We report two Australian patients with possible cobalt toxicity related to metal-on-metal total hip replacements. Both patients were treated for osteoarthritis with a DePuy ASR (articular surface replacement) XL Acetabular Hip System prosthesis, which contains cobalt and chromium, and which has recently been recalled from the market. (MJA 2011; 194: 649-651)
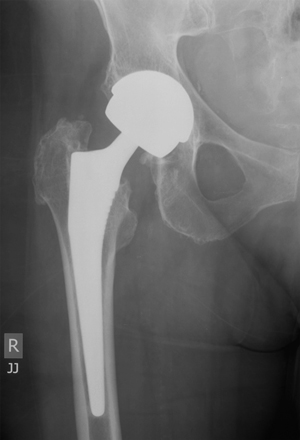
The patient’s original hip replacement had been done in 2006 with an ASR (articular surface replacement) hip prosthesis (ASR XL Acetabular Hip System [DePuy Orthopaedics, Warsaw, Ind, USA]), comprising a large-diameter metal (cobalt and chromium) cup with a large modular metal head on a titanium Corail stem (Box). A revision total hip replacement was performed in March 2011, principally because of her systemic symptoms and elevated cobalt and chromium levels. The ASR metal cup and head were removed and the stem retained. Her acetabulum was revised with an all-polyethylene cemented cup and the head was changed to a ceramic one. At the time of surgery, 30 mL of turbid fluid was aspirated from the joint and debridement of metal stained tissue was undertaken. The concentration of cobalt in the joint fluid was 4218 nmol/L and chromium was 217 000 nmol/L. Cerebrospinal fluid (CSF) collected at surgery showed a cobalt level of 9 nmol/L and a chromium level of 13 nmol/L (no reference ranges for these in CSF), showing that the ions had crossed the blood–brain barrier.
Total hip replacement is generally a successful operation, with a long clinical history of good outcomes. Conventional total hip prostheses consist of a metal head that fits into a polyethylene cup. Concerns have been raised, particularly in younger patients, that the plastic cup suffers wear and tear with time and may require revision. In an attempt to decrease wear at the interface of the articulation, metal-on-metal bearings (Box), composed of cobalt and chromium, have gained widespread popularity and have been used extensively, particularly in the United States.
However, some of these metal-on-metal prostheses have not lived up to the promise of increased durability. The Australian National Joint Replacement Registry (NJRR) notes large differences in the performance of different types of metal-on-metal hip prostheses. An acceptable upper rate of failure of hip prostheses is considered to be below 1% per year, with the vast majority of implants studied by the NJRR falling well below this failure rate.1 The ASR prosthesis originally implanted in both patients reported here has recently been withdrawn from the market, after about 100 000 were implanted worldwide. Its revision rate reported on the NJRR in 2010 was 6.4% at 3 years when inserted with a Corail stem and 10.9% at 5 years when inserted as a resurfacing.1 Updated guidance from the British Hip Society’s 2011 conference noted a
higher than anticipated early failure rate [in large-diameter metal-on-metal hip replacements]. These range from [a] 21% revision rate at 4 years (potentially rising to 35% if all currently known painful implants progress to revision) to 49% at 6 years for the ASR XL device. Other devices have a revision or impending revision rate of 12 – 15% at 5 years.2
An additional problem with metal-on-metal bearings is that they release a variety of metal ions into local tissue and the general circulation, with chromium being the most widely reported of these. Increased cobalt levels have been reported to be associated with neurological3,4 (hand tremor, incoordination, cognitive decline, depression, vertigo, hearing loss and visual changes), cardiac (arrhythmias and cardiomyopathy) and endocrine5,6 symptoms. Symptoms of cobaltism have been previously described when cobalt was used to treat refractory anaemia in patients 40 years ago.7 The term “arthroprosthetic cobaltism” has been coined to describe these manifestations in patients with joint replacements.3
The direct effect of cobalt on cells has recently been explored. Exposure to cobalt ions was found to significantly inhibit osteoblast function by reducing alkaline phosphatase activity and calcium deposition, and to rapidly induce the secretion of proteins IL-8 and MCP-1 in primary human osteoblasts.8 This may have implications for bone ingrowth onto implants and osteolysis around the hip replacement. Other reported effects of elevated cobalt levels on cells include abnormal lymphocyte function,9,10 chemokine secretion11 and ischaemic changes in rat brains.12 A relationship between symptoms and peak cobalt levels, or the length of exposure to cobalt, has not been established.
The British Hip Society recommends regular follow-up of patients with metal-on-metal hip replacements for at least 5 years and probably for the life of the prosthesis.2 Clinical manifestations of cobalt toxicity may occur many years after implantation of the prosthesis and appear to stay elevated over extended periods of time.13 Long-term exposure to cobalt may also be associated with cancer.14 Patients should therefore be followed up carefully, with specific questioning about alterations in neurological, cardiac, respiratory and endocrine function.
Provenance: Not commissioned; externally peer reviewed.
- 1. Australian Orthopaedic Association National Joint Replacement Registry. Annual report 2010. Adelaide: AOA, 2010. http://www.dmac.adelaide.edu.au/aoanjrr/documents/aoanjrrreport_2010.pdf (accessed May 2011).
- 2. Hodgkinson J, Skinner J, Kay P. Updated guidance on large diameter metal on metal bearing total hip replacements. London: British Hip Society, British Orthopaedic Association, National Joint Registry and Medicines and Healthcare products Regulatory Agency, 2011. http://www.britishhipsociety.com/pdfs/BHS_MOM_THR.pdf (accessed May 2011).
- 3. Tower SS. Arthroprosthetic cobaltism: neurological and cardiac manifestations in two patients with metal-on-metal arthroplasty: a case report. J Bone Joint Surg Am 2010; 92: 2847-2851.
- 4. Rizzetti MC, Liberini P, Zarattini G, et al. Loss of sight and sound. Could it be the hip? Lancet 2009; 373: 1052.
- 5. Brock T, Stopford W. Bioaccessibility of metals in human health risk assessment: evaluating risk from exposure to cobalt compounds. J Environ Monit 2003; 5: 71N-76N.
- 6. Keegan GM, Learmonth ID, Case CP. Orthopaedic metals and their potential toxicity in the arthroplasty patient: a review of current knowledge and future strategies. J Bone Joint Surg Br 2007; 89: 567-573.
- 7. Schirrmacher UO. Case of cobalt poisoning. BMJ 1967; 1: 544-545.
- 8. Queally JM, Devitt BM, Butler JS, et al. Cobalt ions induce chemokine secretion in primary human osteoblasts. J Orthop Res 2009; 27: 855-864.
- 9. Hart AJ, Hester T, Sinclair K, et al. The association between metal ions from hip resurfacing and reduced T-cell counts. J Bone Joint Surg Br 2006; 88: 449-454.
- 10. Daou S, El Chemaly A, Christofilopoulos P, et al. The potential role of cobalt ions released from metal prosthesis on the inhibition of Hv1 proton channels and the decrease in Staphylococcus epidermidis killing by human neutrophils. Biomaterials 2011; 32: 1769-1777.
- 11. Devitt BM, Queally JM, Vioreanu M, et al. Cobalt ions induce chemokine secretion in a variety of systemic cell lines. Acta Orthop 2010; 81: 756-764.
- 12. Caltana L, Merelli A, Lazarowski A, Brusco A. Neuronal and glial alterations due to focal cortical hypoxia induced by direct cobalt chloride (CoCl2) brain injection. Neurotox Res 2009; 15: 348-358.
- 13. Engh CA Jr, MacDonald SJ, Sritulanondha S, et al. 2008 John Charnley award: metal ion levels after metal-on-metal total hip arthroplasty: a randomized trial. Clin Orthop Relat Res 2009; 467: 101-111.
- 14. International Agency for Research on Cancer. IARC monographs on the evaluation of carcinogenic risks to humans: volume 86. Cobalt in hard metals and cobalt sulfate, gallium arsenide, indium phosphide and vanadium pentoxide. Lyon: IARC, 2006.





We thank the patients for allowing us to publish details of their cases.
Ross Crawford has received consultancy fees and royalties from Stryker, a manufacturer of joint replacement prostheses.