The World Health Organization declared the first influenza pandemic of the 21st century on 11 June 2009. The pandemic (H1N1) 2009 virus is a novel A(H1N1) quadruple-reassortant virus that contains genes from North American and Eurasian lineages of swine, avian, and human influenza A viruses.1,2 The haemagglutinin (HA) of the pandemic (H1N1) 2009 strain belongs to the classic swine lineage, which probably entered swine populations around 1918; it is antigenically similar to triple-reassortant swine influenza viruses found in pigs (and occasionally in humans) in the United States since the late 1990s.1-4 The pandemic (H1N1) 2009 virus is genetically and antigenically different to the seasonal human influenza A(H1N1) viruses circulating over recent decades.3
A consistent feature of influenza pandemics in the 20th century was that both morbidity and mortality disproportionately affected young people, especially during the 1918–1919 pandemic when about half the influenza-related deaths occurred in the 20–40-years age group.5-7 It is thought that influenza A(H1N1) circulated in humans for some years before 1873, so those born before 1873 and still alive in 1918 (ie, aged 45 years or older) may have had some cross-protection.8 Antigen “recycling” may also have provided the partial protection against influenza-related death observed during the 1968 A(H3N2) pandemic among people born before the 1889–1891 pandemic. Studies have confirmed the pre-pandemic presence in 1968 of H3-like antibodies in people born before 1892.9
Although people aged at least 65 years (and even more so those 85 years or older) are at high risk of seasonal influenza-related complications and death, the highest attack rates for pandemic (H1N1) 2009 influenza were in younger adults and children.10,11 The significantly lower proportion of cases in older people may be explained by cross-reacting antibodies to the pandemic virus among this population. Recent pandemic (H1N1) 2009-specific serosurveys have shown significantly higher levels of cross-reacting influenza antibody in pre-pandemic sera from the population aged over 65 years.12-16 The oldest group (≥ 85 years), who were born around the time of the 1918 pandemic or its immediate aftermath, may have even better protection. For pandemic control, it is important to know about pre-existing immunity to the pandemic strain across the general population, and especially among high-risk groups such as older people, so as to better focus prophylaxis with vaccines and antivirals.17
The first study was a cluster randomised controlled trial of influenza treatment and prevention conducted in 2006–2008 in 16 ACFs in the greater Sydney region. Acute and convalescent blood samples were collected from ill residents of the nine ACFs where influenza outbreaks occurred (one in 2006, five in 2007 and three in 2008). All outbreaks were due to seasonal influenza A(H3N2) or B; none were due to seasonal A(H1N1) and no patients seroconverted to the pandemic (H1N1) 2009 virus.18
During early June 2009, we assisted with managing an outbreak of influenza-like illness in an ACF in rural New South Wales. It was shown to be mainly due to rhinovirus, with 10 residents testing positive to rhinovirus and one to pandemic (H1N1) 2009 using nucleic acid testing.19 After consent was obtained, acute and convalescent blood samples were collected from the 28 ACF residents within 1 week of the first case; for the purpose of this study, the acute samples were classified as pre-pandemic (in any case, only one patient seroconverted).
A pandemic (H1N1) 2009-specific HAI assay was performed using a gamma-irradiated preparation of influenza A/California/07/2009 virus.20 We have no evidence that gamma irradiation affects the estimation of antibody levels.
Briefly, serum specimens were treated with receptor-destroying enzyme to remove inhibitors, diluted 1:10, and heat inactivated. Serial doubling dilutions (and appropriate controls) were reacted with antigen in a microtitre tray before a 1% v/v suspension of human group O red blood cells was added. Endpoints were read by two independent operators as the last dilution showing complete inhibition of haem-agglutination after 1–2 hours. For the purpose of this study, titres ≥ 1:40 were considered to reduce the risk of clinical infection with pandemic (H1N1) 2009 influenza by 50%.21 All testing was performed in a single laboratory.
In total, 259 serum samples from individuals aged 60 years or older (range, 60–101 years) were tested (Box 1). Overall, the proportion of individuals with cross-reacting antibody titres ≥ 1:40 was 37.5% (97/259; 95% CI, 31.6%–43.3%). Respectively, the proportions with cross-reacting antibody titres of < 1:10, 1:10, 1:20, 1:40 and ≥ 1:80 were 23.9% (95% CI, 19.1%–29.5%), 17.3% (95% CI, 13.2%–22.4%), 20.8% (95% CI, 16.3%–26.2%), 19.7% (95% CI, 15.3%–24.9%) and 18.1% (95% CI, 13.9%–23.3%).
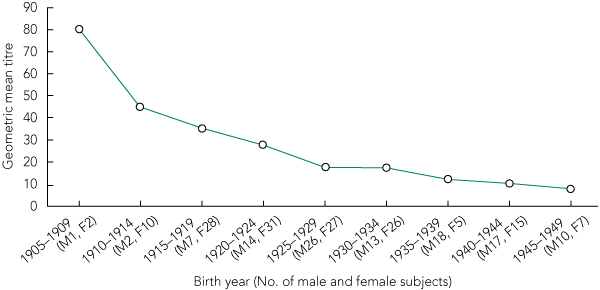
For each of the three studies, there was a significant trend for the proportion of individuals with protective antibody titres (≥ 1:40) to increase with increasing age of the participants. Combined, the trend was highly significant (Box 1). The prevalence of cross-reacting antibody titres ≥ 1:40 was highest in the oldest age groups, with more than 60% of individuals aged ≥ 85 years having titres ≥ 1:40. The prevalence decreased significantly with successively younger cohorts, to only 12% in those aged 60–64 years (Box 1 and Box 2). The biggest step-down and the only significant difference between two consecutive cohorts (P = 0.025) was between the 85–89-years and 80–84-years age groups. Year-by-year examination of the data suggested that this step-down occurred for those born in or after 1924, with this group being less likely to have titres ≥ 1:40 (P = 0.0001). Comparing all individuals by GMT and birth year showed that the older the person, the more likely he or she was to have a high HAI antibody titre to the pandemic (H1N1) 2009 virus (Box 3).
We found that the highest levels of measurable cross-reacting HAI antibodies to pandemic (H1N1) 2009 influenza were in individuals born in the years before or a few years after the 1918–1919 pandemic. The lower proportions with cross-reacting HAI antibodies in those born from 1925 onward may reflect subsequent antigenic drift in the A(H1N1) virus due to immune pressure, possibly with glycosylation of key HA receptor binding sites, in circulating strains.3,22-24
The likely first exposure to influenza A virus for almost all individuals born between 1918 and 1957 (aged 52–91 years in 2009) was to an influenza A(H1N1) strain; for those born between 1957 and 1968 (aged 41–52) it was an influenza A(H2N2) strain; and for those born since the last pandemic in 1968 (aged < 41) it was an influenza A(H3N2) strain. The influenza A(H1N1) subtype reappeared in 1977 and co-circulated with A(H3N2) over the subsequent decades but did not replace the dominant A(H3N2) subtype.25
We suggest that the HAI antibody cross-reactivity against the pandemic (H1N1) 2009 virus in older individuals is due to prior infection caused by the 1918–1919 A(H1N1) pandemic virus or its immediate descendants, and that this immunity lasts for many decades. Most individuals born before 1925 had cross-reacting antibodies to the pandemic (H1N1) 2009 virus above the putative protective level. Even though there is a considerable gap in the available influenza viral isolates and HA sequences between the years 1918 and 1933, the estimated evolutionary speed of the virus would be at least 1% of HA1 amino acids showing changes per year.14 Seemingly, the mutation rate proceeded at such a pace that viruses circulating from the 1930s were already so different from the 1918 virus that cross-immunity was much reduced, helping to explain the large step-down in cross-protection in those born from the mid 1920s onwards.
Recent studies have shown that the pandemic (H1N1) 2009 HA gene is more closely related phylogenetically to the 1918 A(H1N1) virus and classic swine influenza A(H1N1) viruses than to recent seasonal human influenza A(H1N1) viruses.3 It is also antigenically similar to the 1918 A(H1N1) pandemic virus in terms of the immunodominant antibody response to HA.12,13,16 It seems likely that immunity induced by the 1918 A(H1N1) virus provides cross-protection against the pandemic (H1N1) 2009 virus, as shown by the much lower rates of severe influenza in older age groups during the first pandemic wave.26,27 This may or may not be a consequence of “original antigenic sin” (OAS).28 The doctrine of OAS states that the first infection with an influenza virus leaves a lifelong immunological imprint, reinforced by later infections with antigenically related strains. Our data showing high HAI antibody levels in those born in the few years before and after the 1918–1919 pandemic make it likely that almost all these people experienced 1918 A(H1N1) virus infection as their first influenza infection.
However, the observation that the older those born before the 1918–1919 pandemic were in 1919, the more frequently seropositive they were (proportions positive: 100% born before 1910, 67% born 1910–1914, and 54% born 1915–1919), suggests that older people may have more immunity due to more exposures to related A(H1N1) strains and does not obviously support the theory of OAS, as older children are more likely to have previously experienced a different seasonal influenza strain. Even so, some children may not experience influenza until school age, so the principle of OAS may still have operated. Perhaps this finding indicates that the HA was still highly immunogenic despite earlier influenza infection. As well, or alternatively, the ability to survive and mount a strong (probably lifelong) response to pandemic strains may be indicative of broader immunological resilience and higher likelihood of surviving to a great age. The greater level of protection in women (confounded by age, but not entirely) is intriguing and may reflect more intense exposure to children (who transmit influenza) or a sex-specific immunogenicity advantage in women.29
In Australia, 42% of people aged 65 years or older had received the monovalent pandemic (H1N1) 2009 vaccine by December 2009, whereas only 14% of adults aged under 65 years had been vaccinated.30 This is in stark contrast to where the clinical need was and is indicative of the importance of timely serosurveys, irrespective of which strain caused the pandemic.
Our study has several limitations. First, as an opportunistic study, we had to rely on limited sources of pre-pandemic serum samples from older people. Second, we could not collect information on the influenza vaccination history of the subjects, which might be important if there is any cross-reaction between the seasonal influenza strains and the pandemic virus. However, previous studies have shown there is little cross-reaction between pandemic (H1N1) 2009-specific HAI antibodies and antibodies to recent seasonal A(H1N1) strains.13,20 Some of our samples were collected during 2006–2008 seasonal influenza outbreak investigations in ACFs. However, all these outbreaks were caused by seasonal influenza A(H3N2) or B, so the possibility of a cross-reactive antibody response specific to our HAI test is unlikely (and not observed).
Our deductions are reflected in serological findings of HAI antibodies to pandemic (H1N1) 2009 virus in older individuals from other developed countries.12-16 Whether this is applicable to less developed countries needs to be determined. Seasonal influenza control and management policies concentrate mainly on high-risk populations, including those aged 65 years or older. Our findings show that pandemic influenza control measures should have focused more on younger adults who were naive to the pandemic virus and at increased risk of severe disease. A second wave may yet occur in southern hemisphere countries, especially as the pandemic (H1N1) 2009 virus displaced other circulating seasonal influenza strains in 2009,31 although the combined effects of high disease incidence in 2009 and moderately high vaccine uptake may curtail its impact considerably.
Received 3 June 2010, accepted 13 September 2010
- Robert Booy1
- Gulam Khandaker1
- Leon G Heron1
- Jiehui Yin1
- Bridget Doyle2
- Katherine K Tudo3
- Linda Hueston3
- Gwendolyn L Gilbert3
- C Raina MacIntyre4
- Dominic E Dwyer3
- 1 National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases, The Children’s Hospital at Westmead, Sydney, NSW.
- 2 Public Health Unit, Greater Southern Area Health Service, Albury, NSW.
- 3 Centre for Infectious Diseases and Microbiology, Institute of Clinical Pathology and Medical Research, Sydney, NSW.
- 4 School of Public Health and Community Medicine, Faculty of Medicine, University of New South Wales, Sydney, NSW.
*First two equal authors.
The antigen (influenza A/California/07/2009 virus) for HAI testing was kindly provided by the WHO Collaborating Centre for Reference and Research on Influenza, Melbourne. We thank Dr Jeremy McAnulty (Director of Communicable Diseases Branch, NSW Health), Professor Richard Lindley (Professor of Geriatric Medicine, Moran Foundation for Older Australians), all the residents and staff of the ACFs, and laboratory staff at the Centre for Infectious Diseases and Microbiology, Institute of Clinical Pathology and Medical Research. The “Economic and social benefits of treating and preventing influenza in aged care facilities” study in 2006–2008 was jointly funded by the Moran Health Care Group and a linkage grant from the Australian Research Council, together with Sanofi. The NSW Department of Health provided funding to the Centre for Infectious Diseases and Microbiology for the NSW serosurvey.
Robert Booy has received funding from CSL, Roche, Sanofi, GlaxoSmithKline (GSK) and Wyeth to attend and present at scientific meetings; any funding received is directed to a research account at the Children’s Hospital at Westmead. Leon Heron has performed consultancy work for Novartis for which payment was made to the National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases. He has had travel expenses covered by GSK and has conducted sponsored research and investigator-driven research with funding from GSK, Wyeth, Merck, CSL, Roche, and Sanofi Pasteur. Raina MacIntyre has received honararia from Merck, GSK and Wyeth for participation on pneumococcal and influenza advisory boards, as well as payment of travel expenses to attend advisory board meetings, and funding to her institution from GSK and CSL for investigator-driven research.
- 1. Shinde V, Bridges CB, Uyeki TM, et al. Triple-reassortant swine influenza A (H1) in humans in the United States, 2005–2009. N Engl J Med 2009; 360: 2616-2625.
- 2. Dawood FS, Jain S, Finelli L, et al; Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009; 360: 2605-2615.
- 3. Garten RJ, Davis CT, Russell CA, et al. Antigenic and genetic characteristics of swine-origin 2009 A(H1N1) influenza viruses circulating in humans. Science 2009; 325: 197-201.
- 4. Newman AP, Reisdorf E, Beinemann J, et al. Human case of swine influenza A (H1N1) triple reassortant virus infection, Wisconsin. Emerg Infect Dis 2008; 14: 1470-1472.
- 5. Simonsen L, Olson DR, Viboud C, et al. Pandemic influenza and mortality: past evidence and projections for the future. In: Knobler SL, Mack A, Mahmoud A, Lemon SM, editors. The threat of pandemic influenza: are we ready? Workshop summary. Washington, DC: National Academies Press, 2005: 89-114.
- 6. Andreasen V, Viboud C, Simonsen L. Epidemiologic characterization of the 1918 influenza pandemic summer wave in Copenhagen: implications for pandemic control strategies. J Infect Dis 2008; 197: 270-278.
- 7. Taubenberger JK, Morens DM. 1918 Influenza: the mother of all pandemics. Emerg Infect Dis 2006; 12: 15-22.
- 8. Morens DM, Fauci AS. The 1918 influenza pandemic: insights for the 21st century. J Infect Dis 2007; 195: 1018-1028.
- 9. Dowdle WR. Influenza A virus recycling revisited. Bull World Health Organ 1999; 77: 820-828.
- 10. Bautista E, Chotpitayasunondh T, Gao Z, et al; Writing Committee of the WHO Consultation on Clinical Aspects of Pandemic (H1N1) 2009 Influenza. Clinical aspects of pandemic 2009 influenza A (H1N1) virus infection. N Engl J Med 2010; 362: 1708-1719.
- 11. Girard MP, Tam JS, Assossou OM, Kieny MP. The 2009 A (H1N1) influenza virus pandemic: a review. Vaccine 2010; 28: 4895-4902.
- 12. Miller E, Hoschler K, Hardelid P, et al. Incidence of 2009 pandemic influenza A H1N1 infection in England: a cross-sectional serological study. Lancet 2010; 375: 1100-1108.
- 13. Hancock K, Veguilla V, Lu X, et al. Cross-reactive antibody responses to the 2009 pandemic H1N1 influenza virus. N Engl J Med 2009; 361: 1945-1952.
- 14. Ikonen N, Strengell M, Kinnunen L, et al. High frequency of cross-reacting antibodies against 2009 pandemic influenza A(H1N1) virus among the elderly in Finland. Euro Surveill 2010; 15: pii: 19478.
- 15. Rizzo C, Rota MC, Bella A, et al. Cross-reactive antibody responses to the 2009 A/H1N1v influenza virus in the Italian population in the pre-pandemic period. Vaccine 2010; 28: 3558-3562.
- 16. Itoh Y, Shinya K, Kiso M, et al. In vitro and in vivo characterization of new swine-origin H1N1 influenza viruses. Nature 2009; 460: 1021-1025.
- 17. Miller MA, Viboud C, Balinska M, Simonsen L. The signature features of influenza pandemics — implications for policy. N Engl J Med 2009; 360: 2595-2598.
- 18. Rosewell A, Chiu C, Lindley R, et al. Surveillance for outbreaks of influenza-like illness in the institutionalized elderly. Epidemiol Infect 2010; 138: 1126-1134.
- 19. Khandaker G, Doyle B, Dwyer DE, Booy R. Managing outbreaks of viral respiratory infection in aged care facilities — challenges and difficulties during the first pandemic wave [letter]. Med J Aust 2010; 192: 722. <MJA full text>
- 20. Gilbert GL, Cretikos MA, Hueston L, et al. Influenza A (H1N1) 2009 antibodies in residents of New South Wales, Australia, after the first pandemic wave in the 2009 southern hemisphere winter. PLoS One 2010; 5: e12562.
- 21. Hobson D, Curry RL, Beare AS, Ward-Gardner A. The role of serum haemagglutination-inhibiting antibody in protection against challenge infection with influenza A2 and B viruses. J Hyg (Lond) 1972; 70: 767-777.
- 22. Reichert T, Chowell G, Nishiura H, et al. Does glycosylation as a modifier of original antigenic sin explain the case age distribution and unusual toxicity in pandemic novel H1N1 influenza? BMC Infect Dis 2010; 10: 5.
- 23. Wei CJ, Boyington JC, Dai K, et al. Cross-neutralization of 1918 and 2009 influenza viruses: role of glycans in viral evolution and vaccine design. Sci Transl Med 2010; 2: 24ra21.
- 24. Xu R, Ekiert DC, Krause JC, et al. Structural basis of preexisting immunity to the 2009 H1N1 pandemic influenza virus. Science 2010; 328: 357-360.
- 25. Nelson MI, Viboud C, Simonsen L, et al. Multiple reassortment events in the evolutionary history of H1N1 influenza A virus since 1918. PLoS Pathog 2008; 4: e1000012.
- 26. Centers for Disease Control and Prevention. Update: novel influenza A (H1N1) virus infections — worldwide, May 6, 2009. MMWR Morb Mortal Wkly Rep 2009; 58: 453-458.
- 27. Australian Government Department of Health and Ageing. Australian influenza report 2009 – current report – 8 to 14 May 2010. http://www.health.gov.au/internet/main/publishing.nsf/Content/cda-ozflu-no19-10.htm (accessed Dec 2010).
- 28. Francis T Jr. On the doctrine of original antigenic sin. Proc Am Philos Soc 1960; 104: 572-578.
- 29. Klein SL, Jedlicka A, Pekosz A. The Xs and Y of immune responses to viral vaccines. Lancet Infect Dis 2010; 10: 338-349.
- 30. Australian Institute of Health and Welfare. 2009 Adult Vaccination Survey. Provisional topline results for H1N1 vaccination uptake. Canberra: AIHW, 8 Feb 2010. http://www.aihw.gov.au/pophealth/adult_vaccination_survey.cfm (accessed May 2010).
- 31. Blyth CC, Kelso A, McPhie KA, et al. The impact of the pandemic influenza A(H1N1) 2009 virus on seasonal influenza A viruses in the southern hemisphere, 2009. Euro Surveill 2010; 15: pii: 19631.





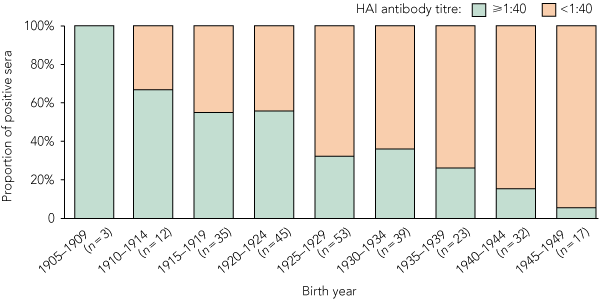
Abstract
Objective: To assess background pre-pandemic cross-reacting antibodies to the pandemic (H1N1) 2009 virus in older populations in Australia.
Design, setting and participants: Data were opportunistically generated from three cross-sectional pre-pandemic studies involving people aged 60 years or older: a 3-year (2006–2008) study of influenza outbreaks in aged care facilities (ACFs) in Sydney; an investigation of a respiratory virus outbreak in an ACF in rural New South Wales in June 2009; and a non-influenza serosurvey undertaken in NSW in 2007 and 2008.
Main outcome measure: Prevalence of pandemic (H1N1) 2009 haemagglutination inhibition (HAI) antibody titres ≥ 1:40 (putative protective level) in pre-pandemic sera.
Results: In total, 259 serum samples from individuals aged 60 years or older (range, 60–101 years) were tested. More than half of the individuals tested were women (151/259; 58.3%). About a third of individuals (37.5%) had cross-reacting HAI antibody titres ≥ 1:40. The prevalence of cross-reacting antibodies was highest in the oldest age groups (≥ 85 years), with more than 60% of these people having HAI antibody titres ≥ 1:40. The proportion of subjects with HAI antibody titres ≥ 1:40 decreased significantly and successively in younger groups to only 12% of those aged 60–64 years.
Conclusions: Our study suggests a pre-existing influenza A antibody reserve in most of the oldest group of people that was cross-reactive to the new pandemic (H1N1) 2009 virus; this is likely to be lifelong and to have provided them with clinical protection against the first wave of the pandemic. Pandemic influenza control measures need to focus more on younger adults naive to the pandemic virus and at increased risk of severe disease.