Immune thrombocytopenic purpura (ITP) is the most common autoimmune-mediated haematological disorder, characterised by antibodies against platelet surface antigens.1 Its aetiology, pathogenesis and molecular receptor targets remain unclear. It has been associated with HIV infection and hepatitis C.1
Helicobacter pylori is a causative agent in a spectrum of gastrointestinal diseases, including peptic ulcer disease, gastric cancer and MALToma (mucosa-associated lymphoid tissue lymphoma). There is growing evidence of an association between H. pylori eradication and platelet recovery in patients with ITP.2-6 The evidence is strongest in Japan, where H. pylori eradication is now recommended as an initial treatment for infected ITP patients.7 Screening and eradication may be a simpler and safer therapeutic option than immunosuppression or splenectomy in certain ITP patients.8,9 Most studies of H. pylori and ITP are from Japan, Spain and Italy; however, the British Society for Haematology now recommends H. pylori screening and eradication as a treatment in ITP (evidence level, III).10 To date, there have been no published reports of such therapy in Australian patients. Local experience is important, given the potential regional variation in strains of H. pylori.
We began routine testing for H. pylori in patients with ITP referred for splenectomy in August 2005. Testing was via gastroscopy, serological testing or urea breath test.11 Patients were treated with standard triple therapy, comprising clarithromycin 500 mg, amoxycillin 1000 mg and oral esomeprazole 20 mg, all twice daily for 1 week. One patient was treated with metronidazole instead of amoxycillin due to penicillin allergy. Eradication was confirmed by urea breath test, with titres of isotope 14C < 50 disintegrations/min 4 weeks after completion of triple therapy indicating a negative result. Patients who had previously undergone successful H. pylori eradication therapy were not treated again.
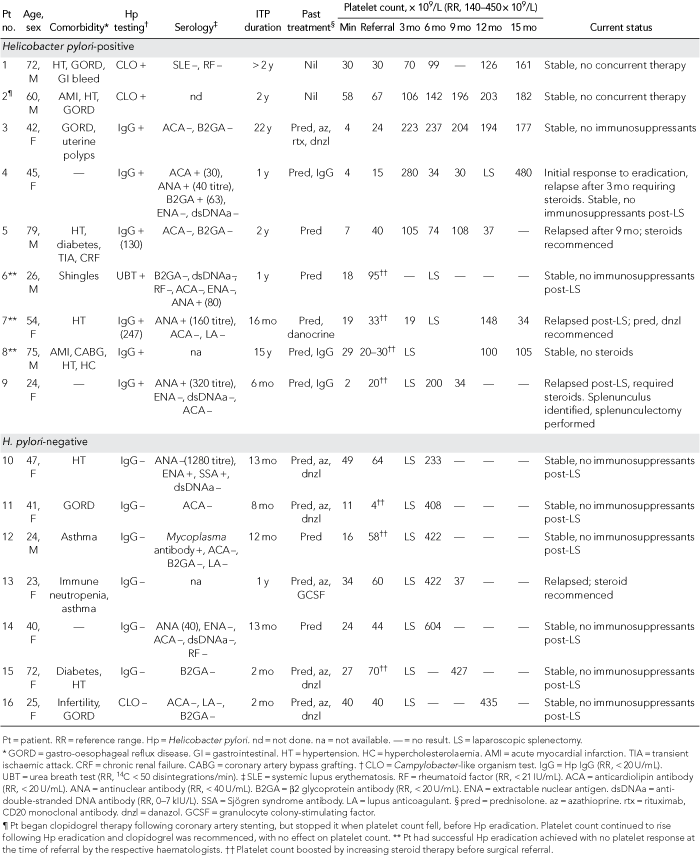
Clinical characteristics of the patients and their responses to treatment are summarised in Box 1.
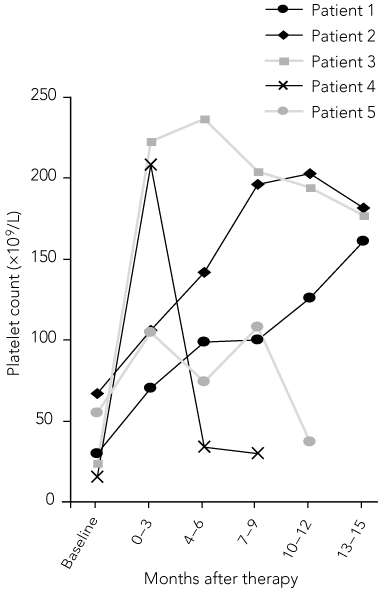
Nine of 16 patients tested positive for H. pylori and all had successful eradication therapy. Of these, five patients showed an initial platelet response, and three of these showed a long-term (> 12 months) response and did not require immunosuppression or splenectomy (Box 2). Only one of these patients had initial severe ITP (platelet count, 4 × 109/L [reference range, 140–450 × 109/L]). Two of three patients who tested positive for anticardiolipin or antinuclear antibodies also tested positive for H. pylori and did not have a sustained platelet response to H. pylori eradication.
The association between ITP and H. pylori was first reported in 1998.12 Possible aetiological links include molecular mimicry and cross-reactivity of bacterial and platelet antigens, in association with host factors such as specific human leukocyte antigen (HLA) class II alleles.13,14 A recent Italian study found that the HLA-DRB1*11, -DRB1*14 and -DQB1*03 alleles occur significantly more often in H. pylori-infected ITP patients and indicate a higher probability of platelet response to eradication therapy.15 ITP patients who had successful H. pylori eradication showed significantly decreased serum platelet-associated IgG levels, suggesting that removal of the H. pylori antigenic stimulus leads to platelet recovery.9
However, response rates have varied. A literature review of 13 major studies published between 1998 and 2004 revealed an overall platelet response rate of 52% following H. pylori eradication therapy when used as an adjunct to standard ITP therapy or in otherwise untreated cases.5 Another review concluded that 58% of 482 patients with ITP were H. pylori-positive and, of these, more than 50% showed a significant platelet response to successful H. pylori eradication.13 Long-term follow-up studies, including one with a median follow-up of 31 months, have also shown good results.2,16,17 A large retrospective analysis in Japan yielded a platelet response rate as high as 63%, with remission sustained after 12 months, even among patients whose ITP was refractory to splenectomy.7 A small randomised controlled trial, also in Japan, showed a significant increase in platelet count in the group who had successful eradication therapy.3 Some studies showed that platelet recovery was better in less severe or recent-onset ITP,18,19 or in the absence of concurrent steroid therapy.20
In contrast, several studies have refuted a significant association between H. pylori and ITP.21-23 For example, a prospective study in the United States found that only one of 14 patients who responded to H. pylori eradication had a rise in platelet count.21
There is substantial variability in ITP response to H. pylori eradication in different countries.5,18,20,24 This may be due to variations in bacterial virulence factors, such as CagA expression or host immunological class II HLA factors.2,24 Most Japanese H. pylori strains are CagA-positive, unlike most American strains.9 Patients infected with CagA-positive strains (as measured by anti-CagA IgG antibodies) are thought to have increased platelet response to H. pylori eradication compared with those who are CagA-negative or who have low serum titres of CagA antibodies.3,5 Serum levels of anti-CagA IgG may be used as a predictor of response to H. pylori treatment.2,25
The prevalence of H. pylori infection in ITP patients varies from 22% in a US study21 to 47% in a study from the United Kingdom and Italy19 to 71% in a Spanish study.23 In a comparison of H. pylori and ITP data from the UK and Italy, the prevalence of H. pylori infection in patients with ITP appeared to be similar to that in the general population.19 The prevalence of H. pylori infection in the general Australian population is 25%–38%,26 but its prevalence in Australians with ITP is yet to be determined. In our series, 56% of ITP patients were H. pylori-positive.
Received 12 October 2007, accepted 2 March 2008
- Vanaja Sivapathasingam1,2
- Michael P Harvey1
- Robert B Wilson1
- 1 Liverpool Hospital, Sydney, NSW.
- 2 Monash Medical Centre, Melbourne, VIC.
None identified.
- 1. Chong BH, Ho S-J. Autoimmune thrombocytopenia: review. J Thromb Haemost 2005; 3: 1763-1772.
- 2. Gasbarrini A, Franceschi F. Does H. pylori infection play a role in idiopathic thrombocytopenic purpura and in other autoimmune diseases? Am J Gastroenterol 2005; 100: 1271-1273.
- 3. Suzuki T, Matsushima M, Masui A, et al. Effect of Helicobacter pylori eradication in patients with chronic idiopathic thrombocytopenic purpura — a randomized controlled trial. Am J Gastroenterol 2005; 100: 1265-1270.
- 4. Suvajdzic N, Stankovic B, Artiko V, et al. Helicobacter pylori eradication can induce platelet recovery in chronic idiopathic thrombocytopenic purpura: prospective cohort study. Platelets 2006; 17: 227-230.
- 5. Jackson S, Beck P, Pineo G, Poon M. Helicobacter pylori eradication: novel therapy for immune thrombocytopenic purpura? A review of literature. Am J Hematol 2005; 78: 142-150.
- 6. Franchini M, Veneri D. Helicobacter pylori-associated immune thrombocytopenia: review. Platelets 2006; 17: 71-77.
- 7. Fujimura K, Kuwana M, Kurata Y, et al. Is eradication therapy useful as the first line of treatment in Helicobacter pylori-positive idiopathic thrombocytopenic purpura? Analysis of 207 eradicated chronic ITP cases in Japan. Int J Hematol 2005; 81: 162-168.
- 8. Kuwana M, Ikeda Y. Helicobacter pylori and immune thrombocytopenic purpura: unsolved questions and controversies. Int J Hematol 2006; 84: 309-315.
- 9. Inaba T, Mizuno M, Take S, et al. Eradication of Helicobacter pylori increases platelet count in patients with idiopathic thrombocytopenic purpura in Japan. Eur J Clin Invest 2005; 35: 214-217.
- 10. British Committee for Standards in Haematology General Haematology Task Force. Guidelines for the investigation and management of idiopathic thrombocytopenic purpura in adults, children and in pregnancy. Br J Haematol 2003; 120: 574-596.
- 11. Vaira D, Vakil N. Blood, urine, stool, breath, money, and Helicobacter pylori. Gut 2001; 48: 287-289.
- 12. Gasbarrini A, Franceschie F, Tartaglione R, et al. Regression of autoimmune thrombocytopenia after eradication of Helicobacter pylori. Lancet 1998; 352: 878.
- 13. Franchini M, Veneri D. Helicobacter pylori infection and immune thrombocytopenic purpura — an update. Helicobacter 2004; 9: 342-344.
- 14. Ishiyama M, Teramura M, Iwabe K, et al. Clonally expanded T-cells in the peripheral blood of patients with idiopathic thrombocytopenic purpura and Helicobacter pylori infection. Int J Hematol 2006; 83: 147-151.
- 15. Veneri D, De Matteis G, Solero P, et al. Analysis of B- and T-cell clonality and HLA class II alleles in patients with idiopathic thrombocytopenic purpura: correlation with Helicobacter pylori infection and response to eradication treatment. Platelets 2005; 16: 307-311.
- 16. Kohda K, Kuga T, Kogawa K, et al. Effect of Helicobacter pylori eradication on platelet recovery in Japanese patients with chronic idiopathic thrombocytopenic purpura and secondary autoimmune thrombocytopenic purpura. Br J Haematol 2002; 118: 584-588.
- 17. Veneri D, Krampera M, Franchini M. High prevalence of sustained remission of idiopathic thrombocytopenic purpura after Helicobacter pylori eradication: a long-term follow-up study. Platelets 2005; 16: 117-119.
- 18. Ando K, Shiamoto T, Tauchi T, et al. Can eradication therapy for Helicobacter pylori really improve the thrombocytopenia in ITP? Our experience and literature review. Int J Hematol 2003; 77: 239-244.
- 19. Stasi R, Rossi Z, Stipa E, et al. Helicobacter pylori eradication in the management of patients with idiopathic thrombocytopenic purpura. Am J Med 2005; 118: 414-419.
- 20. Sato R, Murakami K, Watanabe K, et al. Effect of Helicobacter pylori eradication on platelet recovery in patients with chronic idiopathic thrombocytopenic purpura. Arch Intern Med 2004; 164: 1904-1907.
- 21. Michel M, Cooper N, Jean C, et al. Does Helicobacter pylori initiate or perpetuate immune thrombocytopenic purpura? Blood 2004; 103: 890-896.
- 22. Michel M, Khellaf M, Desforges, et al. Autoimmune thrombocytopenic purpura and Helicobacter infection. Arch Intern Med 2002; 162: 1033-1036.
- 23. Jarque I, Andreu R, Llopis J, et al. Absence of platelet response after eradication of Helicobacter pylori infection in patients with idiopathic thrombocytopenic purpura. Br J Haematol 2001; 115: 1002-1003.
- 24. Takahashi T, Yujiri T, Tanizawa Y. Helicobacter pylori and chronic ITP: the discrepancy in the clinical responses to eradication therapy might be due to differences in the bacterial strains. Blood 2004; 104: 594.
- 25. Kodama M, Kitadai Y, Ito M, et al. Immune response to CagA protein is associated with improved platelet count after Helicobacter pylori eradication in patients with idiopathic thrombocytopenic purpura. Helicobacter 2007; 12: 36-42.
- 26. Lam VT, Trinh LK, Wilson R. Helicobacter infection and treatment outcome in an urban Australian population. ANZ J Surg 2006; 76: 710-714.





Abstract
Objective: To determine whether Helicobacter pylori eradication is an effective treatment for Australian patients with chronic immune thrombocytopenic purpura (ITP).
Design, setting and patients: Retrospective analysis of clinical records of a consecutive series of ITP patients referred to a gastrointestinal surgeon in a tertiary referral hospital for laparoscopic splenectomy between August 2005 and November 2007.
Main outcome measures: Platelet response (measured at least 3-monthly) following successful H. pylori eradication therapy (confirmed by urea breath test 4 weeks later).
Results: Of 16 patients, seven were H. pylori-negative and underwent laparoscopic splenectomy. Nine were H. pylori-positive and successfully underwent H. pylori eradication therapy; five of the nine had an initial platelet response. Four patients had platelet counts > 100 × 109/L (reference range, 140–450 × 109/L) and were off all immunosuppression at 9 months; three had a sustained response beyond 12 months. One patient had an initial response at 3 months (15 × 109/L to 208 × 109/L), but relapsed 4 months after H. pylori eradication and underwent splenectomy with platelet count recovery. The remaining four patients showed no platelet response and subsequently underwent splenectomy.
Conclusion: Larger prospective studies are needed to fully ascertain the role of H. pylori in Australian patients with ITP. However, H. pylori eradication is simple and safe. H. pylori screening and eradication should be considered before immunosuppression or splenectomy.