Intravenous fluid therapy has been used for almost 200 years and remains a fundamental part of hospital patient care.1 However, approaches to the administration of water and sodium vary,2,3 with justification for any particular intravenous fluid regimen being based primarily on physiological concepts rather than evidence. Inappropriate administration of intravenous fluids — either the incorrect volume (too much or too little) or the incorrect type of fluid — is a significant cause of patient morbidity and mortality. Here, we recommend strategies to avoid inappropriate intravenous fluid therapy.
fixed fluid replacement regimens, adjusted by individualised assessment of fluid losses; and
variable, algorithmic approaches, where volume replacement is optimised by targeting a specific circulatory parameter.4,5
Fixed fluid regimens predominate in common clinical settings and include:
standard perioperative fixed fluid replacement regimens, as described in student texts, which usually advocate 3 L per day of fluid of varying composition;5
restrictive fluid regimens, which have long been advocated for postoperative management of pneumonectomy and lobectomy, and more recently for bowel surgery;6-9 and
liberal acute fluid resuscitation in severe burns.10
In general, fixed fluid regimens should be considered guides to safe volume replacement, with the actual amounts to be given determined by clinical response, including serial observations of heart rate, blood pressure and urine output. However, clinical assessment of volume requirement and clinical response are confounded by extremes of age, pre-existing disease, severity of acute illness or major surgery, and a continuum of general physiological changes, ranging from the normally adaptive stress response to life-threatening systemic inflammation with shock.11-14 Recent studies support the safety of more restrictive perioperative fluid regimens in uncomplicated elective surgery (Box 1).
Recent evidence also suggests that volume replacement targeting a specific circulatory parameter may improve patient outcome.15-17 These targets involve invasive monitoring of cardiac chamber filling pressures (central venous pressure and pulmonary artery wedge pressure) and cardiac output. Recent ultrasound techniques, such as continuous transoesophageal Doppler, allow less invasive monitoring of circulatory parameters, such as stroke volume. These regimens are necessarily restricted to critically ill patients in intensive care units and will not be discussed further here.
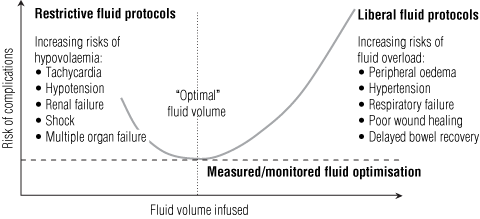
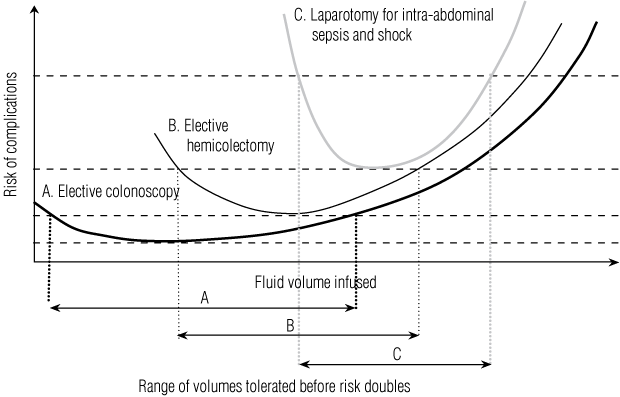
In both fixed and targeted regimens, there is a risk of either insufficient or excessive volume replacement. This risk is likely to be small in previously healthy patients with minimal acute illness. However, a patient’s tolerance to relative hypo- or hypervolaemia decreases with increasing severity of acute illness18 (Box 2 and Box 3).
Extreme hypovolaemia manifests clinically as shock, which may be treated with crystalloids or colloids. A recent Australian study showed that, in intensive care patients, use of either 4% albumin or 0.9% (isotonic) saline for fluid resuscitation resulted in similar outcomes at 28 days.20 Of note, the ratio of the volume of albumin to saline administered was about 1 : 1.4.
The clinical impact of a lesser degree of hypovolaemia can range from thirst and postural hypotension, increased incidence of perioperative nausea and vomiting, through to prolonged hospitalisation.21-23
There is no universally accepted definition of over-resuscitation or “fluid overload”,5,13,24 which is often understood to mean respiratory or heart failure secondary to excessive positive fluid balance.
Symptoms and signs attributed to fluid overload are likely to be determined by the magnitude of positive fluid balance, the severity of underlying cardiorespiratory disease and the nature and severity of acute illness. Limited human data exist, but volunteer studies suggest that infusion of more than 2–3 L isotonic saline in euvolaemic humans results in symptoms of mild nasal stuffiness, periorbital discomfort and a small, asymptomatic decrease in lung mechanics and gas exchange.25,26
As volume loading increases, maintenance of the plasma oncotic pressure may become important in protecting against pulmonary oedema. Acute illness is often associated with hypoalbuminaemia, and it has been shown that when filling pressure is increased, a low plasma oncotic pressure allows pulmonary oedema to develop at lower filling pressures.27
Unless clearly indicated, a more restrictive approach should be taken to maintenance fluid administration than the “mandatory 3 L per day” that is frequently prescribed (Box 1).
An ill patient whose condition is complicated may benefit from a targeted fluid replacement strategy.
When assessing a patient’s volume status and the relative risks of insufficient or excessive volume replacement, pre-existing cardiorespiratory and renal disorders, and the severity of pathophysiological changes associated with the acute illness itself must be taken into account.
If fluid overload is suspected, fluid restriction and/or diuretic therapy may be indicated; however, in more complex clinical situations attempts to achieve a negative fluid balance are not necessarily sufficient, effective or appropriate, as there may be a maldistribution of fluid and, while the patient overall may have fluid overload, there may still be a deficit of fluid in the vascular compartment.
Hypotonic fluids, such as 5% glucose and 4% glucose in 0.18% saline have low (or no) effective solutes when added to the ECF and cause net water movement into cells. Hypertonic fluids, such as 3% saline, added to the ECF cause net water movement from cells. Isotonic (0.9%) saline is isotonic to normal ECF and causes no net water movement. Hartmann’s solution is mildly hypotonic,28 having a calculated osmolality of 273 mOsm/kg, but a measured osmolality (by freezing point depression) of only 254 mOsm/kg.
Hyponatraemia (serum sodium concentration < 135 mmol/L) is the most common electrolyte disorder in hospitalised patients.29 Symptoms of hyponatraemia include headache, lethargy, decreasing level of consciousness and seizures. Death can result from cerebral oedema. Permanent neurological abnormalities may occur with rapid decreases in sodium concentration below 125 mmol/L, or too rapid correction of sodium concentration back towards this level (osmotic demyelination syndromes).
Hypotonic fluid administration to fasting postsurgical patients is the main cause of hyponatraemia in adults and children in hospital.19,30-33 The perioperative stress response, pain, nausea and vomiting all lead to increased secretion of antidiuretic hormone (ADH) which results in inappropriate accumulation of free water from “routine” administration of hypotonic fluids in susceptible individuals. The duration of the effect is from 12 hours after minor surgery to 4 days after major surgery.13
Fatal cases of hyponatraemia in otherwise healthy patients have led to publications questioning the routine use of hypotonic intravenous fluids and proposing they be replaced with isotonic saline in hospitalised patients.34-37
Hyponatraemia should be avoided by not administering hypotonic fluids to patients while they have increased ADH secretion. Once hyponatraemia occurs, management is determined by the cause and severity, the time over which it developed, and the presence of neurological symptoms.29,38 Initial management of acute hyponatraemia without neurological symptoms includes the cessation of all fluids that can exacerbate hyponatraemia. If the patient is euvolaemic or hypervolaemic, then the hyponatraemia is likely to result from inappropriate ADH secretion (SIADH, syndrome of inappropriate ADH) or excessive free water administration. Importantly, patients with SIADH can produce hypertonic urine, and thus neither isotonic (0.9% NaCl solution) nor hypotonic fluids should be administered, as they will exacerbate the hyponatraemia.29 Acute hyponatraemia associated with severe volume depletion (eg, diarrhoea or diuretic use) often corrects spontaneously when the volume depletion is restored with isotonic saline or colloids.39 Hypertonic solutions should only be considered for asymptomatic acute hyponatraemia if a standard approach does not adequately correct the hyponatraemia. The rate of correction of serum sodium concentration in asymptomatic patients with acute hyponatraemia should be gradual and not exceed 8 mmol/L per day.29 Neurological symptoms and persistent or worsening hyponatraemia despite standard treatment should prompt referral to a physician experienced in treatment of acute hyponatraemia.
Severe acute hyponatraemia (serum sodium concentration < 125 mmol/L) associated with neurological symptoms such as headache, drowsiness or seizure is a medical emergency that may result in death from cerebral oedema. The initial treatment goal should be rapid elevation of serum sodium concentration until acute neurological symptoms are controlled, usually by the administration of hypertonic saline (eg, 3% NaCl solution). This is followed by slower correction, as per the treatment of hyponatraemia without neurological symptoms. Permanent brain injury and death due to osmotic demyelination following symptomatic hyponatraemia and its correction are well described.29,40,41 Approaches to the rate of correction of serum sodium in this setting have been recently reviewed.37,42,43
Measure serum sodium concentration daily in all patients receiving maintenance fluids.
Use a staggered regimen for fluid administration, giving isotonic fluids (eg, 0.9% NaCl solution) during the period of high ADH secretion (24–96 hours, depending on the circumstance) and introduce hypotonic fluids only later or if hypernatraemia develops.
Completely avoid all hypotonic fluids in patients whose serum sodium concentration is low or falling rapidly (by > 8 mmol/L per day).
Acute decreases in serum sodium concentration below 125 mmol/L with neurological symptoms should be considered a medical emergency, and treatment should include prompt, controlled correction of serum sodium concentration.
Rapid correction of chronic or asymptomatic hyponatraemia is not indicated.
Hypernatraemia (serum sodium concentration > 145 mmol/L) is much less common than hyponatraemia and occurs when there is either excessive water loss from the ECF (most common) or excessive sodium gain (uncommon, and associated with hypervolaemia). Normal renal concentrating function and thirst often prevent hypernatraemia developing, but these can be impaired in infants and elderly and critically ill patients, increasing the propensity for hypernatraemia.44 Usually, the onset of hypernatraemia is gradual and results from insufficient replacement of free water loss from urine, skin (eg, fever, severe exercise, burns) or bowel (severe diarrhoea). However, diabetes insipidus due to either insufficient ADH release (central diabetes insipidus) or decreased renal distal tubular response to ADH (nephrogenic diabetes insipidus) can cause more rapid rises in serum sodium concentration because of large urinary losses of free water. Brain injury (central) and chronic lithium use (nephrogenic) are the two most common causes of diabetes insipidus.
Symptoms directly attributable to hypernatraemia are neurological and range from lethargy, weakness, and irritability, to seizures, obtundation and death in severe cases. Severe or permanent neurological dysfunction is more likely to occur with acute increases in serum sodium concentration above 160 mmol/L.45,46
Hypernatraemia secondary to water loss is managed by treating the specific cause for ongoing water loss, and is corrected by the administration of hypotonic fluids. These usually contain glucose (eg, 5% glucose, or 4% glucose in 0.18% NaCl solution) and may cause hyperglycaemia in patients with diabetes or if given in excessive quantities. If hyperglycaemia and hypernatraemia are severe, sterile water can be administered intravenously, but this requires central venous access as peripheral administration leads to red cell haemolysis.47 Central diabetes insipidus requires administration of the ADH analogue desmopressin (2–4 µg intravenously) to control urinary free water loss, whereas the nephrogenic form is managed by removing the cause of renal ADH unresponsiveness, if possible, and increasing the intake of hypotonic fluids.
Acute increases in serum sodium concentration above 150 mmol/L should be assessed for a cause and corrected.
Diabetes insipidus is important to recognise as it can cause large, rapid losses of free water with a rapid rise in serum sodium concentration.
In hypernatraemia, as in hyponatraemia, the rate of correction should be proportional to the rate of onset of the hypernatraemia, taking into account the presence and severity of neurological symptoms (maximal rate of correction 10 mmol/L per day). Overly rapid correction may result in cerebral oedema, seizures or death.
1 Examples of common restrictive postoperative fluid regimens*
Significant likelihood of inadequate volume state without sodium administration |
|||||||||||||||
2 Hypothetical curve of the risk of fluid therapy-related complications versus volume of fluid infused
3 Hypothetical risk versus volume replacement curves for an individual patient in different clinical scenarios
- Andrew K Hilton1
- Vincent A Pellegrino2
- Carlos D Scheinkestel3
- Intensive Care Unit, Alfred Hospital, Melbourne, VIC.
None identified.
- 1. Latta T. Letter from Dr Latta to the Secretary of the Central Board of Health, London, affording a view of the rationale and results of his practice in the treatment of cholera by aqueous and saline injections. Lancet 1831; 32 (2): 274-277.
- 2. Holte K, Kehlet H. Fluid therapy and surgical outcomes in elective surgery: a need for reassessment in fast-track surgery. J Am Coll Surg 2006; 202: 971-989.
- 3. Stoneham MD, Hill EL. Variability in post-operative fluid and electrolyte prescription. Br J Clin Pract 1997; 51: 82-84.
- 4. Grocott MPW, Mythen MG, Gan TJ. Perioperative fluid management and clinical outcomes in adults. Anesth Analg 2005; 100: 1093-1106.
- 5. Jacob M, Chappell D, Rehm M. Clinical update: perioperative fluid management. Lancet 2007; 369: 1984-1986.
- 6. Brandstrup B, Tønnesen H, Beier-Holersen R, et al. Effects of intravenous fluid restriction on postoperative complications: comparison of two perioperative regimens. A randomized assessor-blinded multicenter trial. Ann Surg 2003; 238: 641-648.
- 7. Joshi GP. Intraoperative fluid restriction improves outcome after major elective gastrointestinal surgery. Anesth Analges 2005; 101: 601-605.
- 8. Lobo DN, Bostock KA, Neal KR, et al. Effect of salt and water balance on recovery of gastrointestinal function after elective colonic resection: a randomised controlled trial. Lancet 2002; 359: 1812-1818.
- 9. Moller AM, Pedersen T, Svendsen PE, Engquist A. Perioperative risk factors in elective pneumonectomy: the impact of excess fluid balance. Eur J Anaesthesiol 2002; 19: 57-62.
- 10. Pham TN, Cancio LC, Gibran NS; American Burn Association. American Burn Association practice guidelines burn shock resuscitation. J Burn Care Res 2008; 29: 257-266.
- 11. Allison SP, Lobo DN. Fluid and electrolytes in the elderly. Curr Opin Clin Nutr Metab Care 2004; 7: 27-33.
- 12. Desborough JP. The stress response to trauma and surgery. Br J Anaesth 2000; 85: 109-117.
- 13. Gosling P. Salt of the earth or a drop in the ocean? A pathophysiological approach to fluid resuscitation. Emerg Med J 2003; 20: 306-315.
- 14. Rosenthal MH. Intraoperative fluid management — what and how much? Chest 1999; 115: 106S-112S.
- 15. Noblett SE, Snowden CP, Shenton BK, Horgan AF. Randomized clinical trial assessing the effect of Doppler-optimized fluid management on outcome after elective colorectal resection. Br J Surg 2006; 93: 1069-1076.
- 16. Sinclair S, James S, Singer M. Intraoperative intravascular volume optimisation and length of hospital stay after repair of proximal femoral fracture: randomised controlled trial. BMJ 1997; 315: 909-912.
- 17. Wakeling HG, McFall MR, Jenkins CS, et al. Intraoperative oesophageal Doppler guided fluid management shortens postoperative hospital stay after major bowel surgery. Br J Anaesth 2005; 95: 634-642.
- 18. Bellamy MC. Wet, dry or something else? Br J Anaesth 2006; 97: 755-757.
- 19. Ayus JC, Arieff AI. Pathogenesis and prevention of hyponatremic encephalopathy. Endocrinol Metab Clin North Am 1993; 22: 425-446.
- 20. The SAFE Study Investigators. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med 2004; 350: 2247-2256.
- 21. Maharaj CH, Kallam SR, Malik A, et al. Preoperative intravenous fluid therapy decreases postoperative nausea and pain in high risk patients. Anesth Analg 2005; 100: 675-682.
- 22. Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001; 345: 1368-1377.
- 23. Yogendran S, Asokumar B, Cheng DC, Chung F. A prospective randomized double-blinded study of the effect of intravenous fluid therapy on adverse outcomes on outpatient surgery. Anesth Analg 1995; 80: 682-686.
- 24. Holte K, Sharrock E, Kehlet H. Pathophysiology and clinical implications of perioperative fluid excess. Br J Anaesth 2002; 89: 622-632.
- 25. Collins JV, Cochrane GM, Davis J, et al. Some aspects of pulmonary function after rapid saline infusion in healthy subjects. Clin Sci Mol Med 1973; 45: 407-410.
- 26. Holte K, Jensen P, Kehlet H. Physiologic effects of intravenous fluid administration in healthy volunteers. Anesth Analg 2003; 96: 1504-1509.
- 27. Guyton AC, Lindsey AW. Effect of elevated left atrial pressure and decreased plasma protein concentration on the development of pulmonary edema. Circ Res 1959; 7: 649-657.
- 28. Williams EL, Hildebrand KL, McCormick SA, Bedel MJ. The effect of intravenous lactated Ringer’s solution versus 0.9% sodium chloride solution on serum osmolality in human volunteers. Anesth Analg 1999; 88: 999-1003.
- 29. Adrogue HJ, Madias NE. Hyponatremia. N Engl J Med 2000; 342: 1581-1589.
- 30. Moritz ML, Ayus JC. Hospital-acquired hyponatremia: why are there still deaths? Pediatrics 2004; 113: 1395-1396.
- 31. Aronson D, Dragu RE, Nakhoul F, et al. Hyponatremia as a complication of cardiac catheterization: a prospective study. Am J Kidney Dis 2002; 40: 940-946.
- 32. Ayus JC, Wheeler JM, Arieff AI. Postoperative hyponatremic encephalopathy in menstruant women. Ann Intern Med 1992; 117: 891-897.
- 33. Chung HM, Kluge R, Schrier RW, Anderson RJ. Postoperative hyponatremia. A prospective study. Arch Intern Med 1986; 146: 333-336.
- 34. Arieff AI, Ayus JC, Fraser CL. Hyponatraemia and death or permanent brain damage in healthy children. BMJ 1992; 304: 1218-1222.
- 35. Ayus JC, Arieff AI. Postoperative hyponatremia. Ann Intern Med 1997; 126: 1005-1006.
- 36. McAloon J, Kottyal R. A study of current fluid prescribing practice and measures to prevent hyponatraemia in Northern Ireland’s paediatric departments. Ulster Med J 2005; 74: 93-97.
- 37. Moritz ML, Ayus JC. Hospital-acquired hyponatremia — why are hypotonic parenteral fluids still being used? Nat Clin Pract Nephrol 2007; 3: 374-382.
- 38. Palmer BF, Gates JR, Lader M. Causes and management of hyponatremia. Ann Pharmacother 2003; 37: 1694-1702.
- 39. Han DS, Cho BS. Therapeutic approach to hyponatremia. Nephron 2002; 92 Suppl 1: 9-13.
- 40. Heng AE, Vacher P, Aublet-Culelier B, et al. Centropontine myelinolysis after correction of hyponatremia: role of associated hypokalemia. Clin Nephrol 2007; 67: 345-351.
- 41. Nathan BR. Cerebral correlates of hyponatremia. Neurocrit Care 2007; 6: 72-78.
- 42. Miller M. Hyponatremia and arginine vasopressin dysregulation: mechanisms, clinical consequences, and management. J Am Geriatr Soc 2006; 54: 345-353.
- 43. Snell DM, Bartley C. Osmotic demyelination syndrome following rapid correction of hyponatraemia. Anaesthesia 2008; 63: 92-95.
- 44. Adrogue HJ, Madias NE. Hypernatremia. N Engl J Med 2000; 342: 1493-1499.
- 45. Schrier RW, editor. Renal and electrolyte disorders. 6th ed. New York: Lippincott Williams and Wilkins, 2003.
- 46. Rose BD, Post TW. Clinical physiology of acid–base and electrolyte disorders. 5th ed. New York: McGraw–Hill, 2001.
- 47. Worthley LI. Hyperosmolar coma treated with intravenous sterile water. A study of three cases. Arch Intern Med 1986; 146: 945-947.





Abstract
Inappropriate intravenous fluid therapy is a significant cause of patient morbidity and mortality and may result from either incorrect volume (too much or too little) or incorrect type of fluid.
Fluid overload has no precise definition, but complications usually arise in the context of pre-existing cardiorespiratory disease and severe acute illness.
Insufficient fluid administration is readily identified by signs and symptoms of inadequate circulation and decreased organ perfusion.
Administration of the wrong type of fluid results in derangement of serum sodium concentration, which, if severe enough, leads to changes in cell volume and function, and may result in serious neurological injury.
In patients whose condition is uncomplicated, we recommend a restrictive approach to perioperative intravenous fluid replacement, with initial avoidance of hypotonic fluids, and regular measurement of serum concentration of electrolytes, especially sodium.