Murine typhus (MT) occurs worldwide, but, in Australia, is only regularly diagnosed in south-west Western Australia. Meningoencephalitis is an uncommon complication of MT, often accompanied by rash or systemic involvement. We report a case of MT presenting exclusively with meningoencephalitis, raised intracranial pressure, papilloedema and bilateral 6th cranial nerve palsies. MT should be considered in patients with “aseptic” meningitis or meningoencephalitis, even in the absence of other typical features of a typhus-like illness. (MJA 2011; 194: 652-654)
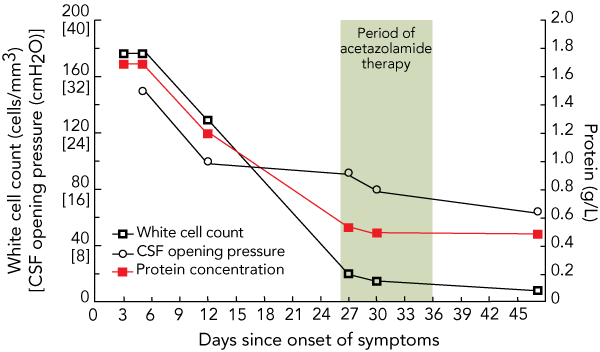
He was diagnosed with meningoencephalitis after a lumbar puncture on the day of admission showed a predominantly lymphocytic pleocytosis of 180 lymphocytes/mm3 (reference range [RR], < 5 lymphocytes/mm3) with an elevated protein level of 1.7 g/L (RR, < 0.45 g/L) and a normal glucose concentration (Box 1). He was admitted to the intensive care unit (ICU) and remained there for a week because of fluctuating confusion and fever. During that week he had two further episodes like the one described before his presentation, including on Day 10 of admission requiring readmission to the ICU. Empiric treatment with a number of antimicrobial medications was commenced at standard doses in the following order: ceftriaxone (intravenously, Days 3 to 17 of illness); ampicillin (intravenously, Days 4 to 9); aciclovir (intravenously, Days 4 to 17); vancomycin, oseltamivir and azithromycin (intravenously, Days 10 to 17). Phenytoin therapy (1000 mg intravenous loading dose, followed by 300 mg orally, daily) was commenced on the day of admission owing to the possible seizure activity before presentation. He was discharged from hospital on Day 17 of the illness after the fever and headaches resolved.
On presentation, the results of blood investigations including a full blood count, liver function tests, a C-reactive protein test, renal function tests and electrolyte level measurements were normal. A transient leukocytosis developed after admission and peaked on Day 6 of admission at 14.0 × 109/L, with a predominant neutrophilia. At no time was there significant derangement of liver function tests. Findings on brain imaging, including gadolinium enhanced magnetic resonance imaging of the brain on two occasions, magnetic resonance venogram and magnetic resonance angiogram were all normal. Serial lumbar punctures were performed (Box 1) showing the reducing white cell count and protein level. Cerebrospinal fluid (CSF) opening pressure was elevated to 30 cmH2O (RR, < 18 cmH2O) measured on Day 5 and this normalised by Day 27. Results of serological tests for human immunodeficiency virus, leptospirosis, Q fever, mycoplasma, brucellosis, psittacosis, and Ross River fever, which were performed during both the acute and convalescent phases of the illness, were negative. Repeated tests of the CSF for cryptococcal antigens were negative. CSF cultures for bacteria, fungi and mycobacteria were also negative, as was polymerase chain reaction testing for herpes viruses and enterovirus. Indirect immunofluorescence detecting total antibody against rickettsiae in the typhus group (Rickettsiae typhi and R. prowazekii), spotted fever group (R. honei and R. australis) and scrub typhus (Orientia tsutsugamushi) was performed in parallel on serum collected on Days 9 and 41. Antibodies against R. typhi were detected at a titre of 1/256 on Day 9, rising to ≥ 1/1024 on Day 41. Antibodies in each CSF specimen showed cross-reactivity with R. prowazekii at equivalent titres, but not with other rickettsial species.
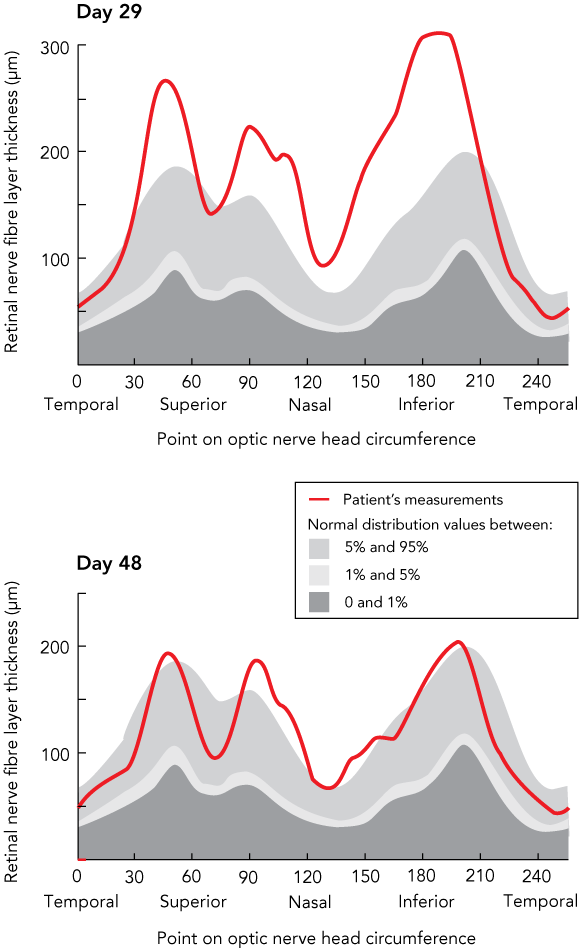
The patient first noticed diplopia and reduced visual acuity on Day 10, which peaked on Day 20 of his illness. At this point, bilateral partial 6th nerve palsies and papilloedema were noted. Because of persistent visual symptoms and papilloedema, therapy with 250 mg acetazolamide orally, twice daily was commenced on Day 26. Papilloedema was documented on Day 28 by ocular coherence tomography (OCT), a measure of retinal nerve fibre layer thickness and indicative of the degree of optic disc swelling (Box 2). The 6th nerve palsies resolved after Day 30 and regular fundoscopy and repeat retinal OCT showed the papilloedema continued to resolve (Box 2). Acetazolamide therapy was ceased on Day 35. The patient had not experienced recurrence of the headaches or neurological symptoms at 6 months after the onset of the illness.
MT (endemic typhus) was first described in 1922 with a series of cases from South Australia.1 There have been further outbreaks in the Darling Downs region of south-east Queensland and south-west Western Australia.2 In each series, exposure to rodents was reported in a significant proportion of cases. MT has been reported worldwide in diverse geographic areas.3 Several areas continue to regularly report new cases including Perth in WA,4 South-East Asia,5 and Texas in the United States.6 The first case of MT in Victoria was recently reported,7 and no locally acquired cases of MT have been reported from NSW since 1944.8
The disease is caused by R. typhi, which is an obligate intracellular gram-negative bacterium with an incubation period of 7–14 days. There are two major sources of transmission. The major route of infection worldwide is the rat–flea–rat cycle.9 Rat fleas (Xenopsylla cheopis) transmit R. typhi to a roof or Norwegian rat (Rattus rattus and Rattus norvegicus, respectively). Humans are infected when flea faeces containing R. typhi are inoculated into the site of a flea bite. Alternatively, the organism may be inhaled from an environment contaminated with flea faeces. Another transmission cycle has been identified, which involves predominantly cats and opossums and their fleas (Ctenocephalides felis).10 In the case we present here, it is likely that the patient was infected as a consequence of close contact with rodents, with a respiratory route of infection being most likely.
Although serological testing does not separate MT and epidemic typhus, MT has been distinguished by its milder course and the lower associated mortality.1 Fever is almost universal, and about 75% of affected patients complain of headache. Rash is observed in about 50% of cases, and is usually a macular or maculopapular rash that starts on the trunk and spreads to the extremities. A number of other features may accompany the illness, including myalgia, arthralgia, gastrointestinal symptoms, jaundice and hepatic dysfunction.11 Among the antibiotics given to our patient, azithromycin was the only one with activity against rickettsiae, and may have altered the course of the illness. However, without treatment, MT is usually a self-limiting illness, with symptoms resolving after 2 weeks.1
Neurological complications like those described in this report are uncommon in MT. Aseptic meningitis or meningoencephalitis has been reported in 2%–14% of cases.12,13 There is often a coexisting rash or other systemic features of typhus,12 and isolated meningitis or meningoencephalitis has been reported previously in only six patients.13,14 Papilloedema and raised intracranial pressure can occur with the meningitis, and 6th nerve palsy has been reported in a single case.13 Facial nerve palsy has also been described in association with meningitis in MT.14 Neurological dysfunction is usually reversible. but long-lasting deficits have been reported.13,15 In affected patients, the CSF typically shows a predominantly monocytic pleocytosis with variable elevation of protein concentration.13 The raised intracranial pressure and optic disc swelling in our patient were transient, and it is likely that intracranial pressure changes associated with meningitis are monophasic like MT infection generally. This has important implications for the extent and duration of treatment of associated raised intracranial pressure.
Provenance: Not commissioned; externally peer reviewed.
- 1. Hone FS. A series of cases closely resembling typhus fever. Med J Aust 1922; 1: 1-13.
- 2. Graves S, Stenos J. Rickettsioses in Australia. Ann N Y Acad Sci 2009; 1166: 151–155.
- 3. Civen R, Ngo V. Murine typhus: an unrecognised suburban vectorborne disease. Clin Infect Dis 2008; 46: 913-918. doi: 10.1086/527443
- 4. Beaman MH, Marinovitch N. Murine typhus in metropolitan Perth. Med J Aust 1999; 170: 93-94.
- 5. Suputtamongkol Y, Suttinont C, Niwatayakul K, et al. Epidemiology and clinical aspects of rickettsioses in Thailand. Ann N Y Acad Sci 2009; 1166: 172-179.
- 6. Adjemian J, Parks S, McElroy K, et al. Murine typhus in Austin, Texas, USA, 2008. Emerg Infect Dis 2010; 16: 412-417.
- 7. Jones SL, Athan E, O’Brien D, et al. Murine typhus: the first reported case from Victoria. Med J Aust 2004; 180: 482. <MJA full text>
- 8. Endemic typhus [report by Chairman of National Health and Medical Research Counci]. Med J Aust 1944; 1: 592.
- 9. Chaniotis B, Psarulaki A, Chaliotis G, et al. Transmission of cycle of murine typhus in Greece. Ann Trop Med Parasitol 1994; 88: 645-647.
- 10. Boostrom A, Beier MS, Macaluso JA, et al. Geographic association of Rickettsia felis-infected opossums with human murine typhus, Texas. Emerg Infect Dis 2002; 8: 549-554.
- 11. Dumler SJ, Taylor JP, Walker DH. Clinical and laboratory features of murine typhus in South Texas, 1980 through 1987. JAMA 1991; 266: 1365-1370.
- 12. Silpapojakul K, Ukkachoke C, Krisanapan S, Silpapojakul K. Rickettsial meningitis and encephalitis. Arch Intern Med 1991; 151: 1753-1757.
- 13. Masalha R, Merkin-Zaborsky H, Matar M, et al. Murine typhus presenting as subacute meningoencephalitis. J Neurol 1998; 245: 665-668.
- 14. Vander T, Medvedovsky M, Valdman S, Herishanu Y. Facial paralysis and meningitis caused by Rickettsia typhi infection. Scand J Infect Dis 2003; 35: 886-887. doi: 10.1080/00365540310016853
- 15. Lew HL, Lane B, Zeiner H. Neuroimaging and clinical manifestations of bilateral temporal encephalopathy secondary to murine typhus infection. Am J Phys Med Rehabil 2005; 84: 310-311.





None identified.