Chromobacterium violaceum infection is uncommon but potentially fatal, with a clinical picture similar to melioidosis but with different antibiotic sensitivities and treatment. This infection can involve any organ, but we believe this is the first reported case of C. violaceum endocarditis.
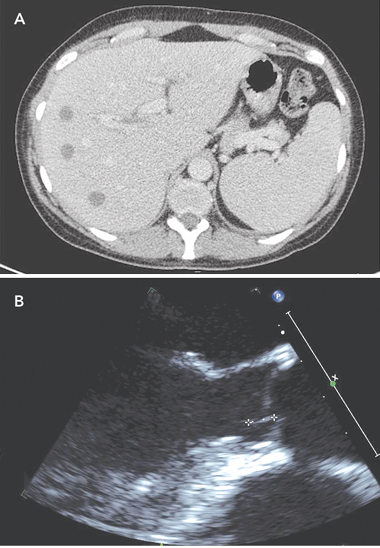
On Day 4, a computed tomography (CT) scan of the abdomen and pelvis showed multiple discrete hypodense lesions measuring up to 1.7 cm throughout the liver, and a bulky spleen (Box 1, A). Deep-purple and black colonies grew on Mueller–Hinton blood agar and antibiotic sensitivity plates. Chromobacterium violaceum, sensitive to ciprofloxacin, meropenem and co-trimoxazole, but resistant to gentamicin, was identified, and confirmed by testing on the VITEK 2 system (bioMérieux, Durham, NC, USA). B. pseudomallei, the causative organism of melioidosis,1 is not usually pigmented. The patient was commenced on intravenous meropenem.
Abatement of symptoms was slow, but the patient’s CRP level fell to 17 mg/L on Day 13. A repeat CT scan of the abdomen and pelvis on Day 10 showed generalised reduction in size of the liver abscesses. A transthoracic echocardiogram on Day 11 revealed mild mitral regurgitation. A contrast CT brain scan performed on Day 13 showed no abnormalities. A transoesophageal echocardiogram (TOE) performed on Day 15 demonstrated a small, 8 mm × 1 mm, linear mobile echodensity arising from the left ventricular outflow tract between the commissures of the left and right coronary cusps, consistent with a small vegetation (Box 1, B). An electrocardiogram was normal.
C. violaceum is a gram-negative, facultative anaerobic, non-sporing coccobacillus. It is commonly found in water and soil in tropical and subtropical regions of South-East Asia, South America and northern Australia. Apart from South America, it is endemic in the same regions as B. pseudomallei.1 Human disease is rare, and is most frequent in the wet season. Trauma is often an antecedent event, with abscess formation at the site. Systemic infections can be rapidly progressive, leading to metastatic abscess formation identical to that seen in melioidosis, with past documented mortality rates of 60%.2 We believe endocardial C. violaceum infection has not been reported previously. Immunodeficiency predisposes to the infection, although most patients with C. violaceum infections have no underlying immunodeficiency.
Ceftazidime and meropenem have become the drugs of choice for treating B. pseudomallei infection.3 White and colleagues demonstrated a 50% reduction in mortality from melioidosis using ceftazidime compared with combined chloramphenicol, co-trimoxazole and doxycycline.4 Sookpranee et al demonstrated the benefit of ceftazidime combined with co-trimoxazole compared with combined co-trimoxazole, chloramphenicol and doxycycline.5 Currie recommends ceftazidime, meropenem or imipenem as initial intensive therapy for melioidosis.6 Cheng et al found meropenem and ceftazidime equally effective in 6 years’ experience with 214 patients, although meropenem was preferred in critically ill patients.7 Resistance of B. pseudomallei to amikacin has been documented.8
In contrast to B. pseudomallei, C. violaceum infection is uncommon, with about 150 cases reported in the world literature, compared with over 200 cases of melioidosis documented in one series alone.7 No reports of controlled therapeutic trials have been published. Susceptibility to third-generation cephalosporins and aminoglycosides varies.9 However, there are documented cases of ceftazidime resistance from Australia,10 India11 and Brazil,12 with resulting fatalities. Amikacin in combination with gatifloxacin has been found to be successful for the treatment of puerperal sepsis from C. violaceum.13 Other case reports record cures with co-trimoxazole, quinolones, tetracyclines and chloramphenicol.14 Currently, there are limited published studies confirming the success of meropenem in treating C. violaceum infection.14
In conclusion, physicians should not rely on ceftazidime alone for treating a febrile traveller with suspected melioidosis, until infection with C. violaceum has been ruled out by microbiological cultures. There is a minor role for amikacin as combination therapy for C. violaceum infections (but no role for B. pseudomallei). Co-trimoxazole in combination with other agents may be an alternative if carbapenems are contraindicated. To exclude endocarditis, transoesophageal echocardiography should be considered. A summary of antibiotic recommendations for B. pseudomallei and C. violaceum infections is provided in Box 2.
- 1. Dance DA, Smith MD, Aucken HM, Pitt TL. Imported melioidosis in England and Wales. Lancet 1999; 353: 208.
- 2. Ponte R, Jenkins SG. Fatal Chromobacterium violaceum infections associated with exposure to stagnant waters. Pediatr Infect Dis J 1992; 11: 583-586.
- 3. Falade OO, Antonarakis ES, Kaul DR, et al. Clinical problem-solving. Beware of first impressions. N Engl J Med 2008; 359: 628-634.
- 4. White NJ, Dance DA, Chaowagul W, et al. Halving of mortality of severe melioidosis by ceftazidime. Lancet 1989; 2: 697-701.
- 5. Sookpranee M, Boonma P, Susaengrat W, et al. Multicenter prospective randomized trial comparing ceftazidime plus co-trimoxazole with chloramphenicol plus doxycycline and co-trimoxazole for treatment of severe melioidosis. Antimicrob Agents Chemother 1992; 36: 158-162.
- 6. Currie BJ. Melioidosis: an important cause of pneumonia in residents of and travellers returned from endemic regions. Eur Respir J 2003; 22: 542-550.
- 7. Cheng AC, Fisher DA, Anstey NM, et al. Outcomes of patients with melioidosis treated with meropenem. Antimicrob Agents Chemother 2004; 48: 1763-1765.
- 8. Wang YS, Wong CH, Kurup A. Cutaneous melioidosis and necrotizing fasciitis caused by Burkholderia pseudomallei. Emerg Infect Dis 2003; 9: 1484-1485.
- 9. Chang CY, Lee YT, Liu KS, et al. Chromobacterium violaceum infection in Taiwan: a case report and literature review. J Microbiol Immunol Infect 2007; 40: 272-275.
- 10. Huffam SE, Nowotny MJ, Currie BJ. Chromobacterium violaceum in tropical northern Australia. Med J Aust 1998; 168: 335-337.
- 11. Dutta S, Dutta SK. Multidrug resistant chromobacterium violaceum: an unusual bacterium causing long standing wound abscess. Indian J Med Microbiol 2003; 21: 217-218.
- 12. de Siqueira IC, Dias J, Ruf H, et al. Chromobacterium violaceum in siblings, Brazil. Emerg Infect Dis 2005; 11: 1443-1445.
- 13. Mahapatra A, Roy D, Poddar N, et al. Chromobacterium violaceum: a case report of an unusual bacterium causing puerperal sepsis. Indian J Pathol Microbiol 2006; 49: 450-452.
- 14. Jitmuang A. Human Chromobacterium violaceum infection in Southeast Asia: case reports and literature review. Southeast Asian J Trop Med Public Health 2008; 39: 452-460.





None identified.