Clinical record
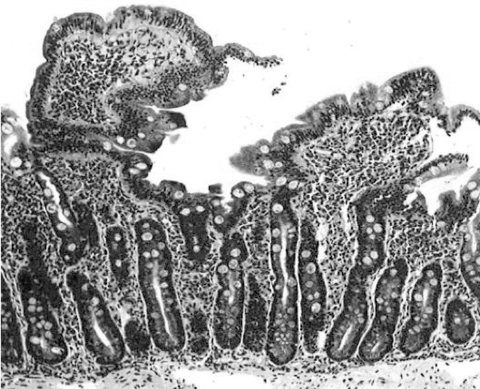
Titration of thyroxine to a weekly dose of 1400 μg and vitamin D3 replacement dosage of 6000 IU daily, over a 6-month period with confirmed compliance, failed to ameliorate hypothyroidism and vitamin D deficiency. In view of her history, previous investigations and progress, serological testing for coeliac disease was carried out. Results were positive for endomysial IgA antibodies, with normal total IgA antibody titre. Also, histological analysis of a small bowel biopsy specimen was consistent with coeliac disease (Figure).
A 6-month gluten-free diet resulted in complete resolution of lethargy, diarrhoea and myalgia. Hypothyroidism was corrected with a weekly dose of 750 μg thyroxine, and both secondary hyperparathyroidism and impaired fasting glycaemia resolved (Table).
While the prevalence of coeliac disease in the European population has been estimated to range from 1 in 150 to 1.5 in 1000,1 the prevalence in the Indian population is unknown due to lack of population-based data,2 leading to the common misconception that coeliac disease is rare. However, recent studies using new serological screening methods have demonstrated gross underdiagnosis of coeliac disease in the past,1-3 and an unexpectedly frequent prevalence in countries populated by non-Europeans. The highest reported population prevalence is in the Saharawi people of Arab–Berber origin, who live in the Sahara desert, with a prevalence of 5.6% — almost tenfold higher than that reported from most European countries.4 Similarly, recent studies in India found a prevalence of 9%–26% in patients who presented with chronic diarrhoea or malabsorption.5,6
The pathogenesis of coeliac disease is related to intolerance of gluten that results in a T-lymphocyte-mediated, small intestinal enteropathy in genetically predisposed individuals, who commonly express the HLA-DQ2 or HLA-DQ8 haplotypes.7 Such genetic predisposition is common in Europeans, but also occurs in Indian patients, as evident from detection of the HLA-DQ2 heterodimer in 14 out of 15 North Indian patients with coeliac disease.8 The HLA-DQ2 haplotype is also found in almost 25% of the healthy North Indian population, similar to the Western population,9 which has led to speculation that some Indians share ancestral origin with “ancient Caucasians” from the Fertile Crescent.9,10
Environmental challenge from the increasingly popular Western diet, which is gluten-rich, may also contribute to observed changes in the epidemiology of coeliac disease. For example, “summer diarrhoea” has been described in communities of Punjabis and Gujaratis from India. Traditionally, wheat replaces maize during summer in India, and Punjabis and Gujaratis residing in England and Canada have been reported to develop coeliac disease when exposed to a gluten-rich diet.11,12
Another misconception about coeliac disease patients is that they are all underweight. In one study, the mean body mass index of 371 patients with coeliac disease was 24.6 kg/m2, with only 5% being underweight, while 39% and 13% were in the overweight and obese ranges, respectively.13 Our patient’s body mass index was in the overweight range, which may have contributed to the delay in diagnosis until she was referred to our clinic.
The complex symptomatology of our patient illustrates the non-specific, extraintestinal manifestations of coeliac disease, including lethargy, neuropsychiatric complaints, and myalgia from vitamin D deficiency; the latter may also lead to impaired insulin secretion and result in impaired fasting glycaemia.14 A high index of suspicion is required to screen for the disease, which is associated with significant morbidity.
Lessons from practice
Coeliac disease is not limited to Europeans; it has been increasingly reported in patients from non-European backgrounds.
Not all patients with coeliac disease are underweight, and a significant minority are overweight or obese.
Serological testing is non-invasive and should be considered in patients with suggestive symptoms or associated autoimmune conditions, regardless of racial background.
Presentation of coeliac disease can be non-specific, and is not limited to gastrointestinal manifestations. A high index of suspicion is required to screen for the condition in patients with predominant extraintestinal presentation.
- Paul Lee1,2
- Katherine Samaras1,3,4
- 1 Department of Endocrinology, St Vincent’s Hospital, Sydney, NSW.
- 2 Pituitary Research Unit, Garvan Institute of Medical Research, Sydney, NSW.
- 3 University of New South Wales, Sydney, NSW.
- 4 Diabetes and Obesity Clinical Group, Garvan Institute of Medical Research, Sydney, NSW.
- 1. Fasano A, Berti I, Gerarduzzi T, et al. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study. Arch Intern Med 2003; 163: 286-292.
- 2. Bhatnagar S, Tandon N. Diagnosis of celiac disease. Indian J Pediatr 2006; 73: 703-709.
- 3. Sood A, Midha V, Sood N, et al. Increasing incidence of celiac disease in India. Am J Gastroenterol 2001; 96: 2804-2805.
- 4. Catassi C, Doloretta Macis M, Rätsch IM, et al. The distribution of DQ genes in the Saharawi population provides only a partial explanation for the high celiac disease prevalence. Tissue Antigens 2001; 58: 402-405.
- 5. Ranjan P, Ghoshal UC, Aggarwal R, et al. Etiological spectrum of sporadic malabsorption syndrome in northern Indian adults at a tertiary hospital. Indian J Gastroenterol 2004; 23: 94-98.
- 6. Sachdev A, Srinivasan V, Maheswary S, et al. Adult onset celiac disease in north India. Trop Gastroenterol 2002; 23: 117-119.
- 7. Schuppan D. Current concepts of celiac disease pathogenesis. Gastroenterology 2000; 119: 234-242.
- 8. Agrawal S, Gupta A, Yachha SK, et al. Association of human leucocyte-DR and DQ antigens in coeliac disease: a family study. J Gastroenterol Hepatol 2000; 15: 771-774.
- 9. Kaur G, Sarkar N, Bhatnagar S, et al. Pediatric celiac disease in India is associated with multiple DR3-DQ2 haplotypes. Hum Immunol 2002; 63: 677-682.
- 10. Catassi C. Where is celiac disease coming from and why? J Pediatr Gastroenterol Nutr 2005; 40: 279-282.
- 11. Sher KS, Fraser RC, Wicks AC, Mayberry JF. High risk of coeliac disease in Punjabis. Epidemiological study in the south Asian and European populations of Leicestershire. Digestion 1993; 54: 178-182.
- 12. Freeman HJ. Biopsy-defined adult celiac disease in Asian-Canadians. Can J Gastroenterol 2003; 17: 433-436.
- 13. Dickey W, Kearney N. Overweight in celiac disease: prevalence, clinical characteristics, and effect of a gluten-free diet. Am J Gastroenterol 2006; 101: 2356-2359.
- 14. Pittas AG, Lau J, Hu F, Dawson-Hughes B. The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J Clin Endocrinol Metab 2007; 92: 2017-2029.