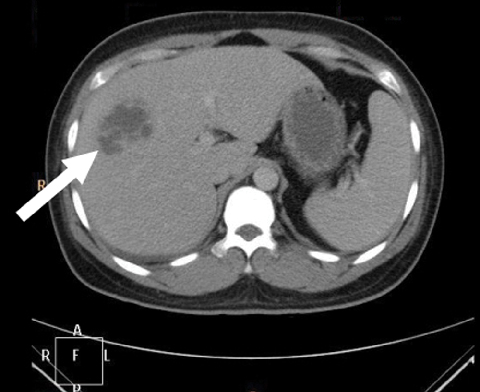
Liver abscess due to Klebsiella pneumoniae infection has been widely reported in Asia, but rarely reported in Australia until now. We describe four previously well Asian-born patients who presented across Australia with community-acquired K. pneumoniae liver abscesses. With prompt recognition, appropriate antibiotics and early drainage, outcome is significantly improved, although vigilance for metastatic complications is essential.
A community-acquired Klebsiella pneumoniae primary invasive liver abscess syndrome has been recognised in Asia for more than 20 years, with almost 1000 reported presentations published by 2008; it has been reported less frequently in other regions.1 K. pneumoniae infection accounted for over 80% of primary liver abscesses reported from Taiwan in the 1990s.2 Increasingly, cases have been seen outside Asia, primarily among patients of Asian ethnicity, including in the United States.1,3 It has only rarely been reported in Australia until now.4,5
Of interest are an absence of prior hepatobiliary disease, an association with diabetes, and a risk of metastatic spread.6 Community-acquired KPLA has been associated with severe metastatic complications, including endophthalmitis.
The reasons for the changing epidemiology away from Escherichia coli as the leading cause of pyogenic liver abscess are unclear, although selective pressure for Klebsiella through widespread amoxicillin use, to which it is almost universally resistant, has been postulated.7 A genetic predisposition is possible, given the disease is seen almost exclusively in patients of Asian ethnicity, even outside Asia, and very rarely in those of Caucasian origin.8,9
K. pneumoniae is frequently found as part of normal faecal flora, and spread to the liver is thought to occur from the intestines via the portal system.1 Ordinarily, any bacteria reaching the liver would then be phagocytosed and killed, and failure of this defence is presumed to lead to the formation of liver abscesses.1 Diabetes was present in about 50%–70% of patients reported from Taiwan,6 presumably conferring susceptibility by impairing neutrophil-mediated defence,10 and this also appears to be a risk factor for metastatic complications.6 It is interesting to note that none of the patients in our small sample had diabetes.
Bacterial virulence is also of major importance, as the condition often affects previously healthy individuals. The presence of capsular polysaccharides of K. pneumoniae serotype K1 or K2 has been strongly associated with virulence through resistance to phagocytosis;10,11 our patients were all infected with one of these two serotypes.
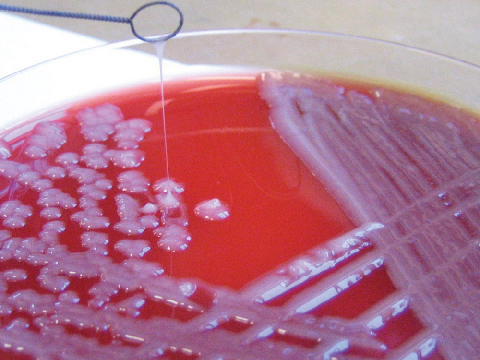
Commonly, K. pneumoniae strains causing liver abscess are hypermucoviscous, as defined by an unusual, highly mucoid colony appearance on culture, a feature strongly associated with the K1 or K2 serotype.11 This stickiness is the basis of the “string test”, which can be performed easily in the laboratory. The string test is a quick, useful investigation in this setting (Box 3).7 A colony that stretches more than 5 mm using a standard inoculation loop tests positive for hypermucovisosity.3
The geographical distribution of KPLA may be explained by the finding that K. pneumoniae isolates from Taiwan were far more likely to have a hypermucoviscous phenotype and to belong to K1 or K2 serotypes than those in other countries except South Africa, where invasive disease is also seen.12 Indeed, of the many K. pneumoniae capsular serotypes isolated from patients in an Australian tertiary hospital inpatient setting, K1 and K2 accounted for only 10 of 293 (3.5%) presentations.13
All our patients had recently been in Asia, which raises the possibility of exposure to these virulent strains of the organism. However, case reports from the US have involved emigrants from Vietnam and Korea who had not travelled home for some years.1
A third-generation cephalosporin such as ceftriaxone is usually an effective treatment, with good penetration of vitreous fluid and cerebrospinal fluid, allowing it to reach metastatic lesions in these locations.12 In the case of endophthalmitis, systemic antibiotics should be combined with intravitreal injections. Treatment is required until clinical state, biochemistry and radiology indicate resolution, often requiring antibiotics for 4 to 6 weeks.
Metastatic spread not uncommonly complicates KPLA; reports from Taiwan estimate the frequency of this at between 3.5% and 20%.12,14 Endophthalmitis, lung abscesses and meningitis are the more common complications. Ophthalmological and other organ review is therefore indicated when KPLA is diagnosed. Visual recovery in patients with endophthalmitis is often poor; a high index of suspicion and early intervention before visual changes are noted may improve outcome.
Response to antibiotics and drainage is generally good. In contrast with patients with underlying biliary tract disease, long-term recurrence rates in patients with spontaneously occurring liver abscess appear to be low.15
1 Clinical and microbiological details of four patients with community-acquired Klebsiella pneumoniae liver abscesses
- 1. McIver CJ, Janda JM. Pathogenesis and laboratory identification of emerging hepatovirulent Klebsiella pneumoniae. Clin Microbiol Newsl 2008; 30: 127-131.
- 2. Lau YJ, Hu BS, Wu WL, et al. Identification of a major cluster of Klebsiella pneumoniae isolates from patients with liver abscess in Taiwan. J Clin Microbiol 2000; 38: 412-414.
- 3. Lederman ER, Crum NF. Pyogenic liver abscess with a focus on Klebsiella pneumoniae as a primary pathogen: an emerging disease with unique clinical characteristics. Am J Gastroenterol 2005; 100: 322-331.
- 4. Lindstrom ST, Healey PR, Chen SC. Metastatic septic endophthalmitis complicating pyogenic liver abscess caused by Klebsiella pneumonia. Aust N Z J Med 1997; 27: 77.
- 5. Cheng AC, Johnson DF. Multiloculated liver abscesses. Clin Infect Dis 2006; 43: 264.
- 6. Wang JH, Liu YC, Lee SS, et al. Primary liver abscess due to Klebsiella pneumoniae in Taiwan. Clin Infect Dis 1998; 26: 1434-1438.
- 7. Fang CT, Chuang YP, Shun CT, et al. A novel virulence gene in Klebsiella pneumoniae strains causing primary liver abscess and septic metastatic complications. J Exp Med 2004; 199: 697-705.
- 8. Braiteh F, Golden MP. Cryptogenic invasive Klebsiella pneumoniae liver abscess syndrome. Int J Infect Dis 2007; 11: 16-22.
- 9. Keynan Y, Rubinstein E. The changing face of Klebsiella pneumoniae infections in the community. Int J Antimicrob Agents 2007; 30: 385-389.
- 10. Lin JC, Siu LK, Fung CP, et al. Impaired phagocytosis of capsular serotypes K1 or K2 Klebsiella pneumoniae in type 2 diabetes mellitus patients with poor glycemic control. J Clin Endocrinol Metab 2006; 91: 3084-3087.
- 11. Lin JC, Chang FY, Fung CP, et al. High prevalence of phagocytic-resistant capsular serotypes of Klebsiella pneumoniae in liver abscess. Microbes Infect 2004; 6: 1191-1198.
- 12. Yu VL, Hansen DS, Ko WC, et al; International Klebsiella Study Group. Virulence characteristics of Klebsiella and clinical manifestations of K. pneumoniae bloodstream infections. Emerg Infect Dis 2007; 13: 986-993.
- 13. Jenney AW, Clements A, Farn JL, et al. Seroepidemiology of Klebsiella pneumoniae in an Australian tertiary hospital and its implications for vaccine development. J Clin Microbiol 2006; 44: 102-107.
- 14. Lee SS, Chen YS, Tsai HC, et al. Predictors of septic metastatic infection and mortality among patients with Klebsiella pneumoniae liver abscess. Clin Infect Dis 2008; 47: 642-650.
- 15. Cheng HC, Chang WL, Chen WY, et al. Long-term outcome of pyogenic liver abscess: factors related with abscess recurrence. J Clin Gastroenterol 2008; 42: 1110-1115.





We thank Kerrie Stevens of the Microbiological Diagnostic Unit Public Health Laboratory, University of Melbourne, for performing serotyping of the above four isolates.
None identified.