* The authors are members of the Australian Iron Deficiency Expert Group, comprising health professionals from diverse backgrounds with a shared interest in alleviating the burden and optimising management of iron deficiency anaemia (IDA) in Australia. Formation of the expert group was an initiative of the Australian and New Zealand Society of Blood Transfusion, supported by the Australian Red Cross Blood Service, New South Wales Clinical Excellence Commission (Blood Watch program), Victorian Department of Health (Blood Matters program) and South Australian Department of Health (BloodSafe program). During a meeting in May 2009, the expert group conducted a barrier analysis around improving the diagnosis and management of IDA in Australia. Many common clinical misconceptions were identified and thus developing a national education strategy was given high priority. A writing group was formed to provide a clinical update specific for the Australian setting.
Dietary iron comprises haem (animal sources) and non-haem iron (cereal and vegetable sources). Haem iron (bound to Hb and myoglobin) is better absorbed than non-haem iron. Non-haem iron is absorbed by intestinal luminal cells through a specific transporter (divalent metal transporter, located on the apical membrane of intestinal enterocytes), and released into the circulation where it binds to transferrin. Transferrin receptors on erythroblasts accept iron-transferrin complexes; these undergo endocytosis and the iron is incorporated into Hb. Although the specific mechanism of haem-iron absorption remains unclear, putative transporters have been identified.1
Iron absorption is upregulated by iron deficiency and increased erythropoiesis, and downregulated in inflammation and iron repletion, mediated by the recently described regulator of iron homeostasis, hepcidin, which blocks iron release from enterocytes and macrophages.2 Body iron stores are regulated through iron absorption. Non-haem iron is best absorbed in the ferrous form (Fe2+). Reduction of ferric iron (Fe3+) by stomach acid, dietary ascorbic acid and luminal reductases optimises absorption. Non-haem iron absorption is inhibited by simultaneous consumption of phytates (in cereals and legumes), tannins (in tea) and calcium. Simultaneous consumption of haem-iron sources and ascorbic acid enhances absorption. Less than 20% of available iron is absorbed in a typical Western diet, lower still from a vegetarian diet. Recommended daily intakes of iron at different stages of life are listed in Box 1.
Iron deficiency results when iron losses or requirements exceed absorption, and is often multifactorial. It is common in children during rapid growth and erythroid expansion, especially in premature or low birth weight babies, in toddlers and preschool children, and during adolescence. Pregnancy results in an overall additional iron requirement of about 1000 mg.3 Breastfeeding removes 1 mg of iron per day, but losses are mitigated by lactation-induced amenorrhoea if present. Blood loss is the most important cause of iron deficiency in adults. Each millilitre of blood loss (if Hb is 150 g/L) results in loss of around 0.5 mg of iron. Gastrointestinal (GI) blood loss is the most important cause in postmenopausal women and men. While menstrual blood loss commonly causes IDA in premenopausal women, coexistent GI lesions have often been identified. Malabsorption of iron may be caused by intestinal mucosal disorders (most frequently, coeliac disease), impaired gastric acid secretion (including use of proton pump inhibitors), and gastric/intestinal bypass procedures. Helicobacter pylori colonisation is also associated with IDA and may impair iron uptake and increase iron loss.4 Anaemia in endurance athletes (“sports anaemia”) has been associated with iron deficiency (due to reduced iron intake, impaired absorption caused by elevated hepcidin, GI bleeding and sweat losses), as well as mechanical haemolysis and haemodilution.5
IDA is associated with impaired cognitive development in preschool-aged children and diminished work productivity and cognitive and behavioural problems in adults.6,7 Among pregnant women, IDA has been associated with increased risks of low birth weight, prematurity and maternal morbidity.8 Non-anaemic tissue iron deficiency may cause impairment in both adults9 and children.6 In a recent randomised trial, patients with chronic heart failure and iron deficiency (both with and without anaemia) treated with intravenous (IV) iron carboxymaltose experienced improvements in symptoms, functional capacity and quality of life, independent of Hb concentrations.10
Few studies have reported the prevalence of iron deficiency or anaemia in the Australian population. Among toddlers, studies in Sydney and Adelaide suggest IDA prevalence ranges from 1%–6%, and up to 14% in Asian groups.11-13 Other studies show the prevalence of anaemia (all causes) among non-pregnant young women to be 10%,14 and 11% in pregnant women not taking iron supplements.15 Anaemia is highly prevalent in Indigenous communities; a study in an Aboriginal community of Western Australia identified anaemia among 55% of women and 18% of men.16 United States data show that 11% of men and 10% of women older than 65 years have anaemia, with iron deficiency accounting for 20%.17 Other high-risk groups include blood donors, where the prevalence of iron deficiency among females exceeds 20%.18
Anaemia is defined as a Hb concentration below the reference range (specific for age, sex and gestation) for the laboratory performing the test. The WHO defines anaemia as a Hb level below 130 g/L in men, 120 g/L in women, and 110 g/L in pregnant women and preschool children.19
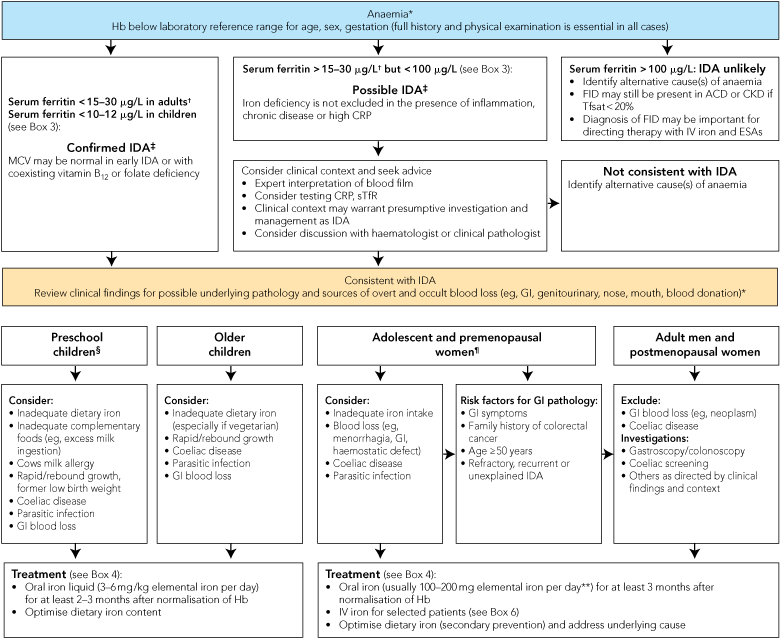
Iron deficiency should be distinguished from other causes of anaemia because of its associations with underlying conditions that mandate specific investigation, and because treatment is simple, safe and effective. Anaemia with low mean corpuscular volume (MCV) or mean corpuscular haemoglobin (MCH) is likely to be due to iron deficiency; the important differential diagnoses are thalassaemic conditions, which should be excluded as clinically appropriate. A strategy for assessment and management of IDA is outlined in Box 2.
Patients with suspected iron deficiency should have iron studies performed and the results correlated with red cell indices. Box 3 provides guidance for interpretation of results of laboratory tests of iron status. The serum ferritin level is the most readily available and useful index of iron deficiency.22 In an anaemic adult, a ferritin level below 15 g/L is diagnostic of iron deficiency, and levels between 15 and 30 g/L are highly suggestive. Lower thresholds (from 10 to 12 g/L) have been used for children.19,20 However, ferritin is also an acute-phase protein and is elevated in inflammation, infection, liver disease and malignancy. This can result in misleadingly elevated ferritin levels in iron-deficient patients with coexisting systemic illness. In the elderly or among patients with inflammation, iron deficiency may still be present with ferritin values up to 60–100 g/L.23,24 Markers such as C-reactive protein may help identify coexisting inflammation. Serum iron levels have significant diurnal variation, are low in both iron deficiency and inflammation, and should not be used to diagnose iron deficiency.
Functional iron deficiency (FID) exists when, despite adequate stores, iron cannot be mobilised for erythropoiesis, mediated by elevated hepcidin. It is commonly seen in patients with end-stage kidney disease, whose response to erythropoietin-stimulating agents may be optimised when ferritin levels exceed 200 g/mL.25 FID may also contribute to anaemia in patients with inflammatory diseases such as rheumatoid arthritis. Percentage hypochromic red cells and reticulocyte haemoglobin content (available on some automated haematology analysers) have been used to assess iron status in patients receiving erythropoietin-stimulating agents and may be a useful indicator of FID.26,27
An additional iron index is the soluble transferrin receptor (sTfR) concentration, which is elevated in tissue iron deficiency and not sensitive to inflammation; the sTfR/log ferritin ratio is highly correlated with body iron stores.28 However, sTfR estimation is limited by variability in interassay cut-offs, availability and slow turnaround in some laboratories. Bone marrow examination remains the gold-standard investigation of IDA in complex cases, although it can usually be avoided. Hepcidin may emerge as a useful aid for assessment of iron status. Where the diagnosis is unclear, clinicians should seek guidance from their pathologist or another relevant expert.
A recommended approach to assessment and management of IDA is outlined in Box 2. The underlying cause must be established in all patients. Importantly, in men and postmenopausal women with IDA, and also in some premenopausal women, evaluation for benign or malignant GI lesions, inflammatory conditions (such as inflammatory bowel disease) and peptic ulceration is indicated. Screening for coeliac disease should be performed.4,29
Increasing dietary iron intake alone is inadequate to treat frank IDA and higher supplemental doses of iron are required (Box 4). However, increasing iron consumption and optimising absorption by minimising inhibitors and maximising enhancers may be valuable for secondary prevention of iron deficiency. Recommended daily dietary intake of iron at different ages is presented in Box 1.
Although more than 100 preparations containing iron are available over the counter in Australia, few contain sufficient elemental iron to treat IDA effectively. The usual recommended dose of oral iron for the treatment of IDA in adults is 100–200 mg of elemental iron daily or in 2 to 3 divided doses;30 recommended doses for children are 3–6 mg/kg/day of elemental iron.20,31 Appropriate formulations are presented in Box 4. Multivitamin-mineral supplements should not be used to treat IDA as iron content is low and absorption may be reduced.
Studies in women and children have shown lower doses of oral iron may be effective and associated with fewer GI side effects.32 In adults, lower doses can be administered through either intermediate-dose tablets (containing around 30–60 mg of elemental iron) or intermittent dosing (eg, second daily to weekly); the latter approach has been recommended by the WHO for some developing countries. Either approach may be useful in patients with mild IDA who experience GI upset with standard doses of iron, but rapid Hb rise is not essential; however, appropriate intermediate-dose iron tablets are not available in Australia. GI upset may also be reduced by taking the dose with food or at night.
When given at equivalent elemental iron doses, different oral iron salts have similar efficacy and tolerability. Based on limited available data, controlled-release iron formulations appear to have fewer GI side effects, but similar discontinuation rates and comparable efficacy;33 release of iron distal to the site of maximal intestinal absorption may theoretically limit response in some patients.
After therapeutic doses of oral iron, reticulocytosis should occur within 72 hours, and Hb levels should rise by about 20 g/L every 3 weeks. It is reasonable to replenish iron stores by continuing treatment for 3–6 months (2–3 months in children20) beyond normalisation of Hb. Causes of failure to respond to oral iron are listed in Box 5. If IDA recurs following repletion of iron stores, a bleeding source must be vigorously sought.
Although IM injection of iron is effective, it is painful, associated with permanent skin staining and no safer than IV infusion. Its use is therefore discouraged34 unless other approaches cannot be practically delivered (eg, when parenteral iron is indicated in remote settings).
While oral iron remains the cornerstone of IDA therapy, some patients require IV iron therapy (Box 6). Underuse of IV iron may have stemmed in part from concerns about the risk of serious allergic reactions — for example, 0.6% of patients treated with high molecular weight iron dextran (no longer available in Australia) had life-threatening allergic reactions.35 Iron polymaltose (Ferrum H [Aspen Pharmacare], Ferrosig [Sigma Pharmaceuticals]) and iron sucrose (Venofer [Aspen Pharmacare]) are the parenteral iron formulations currently available in Australia. A “total-dose” infusion (where iron stores can be repleted in a single treatment episode) can be administered only with iron polymaltose.
Although iron polymaltose (iron dextrin) has been widely used in Australia since the 1960s, there is limited literature concerning its safety, particularly outside the nephrology setting. An Australian audit of the inhospital safety and tolerability of iron polymaltose identified no cases of anaphylaxis or other cardiorespiratory compromise in 401 infusions, and noted infrequent minor side effects during infusion.36 This concurs with the clinical experience of several members of our group. Self-limited side effects that occur up to 2 days after infusion, including headache, fever and arthralgias, are common, affecting 26% of patients in a recent study.37
An average total-dose infusion of iron polymaltose (sufficient to replenish iron stores, commonly 1000 mg–2500 mg for adults), given at the rates recommended in Australian approved product information, takes around 5 hours. A recent survey of policies at different New South Wales health services found a broad range of infusion and premedication practices.38 Few published data indicate the fastest safe rate for either total-dose infusions or smaller dose infusions given in the renal setting (500–1000 mg or less). Several Australian nephrology units have developed protocols and used accelerated infusion rates (Rowan Walker, Deputy Director of Nephrology, Royal Melbourne Hospital, Victoria, and Ashley Irish, Senior Staff Nephrologist, Royal Perth Hospital, Western Australia, personal communications). Delivery of total-dose iron polymaltose infusions at accelerated rates have been safely administered by some of the authors (S C F-B, R T, P R G) in specialist outpatient treatment centres, with and without routine premedication. Accounts of this experience should be published through prospective clinical studies in Australia and New Zealand because use of iron polymaltose outside these countries is limited. We are not aware of accelerated administration of iron polymaltose in children.
Iron sucrose is funded by the Pharmaceutical Benefits Scheme for iron deficiency only in patients with chronic kidney disease who are having an erythropoietin-stimulating agent and react systemically to iron polymaltose. However, it has been licensed for treatment of IDA in Europe for decades and has a well-documented safety profile.39 It cannot be given as a single total-dose infusion (due to the rate of release of vasoactive iron), but is instead given as multiple smaller (commonly 100–200 mg) IV doses. Despite this limitation, it has the potential to provide a flexible alternative to iron polymaltose, being licensed for use as a “slow IV injection” in many countries outside Australia, including the United Kingdom and NZ. Iron sucrose may be useful to partially replace iron stores, helping to ensure more rapid increases in Hb levels when clinically important. Iron stores can then be fully replaced with oral therapy (if tolerated), with IV iron polymaltose or further doses of IV iron sucrose. Some hospital drug committees have approved off-label use of iron sucrose for IDA, including perioperative use.
There are a number of new IV iron preparations. Iron carboxymaltose, which is available in Europe but has not yet been granted marketing approval in Australia, appears to have a low risk of serious adverse effects, and infusions of 1000 mg can be given over 15 minutes.40 Other preparations available internationally include low molecular weight iron dextrans, iron gluconate and ferumoxytol.
Transfusion of red cells remains an overused treatment for IDA.41 It is also expensive and potentially hazardous. In physiologically compensated patients, transfusion carries unnecessary risks and fails to replenish deficient iron stores. Adequate doses of oral iron can improve Hb concentrations within a few weeks, and IV iron can provide more rapid and predictable increases when clinically important. Transfusion is associated with adverse outcomes, including fluid overload (around 1% of patients), and a range of immunological and infectious hazards. Hence, it should be reserved for immediate, targeted management in patients with severe anaemia compromising end-organ function (eg, angina pectoris or cardiac failure) or where IDA is complicated by serious, acute ongoing bleeding. Iron therapy should always follow transfusion to replenish iron stores.
2 Iron deficiency anaemia (IDA): assessment and management*
3 Interpreting laboratory blood test results to assess iron status*
Normal or elevated (elevated ferritin does not imply elevated iron stores) |
|||||||||||||||
4 Commercially available forms of iron therapy in Australia suitable for the treatment of iron deficiency anaemia (IDA)*
Oral formulation for children§ (or adults‡ requiring a liquid) |
|||||||||||||||
FERRUM H (Aspen Pharmacare) FERROSIG (Sigma Pharmaceuticals) |
|||||||||||||||
5 Major reasons for inadequate response to oral iron therapy*
Inadequate iron intake:
Patient not taking oral iron therapy
Patient taking an iron supplement or multivitamin tablet with insufficient iron content
Inadequate iron absorption:
Concomitant consumption of inhibitors of iron absorption (eg, tea, calcium, antacids, tetracycline, within 2 hours of iron ingestion)
Coexisting inflammation with functional iron deficiency
Intestinal mucosal disorders (eg, coeliac disease, inflammatory bowel disease)
Impaired gastric acid secretion (including use of proton pump inhibitors)
Gastric/intestinal bypass procedures
Helicobacter pylori colonisation
Controlled-release iron formulations may contribute (ie, potential for limited iron absorption in some patients)†
Ongoing iron losses or need in excess of dose absorbed:
Occult, undiagnosed or recurrent gastrointestinal blood loss (eg, peptic ulcer, malignancy, angiodysplasia, small bowel lesion, parasites)
Other source of recurrent blood loss (eg, menorrhagia due to uterine pathology or an inherited bleeding disorder such as von Willebrand disorder)
Multiple sources of recurrent blood loss (eg, hereditary haemorrhagic telangiectasia)
Ongoing urinary iron losses (eg, significant valve haemolysis)
Renal failure responding to erythropoietin-stimulating agents
Coexisting condition interfering with bone marrow response:
Superimposed infection, inflammation, malignancy or renal failure
Concomitant B12 or folate deficiency
Coexisting primary bone marrow disease or suppression
Incorrect diagnosis or more than one cause of anaemia:
Anaemia of chronic disease or renal failure
Haemoglobinopathy
Other causes of anaemia (eg, haemolysis, myelodysplastic syndromes, congenital anaemia, endocrine disorders)
* More than one factor is often present. † Role is unclear (limited available data show efficacy comparable to that of non-controlled-release formulations33).
6 Indications for intravenous (IV) iron therapy in patients with iron deficiency anaemia (IDA)
IV iron should be considered in patients with confirmed IDA* and one or more of the following:
Demonstrated intolerance, non-compliance or lack of efficacy with oral iron, despite modification of dose, timing and frequency;
Pregnancy (beyond the first trimester) and postpartum, for the above reasons or to avoid imminent decompensation/transfusion (eg, in women who present late and/or display severe anaemia);
Intestinal malabsorption (eg, inflammatory bowel disease);
Ongoing iron (ie, blood) losses that exceed absorptive capacity;
A clinical need for a rapid iron supply (ie, in patients where optimisation of erythroid response is important to prevent physiological decompensation/ transfusion);
Chronic renal impairment receiving concomitant erythropoietin-stimulating agent therapy.
* Prescribed in consultation with an expert in the use of IV iron and the relevant patient group.
- Sant-Rayn S Pasricha1,2
- Stephen C Flecknoe-Brown4
- Katrina J Allen6
- Peter R Gibson8
- Lawrence P McMahon5
- John K Olynyk11,12,13
- Simon D Roger14
- Helen F Savoia15
- Ramdas Tampi16
- Amanda R Thomson18
- Erica M Wood1,19
- Kathryn L Robinson20,21
- 1 Royal Melbourne Hospital, Melbourne, VIC.
- 2 Australian Red Cross Blood Service, Melbourne, VIC.
- 3 Broken Hill Health Service, Broken Hill, NSW.
- 4 University Department of Rural Health, School of Public Health, Sydney Medical School, Sydney, NSW.
- 5 Department of Paediatrics, University of Melbourne, Royal Children’s Hospital, Melbourne, VIC.
- 6 Gut and Liver Research Group, Infection, Immunity and Environment, Murdoch Childrens Research Institute, Melbourne, VIC.
- 7 Monash University, Melbourne, VIC.
- 8 Box Hill Hospital, Melbourne, VIC.
- 9 Eastern Health, Melbourne, VIC.
- 10 Department of Gastroenterology, Fremantle Hospital, Fremantle, WA.
- 11 University of Western Australia, Perth, WA.
- 12 Curtin Health Innovation Research Institute, Curtin University, Perth, WA.
- 13 Western Australian Institute of Medical Research, Perth, WA.
- 14 Gosford Hospital, Gosford, NSW.
- 15 Royal Children's Hospital, Melbourne, VIC.
- 16 Clinipath Pathology, Perth, WA.
- 17 Royal North Shore Hospital, Sydney, NSW.
- 18 Australian Red Cross Blood Service, Sydney, NSW.
- 19 Department of Clinical Haematology, Faculty of Medicine, Nursing and Health Sciences, Monash University, Melbourne, VIC.
- 20 Australian Red Cross Blood Service, Adelaide, SA.
- 21 The Queen Elizabeth Hospital, Adelaide, SA.
Katrina Allen gave a presentation at an educational forum on iron therapy organised by Aspen Pharmacare in February 2008 and received an honorarium for her time. She received reimbursement for local travel expenses. (Aspen are the Australian sponsors for iron sucrose and iron polymaltose on behalf of Vifor International.) Peter Gibson has received honararia from Aspen Pharmacare for contributing to educational meetings. His institution has received a grant from Vifor International for an investigator-initiated study. Simon Roger has received money for board membership of, and consultancy for, Amgen, Roche, Vifor, Janssen-Cilag and Sandoz. He has also received honoraria, payment for educational presentations and reimbursement for travel expenses from these companies. Kathryn Robinson had an interstate airfare and accommodation for one night paid directly by Aspen Pharmacare for presenting at an educational iron forum organised by Aspen in February 2008; information from her presentation was used for an Aspen educational newsletter but no payment was received.
The Australian Iron Deficiency Expert group received funding for a meeting in May 2009 from the Australian and New Zealand Society of Blood Transfusion, the Australian Red Cross Blood Service, the South Australian Department of Health, the Victorian Department of Health and the NSW Clinical Excellence Commission. The funding was used to pay for the venue and for travel and accommodation of those participants not funded by the institutions they worked for. The participants included clinicians from public hospitals and private practice, and representatives from the Australian Red Cross Blood Service, National Institute of Clinical Studies, National Prescribing Service, National Blood Authority, Therapeutic Goods Authority, and Australian Safety and Quality Commission. No funding from industry was received by the Expert group and no industry representatives were present at the meeting. Three international clinicians attending the Expert meeting were in Australia (funded by Vifor International) for a meeting organised by the WA Department of Health Blood Management Program — the Expert group meeting was organised back-to-back with this meeting to take advantage of their visit.
- 1. West AR, Oates PS. Mechanisms of heme iron absorption: current questions and controversies. World J Gastroenterol 2008; 14: 4101-4110.
- 2. Ganz T. Hepcidin — a regulator of intestinal iron absorption and iron recycling by macrophages. Best Pract Res Clin Haematol 2005; 18: 171-182.
- 3. Bothwell TH. Iron requirements in pregnancy and strategies to meet them. Am J Clin Nutr 2000; 72 (1 Suppl): 257S-264S.
- 4. Goddard AF, James MW, McIntyre AS, Scott BB. Guidelines for the management of iron deficiency anaemia. British Society of Gastroenterology, 2005. http://www.bsg.org.uk/images/stories/docs/clinical/guidelines/sbn/iron_def.pdf (accessed Sep 2010).
- 5. Peeling P, Dawson B, Goodman C, et al. Athletic induced iron deficiency: new insights into the role of inflammation, cytokines and hormones. Eur J Appl Physiol 2008; 103: 381-391.
- 6. Sachdev H, Gera T, Nestel P. Effect of iron supplementation on mental and motor development in children: systematic review of randomised controlled trials. Public Health Nutr 2005; 8: 117-132.
- 7. Murray-Kolb LE, Beard JL. Iron treatment normalizes cognitive functioning in young women. Am J Clin Nutr 2007; 85: 778-787.
- 8. Allen LH. Anemia and iron deficiency: effects on pregnancy outcome. Am J Clin Nutr 2000; 71 (5 Suppl): 1280S-1284S.
- 9. Brownlie T, Utermohlen V, Hinton PS, et al. Marginal iron deficiency without anemia impairs aerobic adaptation among previously untrained women. Am J Clin Nutr 2002; 75: 734-742.
- 10. Anker SD, Comin Colet J, Filippatos G, et al. Ferric carboxymaltose in patients with heart failure and iron deficiency. N Engl J Med 2009; 361: 2436-2448.
- 11. Oti-Boateng P, Seshadri R, Petrick S, et al. Iron status and dietary iron intake of 6–24-month-old children in Adelaide. J Paediatr Child Health 1998; 34: 250-253.
- 12. Karr MA, Mira M, Alperstein G, et al. Iron deficiency in Australian-born children of Arabic background in central Sydney. Med J Aust 2001; 174: 165-168. <MJA full text>
- 13. Karr M, Alperstein G, Causer J, et al. Iron status and anaemia in preschool children in Sydney. Aust N Z J Public Health 1996; 20: 618-622.
- 14. Rangan A, Aitkin I, Blight G, et al. Factors affecting iron status in 15–30 year old female students. Asia Pac J Clin Nutr 1997; 6: 291-295.
- 15. Zhou SJ, Gibson RA, Crowther CA, et al. Effect of iron supplementation during pregnancy on the intelligence quotient and behavior of children at 4 y of age: long-term follow-up of a randomized controlled trial. Am J Clin Nutr 2006; 83: 1112-1117.
- 16. Hopkins RM, Gracey MS, Hobbs RP, et al. The prevalence of hookworm infection, iron deficiency and anaemia in an Aboriginal community in north-west Australia. Med J Aust 1997; 166: 241-244.
- 17. Guralnik JM, Eisenstaedt RS, Ferrucci L, et al. Prevalence of anemia in persons 65 years and older in the United States: evidence for a high rate of unexplained anemia. Blood 2004; 104: 2263-2268.
- 18. Badami KG, Taylor K. Iron status and risk-profiling for deficiency in New Zealand blood donors. N Z Med J 2008; 121 (1274): 50-60.
- 19. World Health Organization. United Nations Children’s Fund. United Nations University. Iron deficiency anaemia: assessment, prevention, and control. A guide for programme managers. Geneva: WHO, 2001. http://www.who.int/nutrition/publications/en/ida_assessment_prevention_control.pdf (accessed Sep 2010).
- 20. Grant CC, Wall CR, Brewster D, et al. Policy statement on iron deficiency in pre-school-aged children. J Paediatr Child Health 2007; 43: 513-521.
- 21. Royal Australian and New Zealand College of Obsteticians and Gynaecologists. College Statement: vitamin and mineral supplements in pregnancy. RANZCOG, 2008. http://www.ranzcog.edu.au/publications/statements/C-obs25.pdf (accessed Sep 2010).
- 22. Guyatt GH, Oxman AD, Ali M, et al. Laboratory diagnosis of iron-deficiency anemia: an overview. J Gen Intern Med 1992; 7: 145-153.
- 23. Guyatt GH, Patterson C, Ali M, et al. Diagnosis of iron-deficiency anemia in the elderly. Am J Med 1990; 88: 205-209.
- 24. Hansen TM, Hansen NE. Serum ferritin as indicator of iron responsive anaemia in patients with rheumatoid arthritis. Ann Rheum Dis 1986; 45: 596-602.
- 25. Besarab A, Amin N, Ahsan M, et al. Optimization of epoetin therapy with intravenous iron therapy in hemodialysis patients. J Am Soc Nephrol 2000; 11: 530-538.
- 26. Jones CH, Richardson D, Ayers S, et al. Percentage hypochromic red cells and the response to intravenous iron therapy in anaemic haemodialysis patients. Nephrol Dial Transplant 1998; 13: 2873-2876.
- 27. Mast AE, Blinder MA, Dietzen DJ. Reticulocyte hemoglobin content. Am J Hematol 2008; 83: 307-310.
- 28. Suominen P, Punnonen K, Rajamaki A, Irjala, K. Serum transferrin receptor and transferrin receptor-ferritin index identify healthy subjects with subclinical iron deficits. Blood 1998; 92: 2934-2939.
- 29. Corazza GR, Valentini RA, Andreani ML, et al. Subclinical coeliac disease is a frequent cause of iron-deficiency anaemia. Scand J Gastroenterol 1995; 30: 153-156.
- 30. Beutler E. Disorders of iron metabolism. In: Lichtman MA, Williams WJ, Beutler E, et al, editors. Williams hematology. 7th ed. New York: McGraw-Hill Medical, 2006: 511-553.
- 31. Department of General Medicine, Royal Children’s Hospital. Oral iron preparations — iron supplementation. Melbourne: Department of General Medicine, Royal Children’s Hospital, 2009. http://www.rch.org.au/genmed/clinical.cfm?doc_id=7883 (accessed Sep 2010).
- 32. Zhou SJ, Gibson RA, Crowther CA, et al. Should we lower the dose of iron when treating anaemia in pregnancy? A randomized dose-response trial. Eur J Clin Nutr 2009; 63: 183-190.
- 33. Zumberg MS, Kahn MJ. Acquired anemias: iron deficiency, cobalamin deficiency, and autoimmune hemolytic anemia. In: Crowther MA, Ginsberg J, Schunemann HJ, et al, editors. Evidence based hematology. Malden MA: Blackwell Publishing, 2008: 197-205.
- 34. Solomons NW, Schumann K. Intramuscular administration of iron dextran is inappropriate for treatment of moderate pregnancy anemia, both in intervention research on underprivileged women and in routine prenatal care provided by public health services. Am J Clin Nutr 2004; 79: 1-3.
- 35. Hamstra RD, Block MH, Schocket AL. Intravenous iron dextran in clinical medicine. JAMA 1980; 243: 1726-1731.
- 36. Newnham E, Ahmad I, Thornton A, et al. Safety of iron polymaltose given as a total dose iron infusion. Intern Med J 2006; 36: 672-674.
- 37. Haines ML, Gibson PR. Delayed adverse reactions to total-dose intravenous iron polymaltose. Intern Med J 2009; 39: 252-255.
- 38. NSW Therepeutic Advisory Group. Iron polymaltose infusions discussion paper: rapid response to iron polymaltose infusions email discussion. NSWtag, 2008. http://www.ciap.health.nsw.gov.au/nswtag/publications/otherdocs/IronPolymaltosediscussionpaper1208.pdf (accessed Sep 2010).
- 39. Chertow GM, Mason PD, Vaage-Nilsen O, et al. Update on adverse drug events associated with parenteral iron. Nephrol Dial Transplant 2006; 21: 378-382.
- 40. Medicines and Healthcare Products Regulatory Agency. Ferinject 50mg iron/ml solution for injection/infusion. Public assessment report — decentralised procedure. MHPRA, 2007. http://www.mhra.gov.uk/home/groups/l-unit1/documents/websiteresources/con014025.pdf (accessed Sep 2010).
- 41. Grey DE, Finlayson J. Red cell transfusion for iron-deficiency anaemia: a retrospective audit at a tertiary hospital. Vox Sang 2008; 94: 138-142.





Abstract
Iron deficiency anaemia (IDA) remains prevalent in Australia and worldwide, especially among high-risk groups.
IDA may be effectively diagnosed in most cases by full blood examination and serum ferritin level. Serum iron levels should not be used to diagnose iron deficiency.
Although iron deficiency may be due to physiological demands in growing children, adolescents and pregnant women, the underlying cause(s) should be sought.
Patients without a clear physiological explanation for iron deficiency (especially men and postmenopausal women) should be evaluated by gastroscopy/colonoscopy to exclude a source of gastrointestinal bleeding, particularly a malignant lesion.
Patients with IDA should be assessed for coeliac disease.
Oral iron therapy, in appropriate doses and for a sufficient duration, is an effective first-line strategy for most patients.
In selected patients for whom intravenous (IV) iron therapy is indicated, current formulations can be safely administered in outpatient treatment centres and are relatively inexpensive.
Red cell transfusion is inappropriate therapy for IDA unless an immediate increase in oxygen delivery is required, such as when the patient is experiencing end-organ compromise (eg, angina pectoris or cardiac failure), or IDA is complicated by serious, acute ongoing bleeding.
Consensus methods for administration of available IV iron products are needed to improve the utilisation of these formulations in Australia and reduce inappropriate transfusion.
New-generation IV products, supported by high-quality evidence of safety and efficacy, may facilitate rapid administration of higher doses of iron, and may make it easier to integrate IV iron replacement into routine care.