We report a case of khat-associated hepatitis in a 32-year-old Somali man living in Australia. This is the first case of hepatoxicity related to khat ingestion reported in Australia.
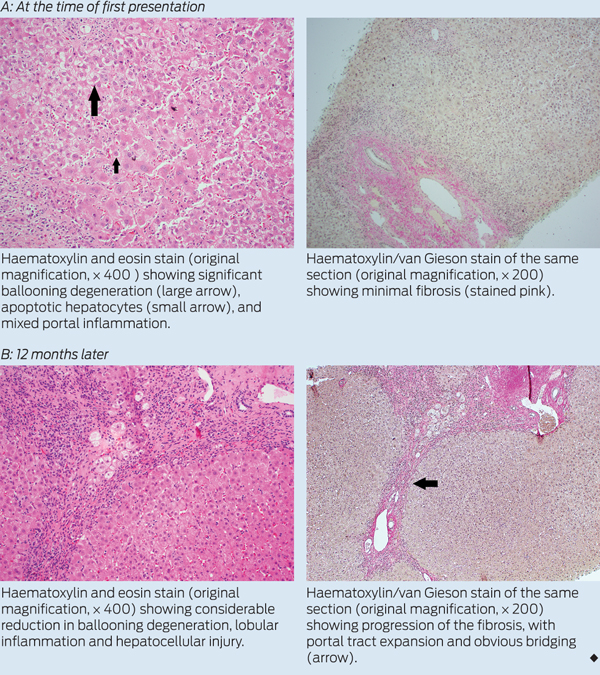
Imaging of the hepatic parenchyma, biliary tree and hepatic vasculature by ultrasonography and computed tomography were unremarkable. A percutaneous liver biopsy, performed on the fifth day of the patient’s admission, showed features that suggested a drug-induced hepatitis, with a differential diagnosis of autoimmune hepatitis. Specifically, there was a moderate portal and marked lobular hepatitis with a mixed inflammatory infiltrate (plasma cells, eosinophils, neutrophils and lymphocytes), mild interface hepatitis and no fibrosis (Box, A). The Ishak score1 for fibrosis stage was F1 and for necroinflammatory activity was 9.
Over the next 12 months, the patient had a further two identical, but less severe, presentations with unexplained hepatitis. Each episode resolved spontaneously between episodes without immunosuppressive or other medication. At each presentation, he was specifically questioned about, but denied, further khat use. His anti-smooth muscle antibody titre subsequently became positive (1:160 [RI, < 1:40]). Although results of a QuantiFERON-TB Gold assay and a Mantoux test were positive, induced sputum and computed tomography of the chest showed no evidence of active tuberculosis. A repeat liver biopsy was performed at the time of his third presentation, 12 months after the initial biopsy. Histological tests again showed features supporting a diagnosis of drug-induced hepatitis, but with some progression of fibrosis with portal-to-portal linkage (Box, B). There were no granulomata.
Khat (Catha edulis) is a plant native to areas in East Africa and the Arabian Peninsula. The plant has stimulant properties which make chewing the leaves of khat popular in Yemen and East African countries.2 In Australia, the practice appears to have become more prevalent with increased immigration, ease of transportation and decreased cost. A recent study reported in the Journal highlighted that khat use among Somali Australians was common, particularly among men.3 Of concern, many users presented to their general practitioners for khat-related adverse events such as mental health problems without disclosing that they were using khat.3 Users knew little about its addictive effects and many considered khat a food, not a drug.3
Since the first report in 2006,4 evidence has emerged to support an association between khat use and hepatotoxicity in humans. Three case series from the United Kingdom and Netherlands describe 19 patients who presented with severe liver injury attributed to khat use.5-7 The true burden is probably very much larger than these series suggest given the widespread use of khat in Somalia and other East African communities, together with a minimal understanding among khat users of its negative consequences. People who use khat may not report this to their GPs because of a genuine belief that it is not linked to any health problems. Further, they may have concerns relating to stigma or illegality.3
While the exact mechanism of khat-associated hepatotoxicity is unknown, evidence from animal studies suggests a direct toxic effect from reactive khat metabolites or an immuno-allergic reaction to these. The active constituents in khat are the alkaloids cathinone and, to a lesser extent, cathine and norephedrine. Fresh leaves contain a higher ratio of cathinone to cathine than dried leaves.8 In Australia, there is no validated test to detect khat or khat metabolites.
A particular manifestation of drug-induced liver injury that can induce chronic liver injury is a drug-induced autoimmune hepatitis. Previous reports suggest that this mechanism might be involved in khat-associated hepatotoxicity. In the Netherlands case series, half of the patients tested positive for autoimmune serological markers.7 Interestingly, in a report of six Somali men with an unusual form of autoimmune hepatitis who presented to two London hospitals between 1996 and 2003,9 a drug history was recorded, but this was before the first documented case of khat hepatotoxicity. Communication with an expert in the field has confirmed that since that publication, further history reveals these patients almost certainly took khat (M Y Morgan, Principal Research Associate, Institute for Liver and Digestive Health, University College London, personal communication).
The natural history of khat-associated hepatotoxicity is uncertain. From the available literature, it appears that the prognosis is unclear, and liver injury may progress to cirrhosis or fulminant hepatic failure.5,6,10 While corticosteroids may be beneficial in some cases of drug-induced autoimmune hepatitis, evidence is lacking to support their use in khat-associated hepatotoxicity.5,6
- 1. Ishak K, Baptista A, Bianchi L, et al. Histological grading and staging of chronic hepatitis. J Hepatol 1995; 22: 696-699.
- 2. Al-Habori M. The potential adverse effects of habitual use of Catha edulis (khat). Expert Opin Drug Safe 2005; 4: 1145-1154.
- 3. Douglas H, Boyle M, Lintzeris N. The health impacts of khat: a qualitative study among Somali-Australians. Med J Aust 2011; 195: 666-669. <MJA full text>
- 4. Brostoff JM, Plymen C, Birns J. Khat — a novel cause of drug-induced hepatitis. Eur J Intern Med 2006; 17: 383.
- 5. Peevers CG, Moorghen M, Collins PL, et al. Liver disease and cirrhosis because of Khat chewing in UK Somali men: a case series. Liver Int 2010; 30: 1242-1243.
- 6. Chapman MH, Kajihara M, Borges G, et al. Severe, acute liver injury and khat leaves. N Engl J Med 2010; 362: 1642-1644.
- 7. Stuyt RJ, Willems SM, Wagtmans MJ, et al. Chewing khat and chronic liver disease. Liver Int 2011; 31: 434-436.
- 8. Toennes SW, Harder S, Schramm M, et al. Pharmacokinetics of cathinone, cathine and norephedrine after the chewing of khat leaves. Br J Clin Pharmacol 2003; 56: 125-130.
- 9. D’Souza R, Sinnott P, Glynn MJ, et al. An unusual form of autoimmune hepatitis in young Somalian men. Liver Int 2005; 25: 325-330.
- 10. Roelandt P, George C, d’Heygere F, et al. Acute liver failure secondary to khat (Catha edulis)-induced necrotic hepatitis requiring liver transplantation: case report. Transplant Proc 2011; 43: 3493-3495.





No relevant disclosures.