Increasing attention is being paid to the importance of peripheral arterial disease (PAD) as a marker of cardiovascular risk.1 PAD, as measured by the ankle–brachial index has been shown to be an independent marker of cardiovascular disease (CVD) in numerous population-based cohorts,2 although the role of the ankle–brachial index as an effective screening tool has yet to be established.3 Most patients with an abnormal ankle–brachial index do not have leg symptoms,4 and it is unclear how widely this measure is used just to identify patients at risk of CVD. For the clinician, intermittent claudication is the most common presentation of PAD. As such, its presentation remains an important opportunity to identify patients at risk of other CVD. Unfortunately, there is evidence that this opportunity is underutilised and the management of risk factors in patients with PAD remains suboptimal.5-8
Most of the studies that have examined the association between claudication and future CVD have focused on middle-aged individuals, and there is a paucity of data for older people.9-11 Given that the prevalence of PAD increases with age, it is important to understand its prognostic significance, and to identify any opportunities for the prevention of cardiovascular events in older age groups. We aimed to assess the prevalence, incidence, risk factors for, and prognosis of claudication in a large cohort of men aged 65 years and over.
We performed an observational cohort study (Box 1). The Health in Men Study (HIMS) arose from a trial of screening for abdominal aortic aneurysm.12 All aspects of the study were approved by the Human Research Ethics Committee of the University of Western Australia.
Briefly, between 1996 and 1999, men aged 65–83 years were identified from the Western Australian electoral roll and invited to attend a screening clinic. Men attending at baseline (n = 12 203) had their height, weight, circumference at waist and hip, and blood pressure measured. The diameter of the infra-renal aorta was measured by ultrasound. Each man completed a questionnaire assessing history and lifestyle relevant to CVD including: smoking status; alcohol consumption; exercise; dietary habits; history of (or treatment for) coronary heart disease (CHD), stroke or transient ischaemic attack, hypertension, dyslipidaemia or diabetes; and aspirin use.13 Prior CVD (a past history of angina, myocardial infarction, coronary intervention or stroke) was identified through the questionnaire or the Western Australian Data Linkage System using the relevant codes in the International Classification of Diseases (ICD). Claudication was assessed with the Edinburgh Claudication Questionnaire (ECQ).14
All surviving men were invited to participate in a follow-up survey which was conducted in 2001 to 2004.13 These men completed a new questionnaire which again included the ECQ. Additional information was obtained through the Western Australian Data Linkage System. Discharges or deaths from CVD, and discharges following procedures for PAD were identified with the relevant codes in the ICD.
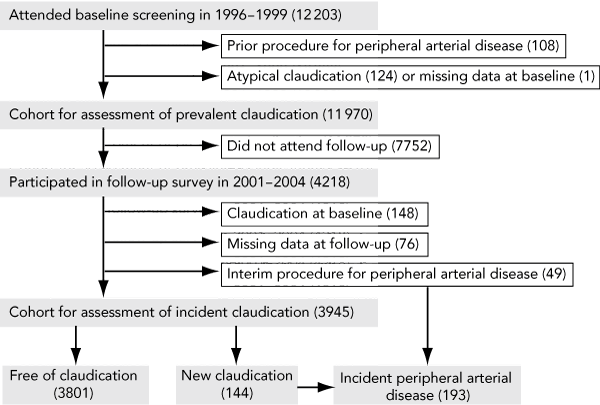
As the focus of our study was risk factors and outcome in men with claudication, rather than any PAD, those who had undergone a procedure for PAD before baseline screening (n = 108) were excluded. Men were considered to have prevalent claudication if they returned a “definite positive” ECQ. Men with atypical (“possible positive”) claudication14 at baseline (n = 124) or missing data (n = 1) were excluded, leaving 11 970 men for analysis of risk factors. The long-term outcome of men with and without claudication was also based on this cohort. Of the 11 970 men, 4218 attended for follow-up (Box 1). Men undergoing a procedure for PAD after baseline may have become free of claudication at follow-up, and could not necessarily be distinguished from men without claudication by their ECQ results. We therefore used the outcome of “incident PAD” rather than incident claudication. This was defined as a positive ECQ at follow-up or a procedure for PAD between baseline and follow-up, and was assessed in men who were free of claudication at baseline.
We used the statistical packages SPSS, version 15 (SPSS Inc, Chicago, Ill, USA), and Stata, version 10 (StataCorp, College Station, Tex, USA) to analyse the data. Risk factors were defined on the basis of answers in questionnaires and supplemented with data from the Western Australian Data Linkage System. Aortic diameter was categorised into < 19 mm, 19–22.9 mm, 23–29.9 mm and ≥ 30 mm on the basis of a previous analysis.15 Differences in categorical variables were assessed with Pearson’s χ2 test, and continuous variables were compared with t tests. Binary logistic regression was used to explore factors associated with claudication at baseline and at follow-up, and covariates were fitted in a forward, stepwise manner in multivariate models.
The overall prevalence of claudication at baseline was 5.3% (95% CI, 4.9%–5.7%), increasing with age from 3.9% (95% CI, 3.3%–4.5%) in men aged 65–69 years to 6.5% (95% CI, 4.8%–8.6%) in men aged 80 years and over (Box 2). After a median follow-up period of 5.7 years (interquartile range, 5.04–6.40 years), 4218 of the 11 970 men with ECQ data participated in the follow-up study (Box 1). After excluding the 148 men with claudication at baseline and the 76 with missing data at follow-up, the crude average annual incidence of incident PAD (claudication or procedure for PAD) was 0.85% (95% CI, 0.72%–0.96%). The cumulative incidence increased with age from 3.6% (95% CI, 2.7%–4.6%) in men aged 70–74 years to 8.6% (95% CI, 4.5%–14.6%) in men aged 85 years and over. During the first 4 years after baseline assessment, 38 men (6.0%) with claudication and 84 (0.07%) without claudication at baseline were subsequently admitted for a procedure for PAD (P < 0.001). Men who did not attend for follow-up were no more likely to undergo a procedure for PAD within 4 years of baseline than those who did attend for follow-up (1.1% [88/7752] v 0.8% [34/4218]; P = 0.087).
The baseline characteristics of men in the screening and follow-up cohorts are summarised in Box 2. With the exception of the proportion who were taking aspirin, those attending follow-up had significantly fewer cardiovascular risk factors than those not attending follow-up (data not shown). Men with claudication at baseline were significantly less likely to have attended for follow-up (23.2% v 35.9%, P < 0.001). Of the men with claudication at baseline, nearly half (47.5%, 303/638) were not taking aspirin. At follow-up, 42.5% (82/193) of the men with incident PAD were not taking aspirin.
The multivariate models of risk factors associated with prevalent claudication and incident PAD are shown in Box 3 and Box 4, respectively. The pattern of positive associations was similar with regard to age, smoking, hypertension, diabetes and history of CVD. Although exercise was associated with a lower prevalence of claudication, it was not associated with a lower incidence of PAD. There was a strong and graded relationship between smoking, and a J-shaped relationship between aortic diameter, and both prevalent claudication and incident PAD.
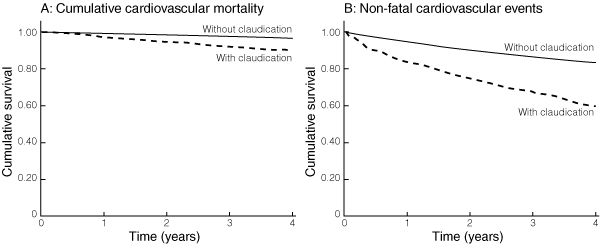
Kaplan–Meier time-to-event curves showed a clear increase in fatal and non-fatal cardiovascular events for men with claudication at baseline (Box 5). The Cox proportional hazards model for mortality from CVD is shown in Box 6. Claudication at baseline was associated with twice the risk of cardiovascular death (hazard ratio [HR], 2.00; 95% CI, 1.52–2.64); the association with non-fatal cardiovascular events was similar (HR, 1.80; 95% CI, 1.57–2.07). There was also a J-shaped relationship between aortic diameter and increasing risk.
The reported prevalence of claudication varies with method of assessment and type of population studied. It rises with age from about 1% of people under the age of 60 years to 3%–8% for those aged 70–74 years, and tends to be about twofold higher in men than in women.4,9,10,16,17 Surprisingly few studies have reported the prevalence of claudication in populations over the age of 75 years. In the Rotterdam Study (which used the World Health Organization Rose Questionnaire for angina pectoris and intermittent claudication) the prevalence of claudication in men was 3.3% in those aged 75–79 years, 4.5% in those aged 80–84 years and 6% in those aged 85 years and over.17 The prevalence of claudication in our study was comparable, but slightly higher (6.9% in those aged 75 years and over); this probably reflects the known greater sensitivity of the ECQ compared with the Rose Questionnaire.14 The true prevalence of claudication is undoubtedly higher because of the self-selection bias of those participating in the study.
The average annual incidence of new symptomatic PAD in men aged 65 years and over was about 0.8%. Most previous studies examining the incidence of claudication have focused on populations aged under 65 years (at baseline); the annual incidence varies considerably, but is probably in the region of 0.5%–2% for those aged 45–65 years.9-11,18 The assessment of incident claudication is challenging as a result of selective loss to follow-up of individuals most at risk of new claudication. To minimise the underestimation of incident claudication, we examined the outcome of incident PAD (either a procedure for PAD or new claudication).
The pattern of risk factors associated with prevalent claudication is consistent with atherosclerosis, and reflects that seen in numerous other studies. The pattern for incident symptomatic PAD was similar, although a history of or treatment for dyslipidaemia were not predictive. This may reflect the adequacy of control of this risk factor. Smoking was a more prominent risk factor for both prevalent claudication and incident PAD than for subsequent cardiovascular mortality. This dominant association between smoking and PAD compared with other cardiovascular risk factors (and outcomes) has been reported previously, and the reason for it is unknown.19 The association we found between diabetes and incident PAD differs from a recent study which showed that diabetes predicts progression of small-vessel PAD but not large-vessel PAD.20 Our result may have been influenced by our inclusion of men undergoing procedures for PAD, some of these procedures being for the combined consequences of PAD and diabetic foot complications.
The positive association between taking aspirin and claudication may be the result of an association with other cardiovascular indications for aspirin. Nevertheless, only about half of the men with prevalent claudication or incident PAD were taking aspirin. This relatively low level of aspirin use is consistent with findings of other studies; for example, a survey in the United Kingdom suggested that only 59% of patients with claudication were prescribed aspirin.5 Aspirin is recommended in all patients with PAD, despite evidence from a recent meta-analysis suggesting that aspirin may not reduce cardiovascular mortality.21
The strong association between claudication and subsequent cardiovascular events was first observed in middle-aged individuals (mostly aged 45–60 years) in cohorts of studies such as Framingham,22 Speedwell9 and Whitehall.23 This observation has been confirmed in studies that included older age groups (up to 75 years).11 Our study, which included 3208 men aged 75 years and over at baseline, confirms that claudication remains a statistically significant independent predictor of cardiovascular events in older men. The protective association with dyslipidaemia, and the insignificant association with hypertension may reflect the adequacy of management of these risk factors.
We have previously shown that aortic diameter predicts all-cause mortality.15 Based on these earlier findings, we included this variable in all our models, using a diameter of 19–22.9 mm as the reference (normal) range. Men with aortic diameters outside this range, including those with enlarged but non-aneurysmal aortic diameters (23–30 mm), were at increased risk of both prevalent claudication of cardiovascular death. The reason for the J-shaped association between aortic diameter and cardiovascular outcomes is unknown, but similar findings have recently been reported in the Cardiovascular Health Study.24
The rate of fatal and non-fatal cardiovascular events remains high in old age, suggesting that there is still scope for primary or secondary prevention. Our study shows that claudication is prevalent (at a rate of about 5%) among older men, and that about 0.8% develop new PAD each year. This is not only a cause of distressing symptoms, but is a significant predictor of cardiovascular outcome. The risk of claudication and cardiovascular mortality is associated with factors that can still be modified in older age including smoking, exercise, diet and aspirin therapy. Although a low ankle–brachial index (usually < 0.9) is a more sensitive marker of PAD than claudication, and has attracted considerable attention as a predictor of cardiovascular risk, there is recent evidence from the REACH (Reduction of Atherothrombosis for Continued Health) Registry that a diagnosis of PAD by a low ankle–brachial index alone does not ensure optimal risk factor control.8 While measuring the ankle–brachial index is relatively simple, eliciting a history of claudication is even simpler and is universally available at no cost. The significance of claudication in the identification of all patients at risk of future CVD, irrespective of age, should not be forgotten.
4 Multivariate logistic regression analysis of risk factors for incident peripheral arterial disease
5 Kaplan–Meier time-to-event curves of cumulative cardiovascular mortality and non-fatal cardiovascular events for 638 men with and 11 332 men without claudication*
 | |||||||||||||||
Received 23 September 2009, accepted 11 February 2010
- Rahul Lakshmanan1
- Zoë Hyde2
- Konrad Jamrozik3
- Graeme J Hankey2
- Paul E Norman1
- 1 University of Western Australia, Fremantle Hospital, Fremantle, WA.
- 2 University of Western Australia, Royal Perth Hospital, Perth, WA.
- 3 University of Adelaide, Adelaide, SA.
Special thanks to all the men who participated in the Western Australian Abdominal Aortic Aneurysm Program. Thanks to the staff, investigators and research students involved in the original screening and PAD trials. We are also grateful for assistance received from the State Electoral Commission, the Australian Bureau of Statistics, the Registrar General of Births, Deaths and Marriages, and the Western Australian Data Linkage Unit, and to hospitals in Perth for providing space in which to conduct screening. The study was funded by research grants from the National Health and Medical Research Council (964145 and 139093) and the Western Australian Health Promotion Foundation.
None identified.
- 1. Golomb BA, Dang TT, Criqui MH. Peripheral arterial disease: morbidity and mortality implications. Circulation 2006; 114: 688-699.
- 2. Ankle Brachial Index Collaboration. Ankle brachial index combined with Framingham risk score to predict cardiovascular events and mortality: a meta-analysis. JAMA 2008; 300: 197-208.
- 3. Beckman JA, Jaff MR, Creager MA. The United States Preventive Services Task Force recommendation statement on screening for peripheral arterial disease. More harm than benefit? Circulation 2006; 114: 861-866.
- 4. Murabito JM, Evans JC, Nieto K, et al. Prevalence and clinical correlates of peripheral arterial disease in the Framingham study. Am Heart J 2002; 143: 961-965.
- 5. Cassar K, Coull R, Bachoo P, et al. Management of secondary risk factors in patients with intermittent claudication. Eur J Vasc Endovasc Surg 2003; 26: 262-266.
- 6. Rehring TF, Sandhoff BG, Stolcpart RS, et al. Atherosclerotic risk factor control in patients with peripheral arterial disease. J Vasc Surg 2005; 41: 816-822.
- 7. Selvin E, Hirsch AT. Contemporary risk factor control and walking dysfunction in individuals with peripheral arterial disease: NHANES 1999–2004. Atherosclerosis 2008; 201: 425-433.
- 8. Cacoub PP, Abola MTB, Baumgartner I, et al. Cardiovascular risk factor control and outcomes in peripheral artery disease patients in the Reduction of Atherothrombosis for Continued Health (REACH) Registry. Atherosclerosis 2009; 204: e86-e92.
- 9. Bainton D, Sweetnam P, Baker I, Elwood P. Peripheral vascular disease: consequence for survival and association with risk factors in the Speedwell prospective heart disease study. Br Heart J 1994; 72: 128-132.
- 10. Bowlin SJ, Medalie JH, Flocke SA, et al. Epidemiology of intermittent claudication in middle-aged men. Am J Epidemiol 1994; 140: 418-430.
- 11. Leng GC, Lee AJ, Fowkes FGR, et al. Incidence, natural history and cardiovascular events in symptomatic and asymptomatic peripheral arterial disease in the general population. Int J Epidemiol 1996; 25: 1172-1181.
- 12. Norman PE, Jamrozik K, Lawrence-Brown MM, et al. Population-based randomised controlled trial on impact of screening on mortality from abdominal aortic aneurysm. BMJ 2004; 329: 1259-1262.
- 13. Norman PE, Flicker L, Almeida OP, et al. Cohort profile: the Health in Men Study (HIMS). Int J Epidemiol 2008; 38: 48-52.
- 14. Leng GC, Fowkes FGR. The Edinburgh Claudication Questionnaire: an improved version of the WHO/Rose questionnaire for use in epidemiological surveys. J Clin Epidemiol 1992; 45: 1101-1109.
- 15. Norman PE, Le MTQ, Pearce C, Jamrozik K. Infra-renal aortic diameter predicts all-cause mortality. Arterioscler Thromb Vasc Biol 2004; 24: 1278-1282.
- 16. Fowkes FGR, Housley E, Cawood EHH, et al. Edinburgh Artery Study: prevalence of asymptomatic and symptomatic peripheral arterial disease in the general population. Int J Epidemiol 1991; 20: 384-392.
- 17. Meijer WT, Hoes AW, Rutgers D, et al. Peripheral arterial disease in the elderly: the Rotterdam Study. Arterioscler Thromb Vasc Biol 1998; 18: 185-192.
- 18. Kollerits B, Heinrich J, Pichler M, et al. Intermittent claudication in the Erfurt Male Cohort (ERFORT) Study: its determinants and the impact on mortality. Atherosclerosis 2008; 198: 214-222.
- 19. Willgendael EM, Teijink JAW, Bartelink M-L, et al. Influence of smoking on incidence and prevalence of peripheral arterial disease. J Vasc Surg 2004; 40: 1158-1165.
- 20. Aboyans V, Criqui MH, Denenberg JO, et al. Risk factors for progression of peripheral arterial disease in large and small vessels. Circulation 2006; 113: 2623-2629.
- 21. Berger JS, Krantz MJ, Kittelson JM, Hiatt WR. Aspirin for the prevention of cardiovascular events in patients with peripheral artery disease. A meta-analysis of randomised trials. JAMA 2009; 301: 1909-1919.
- 22. Kannel WB, Skinner Jr JJ, Schwartz MJ, et al. Intermittent claudication. Incidence in the Framingham Study. Circulation 1970; 41: 875-883.
- 23. Smith GD, Shipley MJ, Rose G. Intermittent claudication, heart disease risk factors, and mortality. The Whitehall Study. Circulation 1990; 82: 1925-1931.
- 24. Freiberg MS, Arnold AM, Newman AB, et al. Abdominal aortic aneurysms, increasing infrarenal aortic diameter, and risk of total mortality and incident cardiovascular disease events. 10-year follow-up data from the Cardiovascular Health Study. Circulation 2008; 117: 1010-1017.





Abstract
Objectives: To assess the prevalence of and risk factors for claudication and its association with subsequent cardiovascular events.
Design, setting and participants: Observational cohort study of 12 203 Western Australian men aged 65 years and over, recruited from 1996 to 1999, and followed up from 2001 to 2004.
Main outcome measures: Prevalence of claudication and incidence of peripheral arterial disease (PAD); risk factors for claudication and its association with subsequent cardiovascular events.
Results: The prevalence of claudication was 5.3% (638 of 11 970 men). At follow-up, after exclusion of 148 men with claudication at baseline and 76 with missing data at follow-up, the crude average annual incidence of new PAD (claudication or procedure for PAD) was 0.85% (95% CI, 0.72%–0.96%). The risk factors for prevalent claudication and incident PAD were similar, with age, smoking, hypertension, diabetes and history of cardiovascular disease dominating. Of the men with claudication at baseline, nearly half (47.5%; 303 of 638) were not taking aspirin. At follow-up, 42.5% (82 of 193) of the men with incident PAD were not taking aspirin. Claudication at baseline was associated with twice the risk of cardiovascular death (hazard ratio, 2.00; 95% CI, 1.52–2.64). There was a J-shaped relationship between aortic diameter, and both prevalent claudication and subsequent cardiovascular events.
Conclusions: Among older men, claudication is prevalent and is associated with factors that can still be modified in older age, including smoking, exercise and diet. Relatively few men with claudication or at risk of PAD use aspirin. Claudication is a significant predictor of cardiovascular outcome.