
- 20. Cuthbertson DJ, Bell JA, Ng SY, et al. Dynapenic obesity and the risk of incident type 2 diabetes: the English Longitudinal Study of Ageing. Diabet Med 2016; 33: 1052-1059.
- 21. Lee CG, Boyko EJ, Strotmeyer ES, et al. Association between insulin resistance and lean mass loss and fat mass gain in older men without diabetes mellitus. J Am Geriatr Soc 2011; 59: 1217-1224.
- 22. Larsen BA, Wassel CL, Kritchevsky SB, et al. Association of muscle mass, area, and strength with incident diabetes in older adults: the Health ABC Study. J Clin Endocrinol Metab 2016; 101: 1847-1855.
- 23. Park SW, Goodpaster BH, Lee JS, et al. Excessive loss of skeletal muscle mass in older adults with type 2 diabetes. Diabetes Care 2009; 32: 1993-1997.
- 24. Park SW, Goodpaster BH, Strotmeyer ES, et al. Accelerated loss of skeletal muscle strength in older adults with type 2 diabetes. Diabetes Care 2007; 30: 1507-1512.
- 25. Gregg EW, Beckles G, Williamson DF, et al. Diabetes and physical disability among older US adults. Diabetes Care 2000; 23: 1272-1277.
- 26. Schwartz AV, Hillier TA, Sellmeyer DE, et al. Older women with diabetes have a higher risk of falls: a prospective study. Diabetes Care 2002; 25: 1749-1754.
- 27. Lee RH, Pieper CF, Colón-Emeric C. Functional impairments mediate association between clinical fracture risk and type 2 diabetes mellitus in older women. J Am Geriatr Soc 2015; 63: 1546-1551.
- 28. Vestergaard P. Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes — a meta-analysis. Osteoporos Int 2007; 18: 427-444.
- 29. Murphy RA, Reinders I, Garcia ME, et al. Adipose tissue, muscle, and function: potential mediators of associations between body weight and mortality in older adults with type 2 diabetes. Diabetes Care 2014; 37: 3213-3219.
- 30. Cetrone M, Mele A, Tricarico D. Effects of the antidiabetic drugs on the age-related atrophy and sarcopenia associated with diabetes type II. Curr Diabetes Rev 2014; 10: 231-237.
- 31. Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes: a patient-centered approach position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2012; 35: 1364-1379.
- 32. Dunstan DW, Daly RM, Owen N, et al. High-intensity resistance training improves glycemic control in older patients with type 2 diabetes. Diabetes Care 2002; 25: 1729-1736.
- 33. Cuff DJ, Meneilly GS, Martin A, et al. Effective exercise modality to reduce insulin resistance in women with type 2 diabetes. Diabetes Care 2003; 26: 2977-2982.
- 34. Pahor M, Guralnik JM, Ambrosius WT, et al. Effect of structured physical activity on prevention of major mobility disability in older adults: the LIFE study randomized clinical trial. JAMA 2014; 311: 2387-2396.
- 35. McGinley SK, Armstrong MJ, Boulé NG, et al. Effects of exercise training using resistance bands on glycaemic control and strength in type 2 diabetes mellitus: a meta-analysis of randomised controlled trials. Acta Diabetol 2015; 52: 221-230.
- 36. Stephens NA, Sparks LM. Resistance to the beneficial effects of exercise in type 2 diabetes: are some individuals programmed to fail? J Clin Endocrinol Metab 2015; 100: 43-52.
- 37. Chmelo EA, Crotts CI, Newman JC, et al. Heterogeneity of physical function responses to exercise training in older adults. J Am Geriatr Soc 2015; 63: 462-469.
- 38. Bouchard C, Blair SN, Church TS, et al. Adverse metabolic response to regular exercise: is it a rare or common occurrence? PLoS ONE 2012; 7: e37887.
- 39. Scott D, Ebeling PR, Sanders KM, et al. Vitamin D and physical activity status: associations with five-year changes in body composition and muscle function in community-dwelling older adults. J Clin Endocrinol Metab 2015; 100: 670-678.
- 40. Okuno J, Tomura S, Yabushita N, et al. Effects of serum 25-hydroxyvitamin D3 levels on physical fitness in community-dwelling frail women. Arch Gerontol Geriatr 2010; 50: 121-126.
- 41. Ellis AC, Alvarez JA, Gower BA, et al. Cardiorespiratory fitness in older adult women: relationships with serum 25-hydroxyvitamin D. Endocrine 2014; 47: 839-844.
- 42. Scott D, Sanders KM, Ebeling PR. Vitamin D, Muscle function, and falls in older adults: does reduced deposition of intramuscular adipose tissue influence the relationship? J Clin Endocrinol Metab 2013; 98: 3968-3970.
- 43. Marcus R, Addison O, LaStayo P. Intramuscular adipose tissue attenuates gains in muscle quality in older adults at high risk for falling. A brief report. J Nutr Health Aging 2013; 17: 215-218.
- 44. Wamberg L, Pedersen S, Rejnmark L, et al. Causes of vitamin D deficiency and effect of vitamin D supplementation on metabolic complications in obesity: a review. Curr Obes Rep 2015; 4: 429-440.
- 45. Mason C, Xiao L, Imayama I, et al. Vitamin D3 supplementation during weight loss: a double-blind randomized controlled trial. Am J Clin Nutr 2014; 99: 1015-1025.
- 46. Phillips SM, Chevalier S, Leidy HJ. Protein “requirements” beyond the RDA: implications for optimizing health. Appl Physiol Nutr Metab 2016; 41: 565-572.
- 47. Halton TL, Liu S, Manson JE, et al. Low-carbohydrate-diet score and risk of type 2 diabetes in women. Am J Clin Nutr 2008; 87: 339-346.
- 48. Daly RM, O’Connell SL, Mundell NL, et al. Protein-enriched diet, with the use of lean red meat, combined with progressive resistance training enhances lean tissue mass and muscle strength and reduces circulating IL-6 concentrations in elderly women: a cluster randomized controlled trial. Am J Clin Nutr 2014; 99: 899-910.
- 49. Fuller JC Jr, Baier S, Flakoll P, et al. Vitamin D status affects strength gains in older adults supplemented with a combination of β-hydroxy-β-methylbutyrate, arginine, and lysine: a cohort study. JPEN J Parenter Enteral Nutr 2011; 35: 757-762.
- 50. Rondanelli M, Klersy C, Terracol G, et al. Whey protein, amino acids, and vitamin D supplementation with physical activity increases fat-free mass and strength, functionality, and quality of life and decreases inflammation in sarcopenic elderly. Am J Clin Nutr 2016; 103: 830-840.





Summary