In Australia, cancer is the leading cause of non-accidental death in childhood, and children with cancer make up the majority of those receiving end-of-life care.1 Over the past three decades, there has been increasing interest internationally in the medical aspects of palliation in childhood cancer, with data suggesting that current approaches are failing to meet the needs of patients and their families.2-4 Commonly reported symptoms, including pain, fatigue, poor appetite and dyspnoea, are typically suffered by over 75% of patients,5-9 and psychological problems, such as sadness and uncontrolled anxiety, have been documented in a high proportion of children dying of cancer.8,9 Medical reports also suggest that, despite no realistic chance of cure, many children continue to receive cancer-directed therapy during the end-of-life period8,9 and die unnecessarily in hospital.9,10
However, the results of international studies may not be directly applicable to children dying of cancer in Australia. Firstly, there are major differences in health care systems between Australia and other countries. Secondly, compared with countries such as Canada, the United States and the United Kingdom, Australia has more limited access to and use of experimental (Phase I and Phase II) anticancer agents and fewer oncologists providing care to children with cancer at the end of life. Thirdly, Australia’s great geographical distances often necessitate increased provision of local and regional services. While symptoms of children dying in an Australian hospital have been well documented11 and remain a concern for families, who usually prefer to care for their children at home,12 there is sparse information on families’ experiences of caring for children dying of cancer in Australia.
Once parents had provided written informed consent, trained clinical psychologists conducted structured, in-person interviews and provided self-report questionnaires. The content of the interview and questionnaires was developed by one of us (J W) and has previously been reported.9 Some measures and language were adjusted for use in the Australian population. A summary of items in the structured interview and self-report questionnaire is given in Box 1. In most cases, one parent was interviewed, but occasionally, both parents asked to be present during the interview. The self-report questionnaire was completed once for each participating family.
Fifty-seven per cent of the children were boys, with a mean age of 6.7 years (SD, 5.2 years) at diagnosis. The average duration of disease was 2.7 years (SD, 2.4 years; range, 1 month to 12.5 years), and the mean age at death was 9.4 years (SD, 5.8 years). Thirty-six per cent of the children had leukaemia, 32% had a brain tumour, and the remaining 32% had other solid tumours (most commonly neuroblastoma, Ewing’s sarcoma or rhabdomyosarcoma). There were no significant differences, for any of these variables, between children from participating families and those from eligible families who did not participate in our study (Box 2). The mean duration of palliative care (defined from the point when the parent accepted there was no realistic chance of cure) was 4.3 months (SD, 8.3 months; range, 1 day to 6 years).
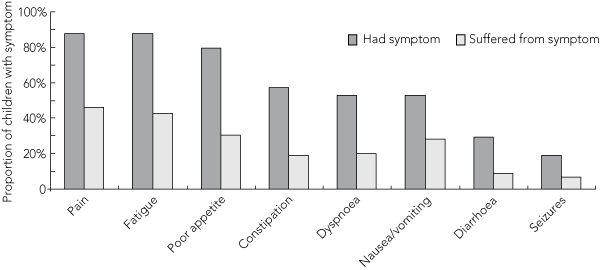
The proportion of children who, according to parental reports, suffered from specific symptoms during the last month of life, and the proportion who suffered “a lot” or “a great deal” as a result, are summarised in Box 3. Children who received cancer-directed therapy during the end-of-life period suffered from a greater number of symptoms than those who did not receive treatment (P = 0.03); however, the severity of symptoms did not differ between these groups. The most frequently reported symptoms were pain, fatigue and poor appetite. The proportion of children who were reported to experience significant suffering from these symptoms varied from 46% (in the case of pain) to 7% (in the case of seizures). Overall, 84% of children were reported to have experienced “a lot” or “a great deal” of suffering from at least one symptom, and 43% from three or more symptoms.
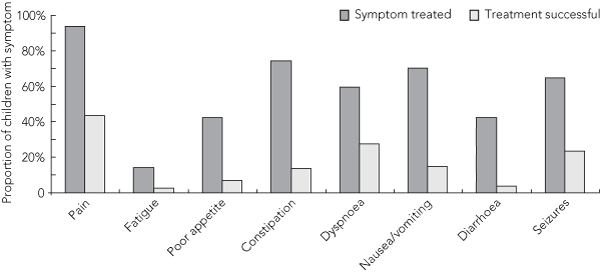
The proportions of children treated for various symptoms in the last month of life and the reported success rates of treatment are shown in Box 4. The most commonly treated symptoms were pain (in 95%), constipation (in 74%) and nausea/vomiting (in 70%), and the least commonly treated symptom was fatigue (in 14%). Symptoms were more likely to be treated in children who experienced a high level of suffering than in other children: fatigue (24% v 5%), dyspnoea (82% v 47%), constipation (94% v 65%), and poor appetite (52% v 36%).
The most successful treatments (expressed as the proportion of treated children whose treatment was successful) were those for pain (47%), dyspnoea (47%) and seizures (36%). The least successful treatments were for diarrhoea (9%), fatigue (18%) and poor appetite (17%) (Box 4). The implementation and success of treatments were not affected by whether the child died in hospital or at home, with one exception: dyspnoea was significantly more likely to be treated in children who died in hospital than in those who died at home (P = 0.02).
Recent guidelines for paediatric palliative care in Australia emphasise that high-quality medical care should be readily available and should focus on meeting the needs of not only patients but also their families and communities.13 To date, it has not been established whether the care of children dying of cancer in Australia achieves these goals. Our study cohort, recruited from the largest of the eight specialist paediatric oncology units in Australia, appears to be representative in age and tumour types of the childhood cancer population. Although data on parental factors such as age, ethnicity, educational level and religion were not available for all families whose children were cared for between 1996 and 2004, we believe that, allowing for the exclusion of non-English-speaking parents, the sample was representative.
A key finding of our study was the lower level of aggressive, cancer-directed treatment during the end-of-life period compared with the level in a similar cohort in the US (Box 5). Whether this reflects a more enlightened approach to palliative care or a reduced capacity to offer novel and experimental treatments is unclear. Certainly, with regard to children in our study who received cancer-directed therapy in the end-of-life period, the primary reasons for most of the treatment were to relieve suffering and extend life. It is noteworthy that these treatments appeared to be ineffective, even for symptom control. It was not possible in our study to determine whether patients who received cancer-directed treatments had medical problems directly related to their tumour burden.
As in other international studies, pain, fatigue and poor appetite were the three most commonly reported symptoms during the end-of-life period. The rates of these symptoms in our Australian population appeared to be very similar to those reported previously in US, European and Japanese populations (Box 5). It was pleasing that, in our study, a substantial proportion of children who experienced significant suffering from symptoms received treatment. However, it was disappointing that current treatments appear inadequate, with success rates of under 50% for all symptoms and under 20% for symptoms such as fatigue, poor appetite, constipation and diarrhoea. Given the positive impact that specialist paediatric palliative care services have had overseas,14 it is hoped that their recent introduction in the state of Victoria will improve symptom control and lessen suffering for children dying of cancer.
A striking difference between our study and previously published international studies was the low proportion of patients who died in hospital and, more specifically, in intensive care units (Box 5). The one study that reported a higher proportion of children dying at home8 was skewed by the elimination of children who died from treatment-related complications and those who were treated for less than 6 months. As in other international studies, nearly all parents in our study indicated a preference for their child to die at home, so the high rate of deaths at home was a positive finding. Furthermore, our finding that most symptoms were just as likely to be treated at home as in hospital, and that treatment was no less likely to be successful, supports the feasibility of providing adequate care at home.15,16 In addition, in view of previously reported evidence of better psychological outcomes in parents whose child dies at home,15,17,18 these findings are reassuring. The universal provision of bereavement services may also be a vital component of recovery from such a traumatic event.
There are a number of shortcomings of our study that restrict interpretation of the results. Our findings, based on a single hospital, may not accurately reflect the experiences in other areas of Australia, particularly the more remote areas of central, northern and western Australia. In addition, the possibility of selection bias cannot be discounted. In particular, the exclusion of non-English-speaking parents and those with psychiatric conditions (which constituted about 10% of parents) may have underestimated important problems relating to communication and access to services, as well as issues of cultural diversity. Recall bias is another factor that cannot be excluded. The information collected was retrospective, relying on accurate recollection by parents some years after the death of their child. Although the significance of events surrounding the death of a child means that parents are not likely to forget them easily, it is possible that their interpretation may have been affected by their own subsequent bereavement experiences. Furthermore, parental impressions of their child’s symptoms and suffering may not accurately reflect the child’s actual experience. Data from adult studies suggest that proxies’ perceptions of patients’ pain alters (downwards) with time19 and is influenced by the caregiver burden20 — factors that were not accounted for in our study.
1 Items in the structured interview and self-report questionnaire
3 Proportion of children who experienced specific symptoms compared with the proportion who, according to parental reports, suffered “a lot” or “a great deal” as a result of these symptoms in the last month of life
4 Proportion of children treated for various symptoms and reported success rates of treatment in the last month of life
Received 27 January 2009, accepted 7 May 2009
- John A Heath1
- Naomi E Clarke1
- Susan M Donath2
- Maria McCarthy1
- Vicki A Anderson3
- Joanne Wolfe4
- 1 Children’s Cancer Centre, Royal Children’s Hospital, Melbourne, VIC.
- 2 Centre for Epidemiology and Biostatistics, Murdoch Childrens Research Institute, Melbourne, VIC.
- 3 Department of Psychology, Royal Children’s Hospital, Melbourne, VIC.
- 4 Department of Pediatrics, Dana Farber Cancer Institute, Boston, Mass, USA.
We wish to thank Marie Dellaportas, Kate Enderby, Jessie Sprot and Vicki Crabb for conducting the interviews, and Lesley Chow for assistance with data management.
None identified.
- 1. Hynson JL, Gillis J, Collins JJ, et al. The dying child: how is care different? Med J Aust 2003; 179 (6 Suppl): S20-S22. <MJA full text>
- 2. Goldman A. Recent advances in palliative care: importance of palliative care for children is being increasingly recognised [letter]. BMJ 2001; 322: 234.
- 3. Kristjanson LJ. Expanding the boundaries of palliative care: an Australian perspective. J Palliat Care 2005; 21: 190-192.
- 4. Mack JW, Wolfe J. Early integration of pediatric palliative care: for some children, palliative care starts at diagnosis. Curr Opin Pediatr 2006; 18: 10-14.
- 5. Hongo T, Watanabe C, Okada S, et al. Analysis of the circumstances at the end of life in children with cancer: symptoms, suffering and acceptance. Pediatr Int 2003; 45: 60-64.
- 6. Jalmsell L, Kreicbergs U, Onelöv E, et al. Symptoms affecting children with malignancies during the last month of life: a nationwide follow-up. Pediatrics 2006; 117: 1314-1320.
- 7. Sirkiä K, Saarinen UM, Ahlgren B, Hovi L. Terminal care of the child with cancer at home. Acta Paediatr 1997; 86: 1125-1130.
- 8. Theunissen JM, Hoogerbrugge PM, van Achterberg T, et al. Symptoms in the palliative phase of children with cancer. Pediatr Blood Cancer 2007; 49: 160-165.
- 9. Wolfe J, Grier HE, Klar N, et al. Symptoms and suffering at the end of life in children with cancer. N Engl J Med 2000; 342: 326-333.
- 10. Fujii Y, Watanabe C, Okada S, et al. Analysis of the circumstances at the end of life in children with cancer: a single institution’s experience in Japan. Pediatr Int 2003; 45: 54-59.
- 11. Drake R, Frost J, Collins JJ. The symptoms of dying children. J Pain Symptom Manage 2003; 26: 594-603.
- 12. Collins JJ, Stevens MM, Cousens P. Home care for the dying child. A parent’s perception. Aust Fam Physician 1998; 27: 610-614.
- 13. Australian Department of Health and Ageing. Paediatric Palliative Care Service Model Review final report. Canberra: DHA, 2004.
- 14. Duncan J, Spengler E, Wolfe J. Providing pediatric palliative care: PACT in action. MCN Am J Matern Child Nurs 2007; 32: 279-287.
- 15. Martinson IM, Armstrong GD, Geis DP, et al. Home care for children dying of cancer. Pediatrics 1978; 62: 106-113.
- 16. Surkan PJ, Dickman PW, Steineck G, et al. Home care of a child dying of a malignancy and parental awareness of a child’s impending death. Palliat Med 2006; 20: 161-169.
- 17. Lauer ME, Camitta BM. Home care for dying children: a nursing model. J Pediatr 1980; 97: 1032-1035.
- 18. Mulhern RK, Lauer ME, Hoffmann RG. Death of a child at home or in the hospital: subsequent psychological adjustment of the family. Pediatrics 1983; 71: 743-747.
- 19. McPherson CJ, Addington-Hall JM. How do proxies’ perceptions of patients’ pain, anxiety, and depression change during the bereavement period? J Palliat Care 2004; 20: 12-19.
- 20. Higginson IJ, Gao W. Caregiver assessment of patients with advanced cancer: concordance with patients, effect of burden and positivity. Health Qual Life Outcomes 2008; 6: 42.





Abstract
Objective: To examine the symptoms, level of suffering, and care of Australian children with cancer at the end of life.
Design, setting and participants: In a study conducted at the Royal Children’s Hospital, Melbourne, parents of children who had died of cancer over the period 1996–2004 were interviewed between February 2004 and August 2006. Parents also completed and returned self-report questionnaires.
Main outcome measures: Proportions of children suffering from and treated for various symptoms; proportion of children receiving cancer-directed therapy at the end of life; proportion of children whose treatment of symptoms was successful; location of death.
Results: Of 193 eligible families, 96 (50%) were interviewed. All interviews were conducted in person, and occurred a mean of 4.5 years (SD, 2.1 years) after the child’s death. Eighty-four per cent of parents reported that their child had suffered “a lot” or “a great deal” from at least one symptom in their last month of life — most commonly pain (46%), fatigue (43%) and poor appetite (30%). Children who received cancer-directed therapy during the end-of-life period (47%) suffered from a greater number of symptoms than those who did not receive treatment (P = 0.03), but the severity of symptoms did not differ between these groups. Of the children treated for specific symptoms, treatment was successful in 47% of those with pain, 18% of those with fatigue and 17% of those with poor appetite. Of the 61 families who felt they had time to plan where their child would die, 89% preferred to have their child die at home. The majority of children (61%) died at home. Of those who died in hospital, less than a quarter died in the intensive care unit.
Conclusions: Relatively high rates of death at home and low rates of unsuccessful medical interventions suggest a realistic approach at the end of life for Australian children dying of cancer. However, many suffer from unresolved symptoms, and greater attention should be paid to palliative care for these children.