The known The human T-lymphotropic virus type 1 (HTLV-1) is endemic to central Australia according to hospital and laboratory data for Indigenous adults admitted to Alice Springs Hospital. However, the data may underestimate the overall community prevalence of HTLV-1 infection in remote communities.
The new The prevalence of HTLV-1 infection in a remote Northern Territory community was high: 30 of 74 adults tested were HTLV-1-positive, and nine had clinical syndromes potentially attributable to HTLV-1 infection.
The implications HTLV-1 infection may be more prevalent among Indigenous Australians and be associated with a greater burden of clinical disease than is currently appreciated.
The human T-lymphotropic virus type 1 (HTLV-1) is an oncogenic retrovirus that preferentially infects CD4+ T-cells.1 Worldwide, at least 5–10 million people are infected with HTLV-1, most dwelling in areas of high endemicity in southern Japan, the Caribbean basin, South America or intertropical Africa.2 Transmission typically follows exposure to infected lymphocytes in blood, or through breastfeeding or sexual intercourse. A minority of people infected with HTLV-1 experience a rapidly progressive haematological malignancy (adult T-cell leukaemia/lymphoma [ATLL])3 or inflammatory disorders4 such as HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP)5 and HTLV-1-associated pulmonary disease.4,6 Although the lifetime disease-specific risks for HAM/TSP and ATLL in Japan and the Caribbean are low (0.3–1.9%7,8 and 1–5%1 respectively), the true burden of HTLV-1-associated diseases has not been determined in a community setting.
HTLV-1 was first identified in central Australia in 19889 and each of the major recognised complications of HTLV-1 have since been described in Indigenous residents of this region.3,5,10,11 HTLV-1-associated pulmonary disease is particularly common6,10,12 and contributes to the highest reported adult prevalence of bronchiectasis worldwide.6 An impaired immune response also contributes to HTLV-1-associated morbidity by increasing the larval burden of Strongyloides stercoralis in people infected with HTLV-1 living in resource-poor areas.1,10 Notwithstanding seropositivity rates that exceed 30% for Indigenous adults admitted to Alice Springs Hospital,10 there has been no coordinated program to reduce viral transmission among Indigenous Australians.
The development of strategies for controlling HTLV-1 transmission in Australia is hampered by limitations of the epidemiological data. Indeed, although the HTLV-1c subtype is thought to be endemic to central Australia, this conclusion is based on laboratory data for Indigenous adults admitted to Alice Springs Hospital,2 and these data may substantially underestimate the prevalence of HTLV-1 infections in some Indigenous communities. Conversely, only two studies have reported community-based seropositivity rates, for 3613 and 1319 Indigenous people from undefined populations. In endemic areas, such as southwestern Japan, mother-to-child transmission is thought to be the primary mode of transmission,14 and this is also assumed to be the case in central Australia.15 Nevertheless, HTLV-1 testing is not currently included in routine antenatal screening.16 Modes of transmission other than breastfeeding may be important for Indigenous Australians, among whom infection rates are highest for hospitalised men.10
Community-based studies are essential for defining the epidemiological associations of HTLV-1 infection in Australia and for accurately determining its prevalence. Such work is complicated by the historical burden of mistrust associated with research in Indigenous communities.17 Here we report the results of a community-based survey of HTLV-1 infection using a culturally safe model that integrated clinical research with a health literacy program; it is the first such community-based survey to be reported. In addition to determining HTLV-1 seroprevalence and its clinical associations, we quantified the number of infected peripheral blood cells with a recently developed HTLV-1c proviral load (PVL) test. A higher HTLV-1 PVL is associated with ATLL and HAM/TSP,1 but data on HTLV-1c PVL are limited, and have not previously been reported for a community-based survey.
Methods
A single remote Northern Territory Indigenous community (estimated resident population [2015], 138) was selected for our pilot study, conducted during two visits, 25–29 August 2014 and 15–19 June 2015.
Phase 1: community engagement
A knowledge translation process and health literacy resources were developed during visits to the community by an Aboriginal research officer (CP) and a non-Indigenous academic (KT). These resources were used by a clinician (LE) during two subsequent visits for discussing (through an interpreter, CP) the major disease associations and the risk of HTLV-1 transmission by sexual contact or exposure to infected blood. Given current uncertainties about the predominant mode of transmission and the clear benefit of breastfeeding in remote Indigenous communities, mother-to-child transmission was not discussed. Health messages were provided separately to men and women.
Phase 2: clinical survey
Male and female Aboriginal team members again provided health literacy information. Individuals were then invited to participate in the study, and consent was obtained in their primary language. The clinical survey incorporated a health questionnaire and a limited physical examination. Data collected included self-reported comorbid conditions and respiratory, gastrointestinal, dermatological and neurological symptoms. All surveys were performed while blinded to the HTLV-1 serostatus of the participants. Clinical records were subsequently reviewed to confirm comorbidity data. Physical examination was restricted to a respiratory examination and limited neurological (gait and lower limb tone, power, reflexes and sensation) and dermatological examinations (generally restricted to the head, back, limbs and abdomen).
Whole blood samples were collected into EDTA-coated tubes. Samples were processed at Alice Springs Hospital and forwarded for Strongyloides serological testing (Western Diagnostic Pathology, Perth). Peripheral blood buffy coats were recovered and stored at −70°C for HTLV-1 studies. Blood was not collected from children under 2 years of age.
Community residence was determined from the community health clinic registry. Chronic lung disease was defined as a daily productive cough (lasting at least one month) with inspiratory crackles on auscultation of the chest. Diarrhoea was defined as the passing of several loose stools each day for more than 2 days.
HTLV-1 serologic and molecular studies
Analyses were performed at the National Serology Reference Laboratory, Melbourne, blinded to the clinical state of participants. Plasma HTLV-1 antibodies were detected by enzyme immunoassay (Murex HTLV I + II, DiaSorin) and titres determined with a twofold endpoint dilution method by particle agglutination (Serodia-HTLV-1, Fujirebio). All samples reactive on either screening assay were confirmed by western blot (WB) (HTLV-I/II Blot2.4, MP Diagnostics). HTLV-1 infection was defined by a positive WB test result.
HTLV-1 PVL was determined by polymerase chain reaction (PCR) using primers and probes that targeted a highly conserved region at the 5′ end of the gag gene in the p19 coding region of HTLV-1c (Mel5; accession number, L02534). SP cells18 containing a single integrated, full-length copy of HTLV-1 and one copy of the albumin gene were used to generate a standard curve for determining HTLV-1 copy numbers and cell numbers. The number of HTLV-1 copies per peripheral blood leucocyte (PBL) was then calculated. PVL was expressed as HTLV-1 copies/105 PBL. The lower limit of detection was 6.5 copies for HTLV-1 (95% confidence interval [CI], 5.4–8.4) and 15.6 for albumin (95% CI, 12.9–20.0).
Statistical analysis
Categorical variables were compared in χ2 or Fisher exact tests as appropriate. Continuous variables were assessed for significant departures from normality. Normally distributed variables were summarised as means and standard deviations, and compared in t tests. Variables with skewed distributions were summarised as medians and interquartile ranges (IQRs), and compared in Wilcoxon rank-sum tests. All analyses were performed in Stata 14.0 (StataCorp).
Ethics approval
The study was developed in collaboration with Primary Health Care Remote, Central Australia Health Service, Alice Springs. All participants with a clinical condition were referred for appropriate follow-up and treatment. The study was approved by the Central Australian Human Research Ethics Committee (reference, HREC-14-242).
Results
One hundred and four people (75% of the estimated resident population) consented to participate in the study. Blood was not collected from four children, one sample was lost in transit, and a 68-year-old man with chronic lung disease and ataxia declined to give blood. HTLV-1 test results were discordant (PCR-positive, WB-negative) for an 11-year-old boy with crusted scabies whose mother and maternal grandmother were infected with HTLV-1, and he was excluded from the study. The final analysis therefore included 97 participants (70% of the resident population): 23 children (13 boys, 10 girls) and 74 adults (39 men, 35 women).
HTLV-1 seropositivity
HTLV-1 seropositivity rates were significantly higher among adults than among children (30 of 74 v 1 of 23; P = 0.001) (Box 1). Rates were highest among adults aged 35 years or more; ten of the 15 men in this age group and seven of the 16 women were infected with HTLV-1 (Box 1).
Clinical associations
Four adult participants did not wait for clinical review; data are therefore presented for 27 adults infected with HTLV-1 and 43 who were not (Box 2). Although half of all adults reported respiratory symptoms, with eight exceptions these were all acute conditions, without clinical evidence of chronic lung disease. A chronic productive cough with physical signs was found in seven adults (four men, three women), each of whom was HTLV-1-positive (Box 2). Four had never smoked, two were current smokers, and one had a past history of smoking. Bronchiectasis was radiologically confirmed in one 52-year-old man and in two women aged 36 and 54 years by chest high resolution computed tomography (cHRCT); the other four HTLV-1-positive adults with chronic lung disease were not examined with cHRCT. A chronic productive cough with physical signs was found in only one (HTLV-1-seronegative) child; bronchiectasis was not apparent on cHRCT, and a diagnosis was made of chronic suppurative lung disease with reactive airway of uncertain aetiology.
The gait of three adults (two women aged 54 and 71 years and a 48-year-old man) was ataxic. All were HTLV-1-positive and had chronic lung disease. The two women were unable to walk without walking aids. Lower limb power and sensation were normal, and there was no increased muscular tone or hyperreflexia.
All five participants who reported diarrhoea were HTLV-1-positive (Box 2); serological results for Strongyloides were positive in two instances. Rates of Strongyloides seropositivity were not significantly different for HTLV-1-positive and -negative adults (HTLV-1-positive, 6 of 28; HTLV-1-negative, 4 of 44; P = 0.172). Three of 15 children tested, including the child with chronic lung disease, were seropositive for Strongyloides and HTLV-1-negative. No participant presented with infective dermatitis, and the only one with crusted scabies was the 11-year-old boy described above.
HTLV-1 proviral load
The HTLV-1 PVL for one WB-positive participant could not be determined for technical reasons. The median HTLV-1 PVL for the remaining 30 HTLV-1-positive participants was 145 (IQR, 16.3–558) copies/105 PBL. The results for three HTLV-1-positive participants who did not wait for clinical examination and for two with symptomatic strongyloidiasis were excluded from further analysis. There was no sex difference in the median HTLV-1 PVL for nine men and eight women who did not present with chronic lung disease, ataxia or strongyloidiasis (men, 51 [IQR, 39–271]) copies/105 PBL; women, 8.6 [IQR, 0.27–179]) copies/105 PBL; P = 0.180). The median HTLV-1 PVL was significantly higher for the seven adults with chronic lung disease than for 17 asymptomatic participants (chronic lung disease, 649 copies/105 PBL [IQR, 162–2220]; asymptomatic adults, 40 copies/105 PBL [IQR, 0.9–229]; P = 0.017) (Box 3). HTLV-1 PVL for each of the three participants with both chronic lung disease and gait ataxia exceeded 1000 copies/105 PBL (1365, 2214 and 3399 copies/105 PBL).
Discussion
We found high rates of HTLV-1 infection among Indigenous adults in a remote Indigenous Australian community. The adult seroprevalence rate in this community (30 of 74 people) is comparable with that of some villages in southwestern Japan prior to public health interventions in that country.19 In contrast to the Japanese results for children, only a single Indigenous child was definitely infected with HTLV-1, and we were unable to test his mother or siblings, as they were absent from the community during the survey. Horizontal transmission may therefore be particularly important in central Australia, as in Jamaica20 and some African countries21 where sexual transmission is the predominant mode. Although our data were derived from a single, small community, HTLV-1 serostatus was determined for 70% of its residents, and the adult seroprevalence rate was very close to that for hospitalised adults from this region.10 Published community-based epidemiological data for HTLV-1 infection in Australia are otherwise limited. HTLV-1 seropositivity rates of 14–15% have been reported for samples of 1319 and 3613 Indigenous residents of central Australia, but the manner in which the participants were selected and the size of the populations from which they were drawn is unclear.
The high rate of HTLV-1 infection in our study community was associated with possibly HTLV-1-linked conditions in nine of 30 HTLV-1-positive adults (chronic lung disease, seven participants; symptomatic strongyloidiasis, two). Seven HTLV-1-positive adults satisfied the case definition for chronic lung disease, and in three instances bronchiectasis was radiologically confirmed. In central Australia, HTLV-1 infection increases the rates of hospitalisation for bronchiectasis, lower respiratory tract infections and asthma;10 in a case–control study, the risk of bronchiectasis was almost twice as high for HTLV-1-infected Indigenous Australians as for uninfected residents.12
Bronchiectasis caused by HTLV-1 is associated with a higher HTLV-1c PVL, and in our study this was also true for participants with chronic lung disease. We have previously reported that HTLV-1 PVL is correlated with the extent of radiologically defined pulmonary injury.10 These observations are consistent with the proposed immunopathology of HTLV-1-linked inflammatory diseases, which are thought to result from an immune response to a high HTLV-1 antigen load.22 In a Japanese cohort, an HTLV-1 PVL of greater than 1000 copies/105 peripheral blood mononuclear cells was predictive of HAM/TSP, and half of all those with this condition had abnormal chest x-rays.23 Gait disturbance is a common manifestation of HTLV-1-associated neurological disease.4 In our study, concurrent gait ataxia and chronic lung disease affected three participants whose HTLV-1 PVL exceeded 1000 copies/105 PBL, and this may reflect the systemic nature of the HTLV-1-mediated inflammatory process.4
The burden of HTLV-1-associated inflammatory diseases has not previously been studied in a community setting. The reported lifetime risk of strictly defined HAM/TSP in people who are HTLV-1-positive ranges between 0.25% for cases solicited from Japanese medical institutions7 to 1.9% based on data provided voluntarily by medical staff to registries in the Caribbean.8 In contrast, neurological and dermatological abnormalities respectively affect more than 30%24 and 70%25 of infected persons followed up in Brazilian outpatient clinics. The true burden of HTLV-1-associated inflammatory disease in a community setting may therefore be far higher than suggested by a strict application of the HAM/TSP case definition to hospitalised patients. Skin diseases were less common among HTLV-1-infected participants in our study than in Brazil.25 This may reflect selection bias in the hospital-based Brazilian cohort, but genetic factors may also contribute to the risk of HTLV-1-induced inflammation in particular organ systems.4
Notwithstanding the success with which we were able to engage with residents of the remote Indigenous community, the study design had a number of limitations. For example, the small sample size precluded a detailed epidemiological analysis that could identify the major modes of HTLV-1 transmission. We were also unable to develop a multivariable model that included other factors that contribute to chronic lung disease, such as smoking and age. A further limitation was our inability to determine the pathological basis for most cases of chronic lung disease and gait ataxia; this would require investigations that are unavailable in a remote community. Similarly, strongyloidiasis was diagnosed serologically because we were unable to collect stool samples prior to treatment. Nevertheless, even if all 18 adult residents not recruited to our study were not infected with HTLV-1, the adult HTLV-1 seroprevalence rate in this community would exceed 30%. Moreover, clinical examinations and HTLV-1 studies were performed without knowledge of HTLV-1 serostatus, and our findings are consistent with studies that have found a strong association between HTLV-1 infection and chronic lung disease in Indigenous Australians.6,10,12
In summary, we found very high HTLV-1 seropositivity rates among adult residents of a remote Indigenous community, and evidence for disease potentially attributable to HTLV-1 in nearly one-third of HTLV-1-positive participants. The project integrated clinical research with a health literacy program, using both Indigenous and non-Indigenous expertise to decolonise research practice.26 Further investigations will apply the lessons learned from this pilot project to other communities, in order to identify the major modes of HTLV-1 transmission and the associated disease burden in remote Australia.
Box 1 – Prevalence of HTLV-1 infection among 97 Indigenous Australian residents of a remote Northern Territory community, according to age group
Age category |
Male |
Female |
|||||||||||||
Children (1–14 years) |
1 of 13 (7%) |
0 of 10 |
|||||||||||||
Adults (15–34 years) |
7 of 24 (29%) |
6 of 19 (32%) |
|||||||||||||
Adults (≥ 35 years) |
10 of 15 (67%) |
7 of 16 (44%) |
|||||||||||||
Box 2 – Demographic data for 97 Indigenous Australian residents of a remote Northern Territory community, according to HTLV-1 serostatus
HTLV-1-positive |
HTLV-1-negative |
P |
|||||||||||||
All participants |
|||||||||||||||
Number |
31 |
66 |
|||||||||||||
Age |
0.001 |
||||||||||||||
Children |
1 |
22 |
|||||||||||||
Adults |
30 |
44 |
|||||||||||||
Sex |
|||||||||||||||
Children |
1 boy |
12 boys, 10 girls |
0.37 |
||||||||||||
Adults |
17 men, 13 women |
22 men, 22 women |
0.57 |
||||||||||||
Clinical survey findings (adults only) |
|||||||||||||||
Any history of smoking |
13/30 |
10/44 |
0.06 |
||||||||||||
Comorbidities |
|||||||||||||||
Diabetes |
12/30 |
8/44 |
0.038 |
||||||||||||
Asthma |
1/30 |
1/44 |
0.78 |
||||||||||||
Heart disease |
2/30 |
0/44 |
0.083 |
||||||||||||
Chronic liver disease |
0/30 |
1/44 |
0.41 |
||||||||||||
Chronic kidney disease |
6/30 |
4/44 |
0.18 |
||||||||||||
Symptoms* |
|||||||||||||||
Respiratory† |
21/27 |
25/43 |
0.090 |
||||||||||||
Diarrhoea‡ |
5/27 |
0/43 |
0.007 |
||||||||||||
Dermatological§ |
5/27 |
3/43 |
0.25 |
||||||||||||
Possibly HTLV-1-associated conditions* | |||||||||||||||
Chronic lung disease¶ |
7/27 |
0/43 |
0.001 |
||||||||||||
Ataxic gait |
3/27 |
0/43 |
0.032 |
||||||||||||
Strongyloides seropositivity** |
6/28 |
4/44 |
0.17 |
||||||||||||
Symptomatic strongyloidiasis†† |
2/27 |
0/43 |
0.082 |
||||||||||||
* Three HTLV-1-positive participants and one HTLV-1-negative participant provided blood but did not wait for medical review. † Wheeze, dry cough or dyspnoea. ‡ Loose bowel actions several times per day for longer than 2 days. § Pruritus or rash attributed to scabies (three participants), impetigo (one), tinea corporis (one) or pityriasis versicolor (one). ¶ Daily productive cough with inspiratory crackles audible on auscultation of the chest. ** Strongyloides serological results were not available for all participants. †† Loose bowel actions several times per day for more than 2 days, together with positive Strongyloides serological result. | |||||||||||||||
Box 3 – HTLV-1 proviral load (PVL) in 24 HTLV-1-positive adults*
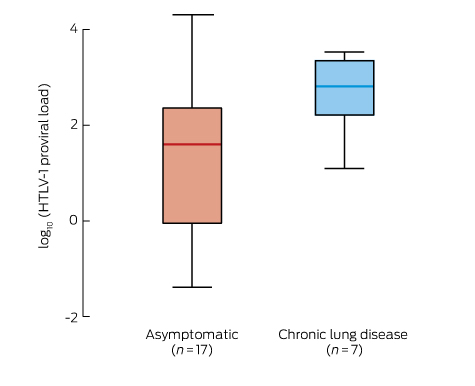
* Expressed as log10(HTLV-1 copies/105 peripheral blood leucocytes). Asymptomatic v chronic lung disease: P = 0.17 (Wilcoxon rank-sum test). Excluded from the analysis were three HTLV-1-positive participants who did not wait for clinical examination, two with symptomatic strongyloidiasis, and one for whom HTLV-1 PVL could not be determined for technical reasons.
Received 9 March 2016, accepted 28 June 2016
- Lloyd J Einsiedel1,2
- Hai Pham2
- Richard J Woodman3
- Clinton Pepperill2
- Kerry A Taylor4
- 1 Northern Territory Rural Clinical School, Flinders University, Alice Springs, NT
- 2 Baker IDI Heart and Diabetes Institute, Alice Springs, NT
- 3 Flinders Centre for Epidemiology and Biostatistics, Flinders University, Adelaide, SA
- 4 Poche Centre for Indigenous Health, Flinders University, Alice Springs, NT
This study was funded by the National Health and Medical Research Council (project grant 1088517) and the Northern Territory Rural Clinical School, which is an initiative of the Australian Department of Health and Ageing.
No relevant disclosures.
- 1. Verdonck K, Gonzalez E, Van Dooren S, et al. Human T-lymphotropic virus 1: recent knowledge about an ancient infection. Lancet Infect Dis 2007; 7: 266-281.
- 2. Gessain A, Cassar O. Epidemiological aspects and world distribution of HTLV-1 infection. Front Microbiol 2012; 3: 388.
- 3. Einsiedel L, Cassar O, Bardy P, et al. Variant human T-cell lymphotropic virus type 1c and adult T-cell leukemia, Australia. Emerg Infect Dis 2013; 19: 1639-1641.
- 4. Martin F, Taylor G, Jacobson S. Inflammatory manifestations of HTLV-1 and their therapeutic options. Expert Rev Clin Immunol 2014; 10: 1531-1546.
- 5. Rajabalendaran N, Burns R, Mollison L. Tropical spastic paraparesis in an Aborigine. Med J Aust 1993; 159: 28-29.
- 6. Einsiedel L, Fernandes L, Spelman T, et al. Bronchiectasis is associated with human T-lymphotropic virus 1 infection in an Indigenous Australian population. Clin Infect Dis 2012; 54: 43-50.
- 7. Kaplan J, Osame M, Kubota H, et al. The risk of development of HTLV-I-associated myelopathy/tropical spastic paraparesis among persons infected with HTLV-I. J Acquir Immune Defic Syndr 1990; 3: 1096-1101.
- 8. Maloney EM, Cleghorn F, Morgan O, et al. Incidence of HTLV-I-associated myelopathy/tropical spastic paraparesis (HAM/TSP) in Jamaica and Trinidad. J Acquir Immune Defic Syndr Hum Retrovirol 1998; 17: 167-170.
- 9. May JT, Stent G, Schnagl RD. Antibody to human T-cell lymphotropic virus type 1 in Australian Aborigines. Med J Aust 1988; 149: 104.
- 10. Einsiedel L, Spelman T, Goeman E, et al. Clinical associations of human T-lymphotropic virus type 1 infection in an Indigenous Australian population. PLoS Negl Trop Dis 2014; 8: e2643.
- 11. Einsiedel L, Cassar O, Gordon L, et al. Human T-lymphotropic virus type 1 infective dermatitis in central Australia. J Clin Virol 2013; 57: 370-373.
- 12. Einsiedel L, Cassar O, Goeman E, et al. Higher human T-lymphotropic virus type 1 subtype C proviral loads are associated with bronchiectasis in Indigenous Australians: results of a case-control study. Open Forum Infect Dis 2014; 1: ofu023.
- 13. Bastian I, Hinuma Y, Doherty RR. HTLV-1 among Northern Territory Aborigines. Med J Aust 1993; 159: 12-16.
- 14. Hino S. Establishment of the milk-borne transmission as a key factor for the peculiar endemicity of human T-lymphotropic virus type 1 (HTLV-1): the ATL Prevention Program Nagasaki. Proc Jpn Acad Ser B Phys Biol Sci 2011; 87: 152-166.
- 15. Gordon C, Hewagama S. Current knowledge about HTLV-1 in Central Australia: proceedings from the first workshop on HTLV-1 in Central Australia. Northern Territory Disease Control Bulletin 2012; 19: 14-15.
- 16. Congress Alukura and Nganampa Health Council. Minymaku kutju tjukurpa. Women’s business manual. 5th edition. Alice Springs: Centre for Remote Health, 2014.
- 17. Glover M, Kira A, Johnston V, et al. A systematic review of barriers and facilitators to participation in randomized controlled trials by Indigenous people from New Zealand, Australia, Canada and the United States. Glob Health Promot 2015; 22: 21-31.
- 18. Rowe T, Dezzutti C, Guenthner PC, et al. Characterization of a HTLV-I-infected cell line derived from a patient with adult T-cell leukemia with stable co-expression of CD4 and CD8. Leuk Res 1995; 19: 621-628.
- 19. Tajima K, Kamura S, Ito S-I, et al. Epidemiological features of HTLV-1 carriers and incidence of ATL in an ATL-endemic island: a report of the community-based co-operative study in Tsushima, Japan. Int J Cancer 1987; 40: 741-746.
- 20. Murphy EL, Wilks R, Hanchard B, et al. A case-control study of risk factors for seropositivity to human T-lymphotropic virus type I (HTLV-I) In Jamaica. Int J Epidemiol 1996; 25: 1083-1089.
- 21. Larsen O, Andersson S, da Silva Z, et al. Prevalences of HTLV-1 infection and associated risk determinants in an urban population in Guinea-Bissau, West Africa. J Acquir Immune Defic Syndr 2000; 25: 157-163.
- 22. Tattermusch S, Bangham C. HTLV-1 infection: what determines the risk of inflammatory disease. Trends Microbiol 2012; 20: 494-500.
- 23. Nagai M, Usuku K, Matsumoto W, et al. Analysis of HTLV-1 proviral load in 202 HAM/TSP patients and 243 asymptomatic HTLV-1 carriers: high proviral load strongly predisposes to HAM/TSP. J Neurovirol 1998; 4: 586-593.
- 24. Tanajura D, Castro N, Oliveira P, et al. Neurological manifestations in human T-cell lymphotropic virus type 1 (HTLV-1)-infected individuals without HTLV-1-associated myelopathy/tropical spastic paraparesis: a longitudinal cohort study. Clin Infect Dis 2015; 61: 49-56.
- 25. Okajima R, Oliveira A, Smid J, et al. High prevalence of skin disorders among HTLV-1 infected individuals independent of clinical status. PLoS Negl Trop Dis 2013; 7: e2546.
- 26. Kelly J, Saggers S, Taylor K, et al. “Makes you proud to be black eh?”: reflections on meaningful indigenous research participation. Int J Equity Health 2012; 11: 40.





Abstract
Objective: Hospital and laboratory data indicate that human T-lymphotropic virus type 1 (HTLV-1) is endemic to central Australia, but no community-based studies of its prevalence or disease burden have been reported. We determined the prevalence rates of HTLV-1 infection and of HTLV-1-associated diseases in a remote Indigenous community.
Setting: A remote Northern Territory community.
Design: All residents were asked to complete a health survey and offered a limited clinical examination, together with serological tests for HTLV-1 and Strongyloides, and HTLV-1 proviral load (PVL) assessment.
Main outcome measures: HTLV-1 seropositivity rates; HTLV-1 PVL (copies/105 peripheral blood leucocytes [PBL]); presentation with HTLV-1-related clinical disease.
Results: HTLV-1 serostatus was determined for 97 of 138 residents (70%). The prevalence of HTLV-1 infection was significantly higher among adults (30 of 74 people tested) than children (1 of 23; P = 0.001). Nine of 30 HTLV-1-positive adults had a clinical syndrome that was potentially attributable to HTLV-1 infection (chronic lung disease, seven; symptomatic strongyloidiasis, two). The median HTLV-1 PVL was significantly higher for adults with chronic lung disease than for those who were asymptomatic (chronic lung disease, 649 copies/105 PBL [IQR, 162–2220]; asymptomatic adults, 40 copies/105 PBL [IQR, 0.9–229]; P = 0.017). Ten of 72 adults tested were seropositive for Strongyloides (six of 28 HTLV-1-positive participants and four of 44 HTLV-1-negative participants; P = 0.17), as were three of 15 children tested; the three children were HTLV-1-negative.
Conclusion: The prevalence of HTLV-1 infection and the rate of disease potentially attributable to HTLV-1 were high among adults in this remote community.