Since 1977, South Australia has had a state-based cancer registry that receives statutory notification of all invasive cancer diagnoses. It also collects survival data for patients with cancer through access to deaths registries. Measuring survival over time provides an indicator of the care provided to patients with cancer. Population data of this kind are more informative about trends in care than are data on individual institutions or patient outcomes. However, it is also important to compare institutional data with population data to monitor treatment outcomes in individual institutions.
The SA Cancer Registry covers the whole state and captures lymphoma diagnosis, incidence and survival data. Its procedures have been described previously.1
Data on deaths are collected through routine notifications, and by electronic searches of official state death records, the National Death Index at the Australian Institute of Health and Welfare, and interstate registries. Completeness of recording of deaths has been checked (by active follow-up, and with deaths reported independently); this showed minimal data were missing, with little effect on calculated survival rates.2,3
During the data collection period there were a number of classifications of lymphoma in use. The World Health Organization classification of tumours of haemopoietic and lymphoid tissues (initially published in 2001 and revised in 2008) is generally regarded as the standard classification of lymphoma.4 The older Registry data available to us used a number of other classifications including the International Working Formulation (IWF).5 This has a relatively simple clinical division of non-Hodgkin lymphoma (NHL) into low, intermediate and high grade. There is evidence that this is a reasonable approach to assessing lymphoma outcomes over time.5-8 Where possible, all cases were categorised according to the IWF.
Stata Statistical Software, release 9.2 (StataCorp LP, College Station, Tex, USA) was used to analyse the data. Case survival rates were calculated with a censoring date at 31 December 2007 (for patients alive on that date). Kaplan–Meier product-limit estimates of disease-specific survival were calculated, treating deaths from other causes as censored observations. Multivariable Cox proportional hazards regression was also undertaken to assess sociodemographic and histological predictors of lymphoma survival outcomes. The regression analysis employed the same censoring criteria as the Kaplan–Meier analyses. The assumptions underlying the analysis, including proportionality and absence of collinearity, were satisfied.9
Previous analyses have shown very similar survival estimates in SA, irrespective of whether relative survival rates or disease-specific survival rates were used, such that disease-specific survival rates could be regarded as a good proxy for relative survival rates at a population level. For example, in a study of patients diagnosed from 1977 to 1996, 10-year lymphoma survival rates were identical (49%) for SA, irrespective of methodology.3
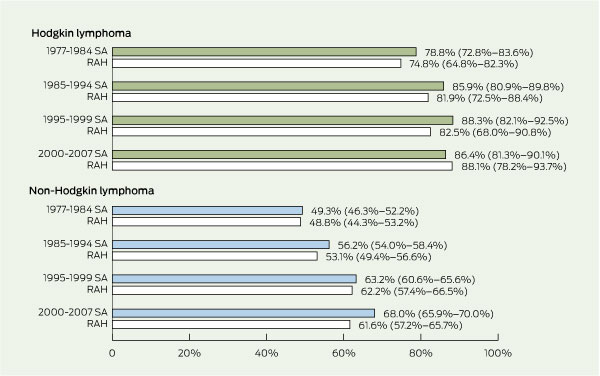
Five-year survival rates of patients with HL in SA increased by 7.6 percentage points from 1977–1984 to 2000–2007 (Box 1). Although there was a trend to an overall increase in survival, neither the overall increase nor the increase between the various periods was statistically significant. At the RAH, the 5-year survival rates of patients with HL increased by 13.3 percentage points in the same period.
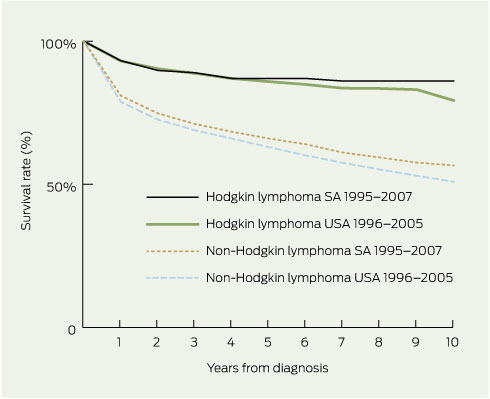
The survival rates of patients with HL in SA (from 1995) were 93.2% at 1 year, 89.8% at 2 years and 86.1% at 10 years, and are comparable with US data from 1996 (Box 2).
Five-year survival rates of patients with HL treated at the RAH for 2000–2007 (88.1%) and in SA as a whole (86.4%) (Box 1) compare favourably with the 85% survival rates reported in Australia during 1998–2004 and in the US in 1999–2005 (Box 3).
For patients in SA, the relative risk of death from HL increased with age from 1.00 (reference value) for those under 40 years to 1.38 for 40–54 year olds rising to 12.21 for those 75 years and over. The relative risk of death from HL at the RAH increased with age in a similar manner (Box 4).
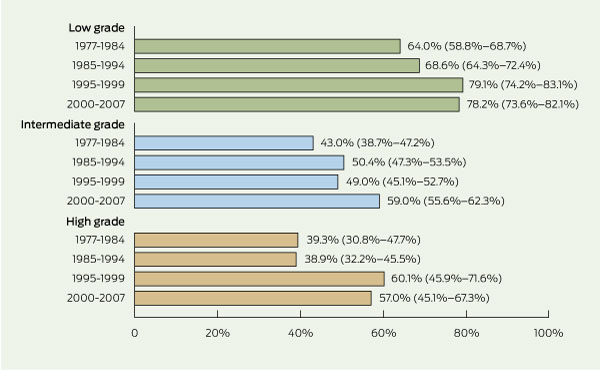
The 5-year survival rates of patients with NHL in SA increased by 18.7 percentage points from 1977–1984 to 2000–2007 (Box 1). Box 2 shows that yearly survival rates for patients with NHL in SA (1995–2007) also compare favourably with NHL survival rates in the US (1996–2005). Survival rates for patients with NHL by IWF grade and diagnosis period are given in Box 5. From 1977–1984 to 2000–2007, the 5-year survival rates for low-, intermediate- and high-grade NHL increased by 14.2, 16.0, and 17.7 percentage points, respectively. For intermediate-grade NHL, the 5-year survival rate increase was statistically significant, but for high-grade NHL only the survival rate increases between 1985–1994 and 1995–1999 were significant.
For patients with NHL, the increase in the relative risk of death between those under 40 years and those over 75 years was not as marked as in patients with HL (Box 4). For patients with low-grade NHL, the relative risk of death increased significantly from a reference value of 1.00 for those aged under 40 years to 6.32 for those 75 years and older. Likewise, for intermediate-grade NHL, the relative risk of death increased with age from 1.00 to 3.39, and for high-grade NHL from 1.00 to 5.64.
Our study shows that over the 30-year study period, there has been a progressive improvement in 5-year survival rates for patients with HL — from 79% to 86% in SA compared with 78% to 85% in Australia as a whole.11 There was minimal deterioration in survival beyond 5 years. In SA, the risk of death was halved in this period. These data are comparable with the Surveillance Epidemiology and End Results (SEER) data from the US.10 The relative risk of death from HL in SA rose progressively with age. Compared with the under-40-years age group, the relative risk of death was six times greater in patients aged 65–74-years and 12 times greater in those 75 years and over. These results confirm that HL can be very effectively treated, and that about 85% of patients are still alive after 10 years, although older patients have poorer outcomes.
The 5-year survival rate for patients with NHL in SA also showed a progressive improvement from just under 50% to 68% in the 30-year period. The risk of death was halved in this period. The data for patients with NHL are comparable with the SEER data for NHL from the US.10 However, the survival patterns in patients with NHL and those with HL are different. The overall survival rate was lower in patients with NHL, and there was a continuous decline in survival rate even beyond 5 years.
There have also been changes in chemotherapy protocols. From 1982 in SA, ABVD (adriamycin, bleomycin, vinblastine, dacarbazine) replaced MOPP (mechlorethamine, vincristine [Oncovin, Aspen], procarbazine and prednisone) as chemotherapy for HL. For intermediate-grade NHL, CHOP chemotherapy (cyclophosphamide, adriamycin [doxorubicin / hydroxydoxorubicin], vincristine [Oncovin], prednisolone) was used from 1986.12-16
Rituximab was introduced in Australia in 1999 and was increasingly used from 2000, and this therapy, alone or in combination with chemotherapy, is most effective in improving survival rates for patients with all grades of B cell NHL, particularly elderly patients with intermediate-grade NHL.17,18
1 Five-year survival rates (95% CI) — South Australian patients with lymphoma and the subgroup treated at the Royal Adelaide Hospital*†
 | |||||||||||||||
2 Survival rates for South Australian patients with Hodgkin (n = 458) and non-Hodgkin lymphoma (n = 4387) by years from diagnosis, compared with rates for United States patients*
 | |||||||||||||||
|
* US Surveillance Epidemiology and End Results (SEER) data.10 | |||||||||||||||
3 Five-year survival rates of patients with lymphoma in South Australia (SA), Australia (Aus)* and the United States (US)†
Received 9 January 2011, accepted 30 May 2011
- Brendon J Kearney1
- Luen Bik To1
- Daniel J Kearney1
- David Roder2
- Colin G Luke3
- Ian D Lewis1
- Pratyush Giri1
- 1 Haematology Directorate, SA Pathology and Haematology Unit, Royal Adelaide Hospital, Adelaide, SA.
- 2 Cancer Council South Australia, Adelaide, SA.
- 3 SA Department of Health, Adelaide, SA.
No relevant disclosures.
- 1. South Australian Cancer Registry. Epidemiology of Cancer in South Australia. Incidence, mortality and survival 1977 to 1999. Incidence and mortality 1999. Adelaide: Openbook Publishers, 2000.
- 2. Bonett A, Roder D, Esterman A. Cancer case-survival rates for South Australia: a comparison with US rates and a preliminary investigation of time trends. Med J Aust 1988; 148: 556-559.
- 3. South Australian Cancer Registry. Epidemiology of cancer in South Australia. Incidence, mortality and survival 1977 to 1996. Incidence and mortality 1996. Adelaide: Openbook Publishers, 1997.
- 4. Jaffe ES, Harris NL, Stein H, Vardiman JW, editors. World Health Organization classification of tumours: pathology and genetics. Tumours of haematopoietic and lymphoid tissue. Lyon: IARC Press, 2001.
- 5. Clarke CA, Undurraga DM, Harasty PJ, et al. Changes in cancer registry coding for lymphoma subtypes: reliability over time and relevance for surveillance and study. Cancer Epidemiol Biomarkers Prev 2006; 15: 630-638.
- 6. Rosenberg SA, Berard CW, Brown BW, et al. National Cancer Institute sponsored study of classifications of non-Hodgkin’s lymphomas. Cancer 1982; 49: 2112-2135.
- 7. Harris NL, Jaffe ES, Stein H, et al. A revised European-American classification of lymphoid neoplasms: a proposal from the International Lymphoma Study Group. Blood 1994; 84: 1361-1392.
- 8. Shipp MA, Mauch PM, Harris NL. Non-Hodgkin’s lymphomas. In: DeVita VT Jr, Hellman S, Rosenberg SA, editors. Cancer: principles and practice of oncology. 5th ed. Philadelphia: Lippincott-Raven, 1997: 2165-2220.
- 9. Armitage P, Berry G. Statistical methods in medical research. Oxford: Blackwell Scientific Publications, 1987.
- 10. Horner MJ, Ries LAG, Krapcho M, et al, editors. SEER cancer statistics review, 1975-2006. Bethesda, Md: National Cancer Institute, 2009 (based on November 2008 Surveillance Epidemiology and End Results data submission, posted to the SEER web site, 2009). http://seer.cancer.gov/csr/1975_2006/ (accessed Nov 2011).
- 11. Australian Institute of Health and Welfare, Cancer Australia and Australasian Association of Cancer Registries. Cancer survival and prevalence in Australia: cancers diagnosed from 1982 to 2004. Canberra: AIHW, 2008. (AIHW Cat. No. CAN 38; Cancer Series No. 42.)
- 12. Carella AM, Prencipe E, Pungolino E, et al. Twelve years experience with high-dose therapy and autologous stem cell transplantation for high-risk Hodgkin’s disease patients in first remission after MOPP/ABVD chemotherapy. Leuk Lymphoma 1996; 21: 63-70.
- 13. Strickland AH, Beresford JA, Arthur GE, et al. Increased survival in patients diagnosed with Hodgkin disease in Tasmania 1972-1992. Aust N Z J Med 1998; 28: 609-614.
- 14. Bonadonna G, Santoro A. ABVD chemotherapy in the treatment of Hodgkin’s disease. Cancer Treat Rev 1982; 9: 21-35.
- 15. Molnar Z, Schneider T, Varady E, Fleischmann T. ABVD chemotherapy of Hodgkin’s disease. Neoplasma 1997; 44: 263-265.
- 16. Pereira A, Montserrat E, Cervantes F, Rozman C. Advanced lymphoma and CHOP chemotherapy. Ann Intern Med 1986; 105: 631.
- 17. Coiffier B, Lepage E, Herbrecht R, et al. MabThera (rituximab) plus CHOP is superior to CHOP alone in elderly patients with diffuse large B-cell lymphoma (DLCL): interim results of a randomised GELA trial. Blood 2000; 96 (Suppl 1): 223A (Abstract no. 950).
- 18. Coiffier B, Thieblemont C, Van Den Neste E, et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomised study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: a study by the Groupe d’Etudes des Lymphomes de l’Adulte. Blood 2010; 116: 2040-2045.





Abstract
Objective: To determine survival rates of patients with lymphoma in South Australia.
Design and setting: De-identified data from the SA Cancer Registry on all patients with lymphoma were analysed, as well as the subgroup treated at the Royal Adelaide Hospital (RAH). For non-Hodgkin lymphoma (NHL), we used the International Working Formulation (IWF) grading. SA and RAH data on survival rates were compared with those for the whole of Australia and the United States.
Patients: All patients diagnosed with lymphoma and treated in SA in 1977–2007.
Outcome measures: 5-year survival rates for patients with lymphoma, by type of lymphoma and age.
Results: Of the total of 8651 patients with lymphoma, 939 were classified as having Hodgkin lymphoma (HL) and 7712 as having NHL. Of those with NHL, 1805 had low-grade, 3576 intermediate-grade, and 510 high-grade NHL. In another 1821 patients, the data were insufficient to make an IWF grading. There was a substantial increase in 5-year survival rates for patients with lymphoma between 1977 and 2007 in SA. While the increase in 5-year survival rates for HL was 7.6 percentage points, survival rates peaked at 88%. For NHL, there was an 18.7 percentage points increase in 5-year survival rates. The first significant increase of 7 percentage points was associated with the introduction of bone marrow transplantation; this was maintained with the increase in 5-year survival rates reaching 14 percentage points by 1995–1999. Since 1999, there has been a further increase of 5 percentage points in 5-year survival rates with the introduction of rituximab.
Conclusion: Outcomes in patients with NHL have improved significantly, most likely because of the use of bone marrow transplantation and rituximab. Hospital- and state-based cancer registry data reflect the reality of population outcomes and the impact of new technologies.