Over the past decade, published studies have shown the benefits of using simulation for educating trainee surgeons, rather than traditional operating-room training methods with actual patients.1 Having fewer trainees in operating rooms not only lowers costs,2 but also reduces the risks and discomfort to the patient caused by the inexperience of trainee surgeons and their longer operating times. Apart from lowering costs, training by simulation provides unique benefits that training with patients cannot provide. These include:
opportunities for controlled exposure — the instructor has control over the order, frequency and complexity of the training;
easily defined metrics for assessing proficiency levels;
“beyond real” capabilities; for example, case simplification, which lets trainees learn specific components of a procedure;1 and
the possibility of continuing a simulated procedure after a mistake has been made, so trainees can learn to recognise when they are on a path to failure.
Existing surgical simulators vary in complexity, with different types suited to the various stages of surgical training. For providing surgeons-in-training with basic technical skills, the primary choice is the Box or part-task trainer, as used in the Fundamentals of Laparoscopic Surgery program of the Society of American Gastrointestinal Endoscopic Surgeons.3 More complex types of simulation are needed to practise the specific techniques and procedures encountered in surgery, and, traditionally, animal and cadaver models have been used. However, acquiring and maintaining high-level skills and gaining accreditation still involves, almost without exception, actual patients. Could current and future developments in computer-based or virtual reality (VR) simulators make them suitable for these purposes?
VR simulators have so far played a fairly limited role in surgical training, mainly because existing VR simulators still fall behind “physical” simulators in several areas. For example, they are considerably more expensive than surgical Box trainers, and cannot yet match them for training certain tasks, such as laparoscopic suturing. They also cannot match animal and cadaver models for tissue behaviour, haptic (tactile) feedback and anatomical realism. Nevertheless, VR simulators have some distinct potential advantages over other simulation types. Metrics are much easier to specify and can be defined in much greater detail, a feature that offsets some of the price difference between VR simulators and Box trainers. Continuing after a mistake has been made is not costly, unlike in animal or cadaver models, and, in general, VR simulators have the potential to be less costly, more available, and do not have the ethical dilemmas of animal and cadaver models.
A comprehensive 2007 review of VR simulators for laparoscopy and colonoscopy training reported that current simulators were still too expensive and too “gameable” (ie, they allowed for too much rote learning, making them unsuitable for advanced skills training).4 In addition, further validation and opportunities for incorporating VR simulators into curricula were needed.4 Another recent study reached similar conclusions — that current VR simulators show benefits for novice-to-intermediate training in colonoscopy and can be used to distinguish experts from novices, but they are not yet ready to replace traditional training or be used for accreditation.5
However, there are now some strong indicators pointing to an expanding role for VR simulators. According to a prospective randomised controlled trial published in 2009, VR simulators are already not only providing basic skills and part-task training at the novice level, but also increasingly at the intermediate skills level, with VR training able to take novices to the performance level of a laparoscopist with intermediate experience.6
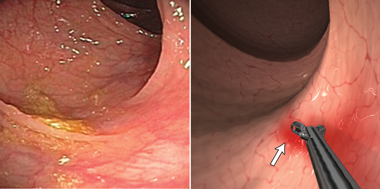
Supporting this expanding role are the continuing advances in the technologies underlying VR simulation, driven mainly by the games and entertainment industries. Developments in these industries have led to the availability of hardware and software that can provide much more realistic simulation models than those currently in use. Evidence for this can already be seen in the next generation of simulator prototypes showcased in recent journals and conferences,7,8 such as the colonoscopy simulator currently being developed at the CSIRO (Commonwealth Scientific and Industrial Research Organisation) Australian e-Health Research Centre. This makes use of the “massively parallel” processing power of current graphics processing units to provide highly detailed, dynamic interactive models of the colon and the colonoscope (Box).7
As every patient is different, and each patient’s pathological condition is subject to change, a great variety of case complexity is required to train advanced skills levels. A simulator that can only generate a few scenarios may only teach the trainee to “rote learn” the map to a specific scenario solution, not how to recognise which aspects of a procedure affect its progress and outcome and in which way. Hence, the simulator must be able to provide a significant variety of case scenarios in all aspects of the simulated procedure (ie, variations in pathology, anatomy, appearance, physical properties and complications). However, it must also be able to repeat a scenario exactly, as it is crucial for trainees, and trainers, to analyse mistakes and learn from them, as shown by Issenberg et al9 in their analysis of the features of high-fidelity simulations that lead to effective learning.
Performance assessment is important in the learning process, and essential in certification. To allow for objective assessment of a trainee’s performance, the simulator must be able to define appropriate metrics from the simulated task for qualitative assessment purposes. These metrics must be relevant to the procedure being trained, as well as accurate and validated. To provide trainees with a fair assessment relative to their peers, exam scenarios must be repeatable within certain limits and validated. Assessment should be used to provide comprehensive feedback to the trainee, as this is the most important feature of effective learning.9
Without this validation, no certifying body, trainer, trainee or patient will accept VR simulators.
- Hans de Visser1
- Marcus O Watson2
- Olivier Salvado1
- Joshua D Passenger1
- 1 Australian e-Health Research Centre, CSIRO ICT Centre, Brisbane, QLD.
- 2 Queensland Health Skills Development Centre, Brisbane, QLD.
We acknowledge the CSIRO Preventative Health National Research Flagship for funding of this research.
The CSIRO has received licence fees for the colonoscopy simulator, as well as funding for overseas travel to demonstrate the simulator, from Surgical Science Sweden AB.
- 1. Hutchins M, Adcock M, Stevenson D, et al. The design of perceptual representations for practical networked multimodal virtual training environments. Proceedings of the 11th International Conference on Human–Computer Interaction; 2005 July 22-27; Las Vegas, USA. Philadelphia: Taylor & Francis Group LLC (Lawrence Erlbaum Associates, Inc), 2005.
- 2. Bridges M, Diamond DL. The financial impact of teaching surgical residents in the operating room. Am J Surg 1999; 177: 28-32.
- 3. Peters JH, Fried GM, Swanstrom LL, et al. Development and validation of a comprehensive program of education and assessment of the basic fundamentals of laparoscopic surgery. Surgery 2004; 135: 21-27.
- 4. Dunkin B, Adrales G, Apelgren K, Mellinger J. Surgical simulation: a current review. Surg Endosc 2007; 21: 357-366.
- 5. Gerson LB. Evidence-based assessment of endoscopic simulators for training. Gastrointest Endosc Clin N Am 2006; 16: 489-509.
- 6. Larsen CR, Soerensen JL, Grantcharov TP, et al. Effect of virtual reality training on laparoscopic surgery: randomised controlled trial. BMJ 2009; 338: b1802.
- 7. de Visser H, Passenger J, Conlan D, et al. Developing a next generation colonoscopy simulator. Int J Image Graphics 2010; 10: 203-217.
- 8. Comas O, Taylor ZA, Allard J, et al. Efficient nonlinear FEM for soft tissue modelling and its GPU implementation within the open source framework SOFA. Proceedings of the International Symposium on Computational Models for Biomedical Simulation; 2008 July 7-8; London, UK. Berlin: Springer (Lecture Notes in Computer Science, Vol 5104: 28-39), 2008.
- 9. Issenberg SB, McGaghie WC, Petrusa ER, et al. Features and uses of high-fidelity medical simulations that lead to effective learning: a BEME systematic review. Med Teach 2005; 27: 10-28.





Abstract
There is increasing evidence that educating trainee surgeons by simulation is preferable to traditional operating-room training methods with actual patients. Apart from reducing costs and risks to patients, training by simulation can provide some unique benefits, such as greater control over the training procedure and more easily defined metrics for assessing proficiency.
Virtual reality (VR) simulators are now playing an increasing role in surgical training. However, currently available VR simulators lack the fidelity to teach trainees past the novice-to-intermediate skills level. Recent technological developments in other industries using simulation, such as the games and entertainment and aviation industries, suggest that the next generation of VR simulators should be suitable for training, maintenance and certification of advanced surgical skills.
To be effective as an advanced surgical training and assessment tool, VR simulation needs to provide adequate and relevant levels of physical realism, case complexity and performance assessment. Proper validation of VR simulators and an increased appreciation of their value by the medical profession are crucial for them to be accepted into surgical training curricula.