By 31 December 2005, 22 361 cases of HIV infection had been diagnosed in Australia, primarily transmitted through sexual contact between men. The number of new diagnoses increased by 41% between 2000 and 2005, with an estimated 15 310 people living with HIV/AIDS.1 About 10% of people living with HIV infection in Australia are women, and many of these are in their reproductive years. With reduced morbidity and mortality since the introduction of effective combination antiretroviral (ARV) therapies and interventions available to virtually eliminate mother-to-child transmission, HIV infection now presents far less of a barrier to parenthood for women with HIV infection in developed countries.
Three key interventions have been shown to significantly reduce mother-to-child transmission of HIV:2
avoidance of breastfeeding (since 1985);3
ARV therapy administered antenatally, during labour and to the newborn child (since 1994);4 and
The patterns of perinatal exposure to HIV in Australia up until 1999 and use of interventions for reducing mother-to-child transmission of HIV have previously been reported.7,8 In this study, we aimed to determine the uptake of interventions to reduce mother-to-child transmission of HIV in Australia, with an additional 6 years of observation, and to identify predictors of uptake.
Univariate and multivariate analyses, restricted to women whose HIV diagnosis occurred before delivery, were used to investigate factors that might be associated with avoidance of breastfeeding since 1985, use of zidovudine during pregnancy since 1994, and elective caesarean section since 1999. Both univariate and multivariate analysis was performed using a clustered logistic regression model as women could be included in the study more than once if they had more than one child. Variables that were insignificant and were not confounders were removed from the model. Statistical significance was set at P < 0.05.
Data were analysed using Stata, version 9 (StataCorp, College Station, Tex, USA).
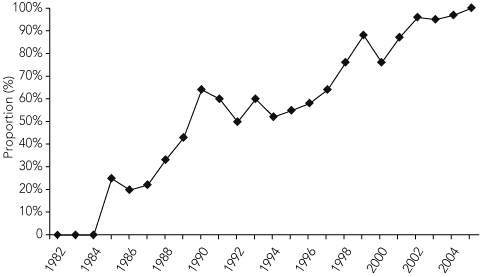
Based on reports received to 31 December 2005, 367 babies were born to 291 mothers with HIV infection in Australia. During this period, 80 mothers were diagnosed with AIDS and 50 mothers died. The proportion of births for which the maternal diagnosis of HIV infection was made antenatally increased steadily over the study period, reaching 100% by 2005 (Box 1).
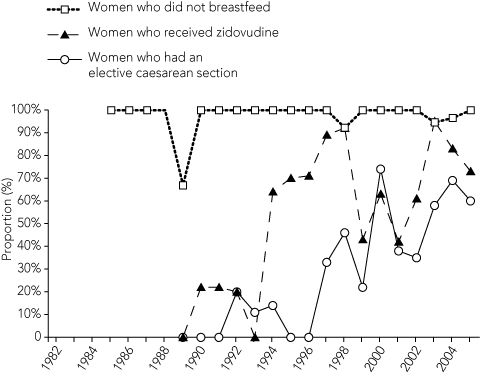
Overall, 255 women in the study population (88%) were diagnosed with HIV before the birth of at least one of their children, and further analysis is based on this group. Avoidance of breastfeeding in this population was at a high level, use of zidovudine during pregnancy increased after 1994, and the proportion of women undergoing elective caesarean section increased after 1999 (Box 2).
In univariate analyses, the year of birth of the child (after 2001) and year of HIV diagnosis in the mother (after 1997) were significantly associated with increased likelihood of use of zidovudine (P < 0.05) (Box 3). Use of zidovudine was less likely if the delivery took place in New South Wales and more likely if it took place in Queensland. Only NSW (adjusted odds ratio [AOR], 0.38; 95% CI, 0.17–0.89; P = 0.02) and the year the child was born (AOR, 3.3; 95% CI, 1.3–8.3; P = 0.01) remained significant in the multivariate model.
For elective caesarean section, year of birth of the child (after 2002) and being non-Indigenous were significantly associated with increased likelihood of uptake, and women were less likely to have an elective caesarean section if delivering in Western Australia (Box 4). The year of birth (AOR, 3.4; 95% CI, 1.1–10.6; P = 0.04) and the location of delivery (AOR, 0.03; 95% CI, 0.01–0.14; P < 0.001) remained significant in the multivariate model.
The data for efficacy and safety of zidovudine during pregnancy have been available since 1994. ARVs reduce mother-to-child transmission of HIV even in women with an HIV viral load < 1000 copies/mL.9 Current United States guidelines10 recommend that women who have never received ARVs should receive zidovudine after the first trimester, regardless of HIV viral load, and that women already on a regimen not including zidovudine should commence it after the first trimester unless clinically contraindicated. Regardless of the antepartum regimen, intravenous zidovudine should be administered during the intrapartum period and to the newborn. In contrast, British guidelines do not recommend intrapartum intravenous zidovudine in women receiving at least three ARVs whose HIV viral load is controlled at < 50 copies/mL.11
Several studies performed before the widespread use of ARVs demonstrated a clear benefit from elective caesarean section.5,6 However, the extent of this benefit in women taking ARVs with an HIV viral load below 50 copies/mL is not clear, and this uncertainty is reflected in current guidelines.10,11 In Australia, there are no national guidelines regarding the preferred mode of delivery in pregnant women with HIV.
Many of the findings reported in this study are consistent with reports from other industrialised countries. The US has also reported that nearly 79% of women received ARVs during pregnancy in areas conducting enhanced surveillance during 1999–2001,12 and that elective caesarean section rates increased (up to 44%) after 1998.13 In the United Kingdom, caesarean section rates of 53% and ARV use of 69% have been reported.14
Received 27 August 2007, accepted 2 March 2008
- Michelle L Giles1,2,3
- Ann M McDonald4
- Elizabeth J Elliott5,6,7
- John B Ziegler8,9
- Margaret E Hellard2,3
- Sharon R Lewin1,3
- John M Kaldor4
- 1 Department of Medicine, Monash University, Melbourne, VIC.
- 2 Centre for Epidemiology and Population Health Research, Burnet Institute, Melbourne, VIC.
- 3 Infectious Disease Unit, Alfred Hospital, Melbourne, VIC.
- 4 National Centre in HIV Epidemiology and Clinical Research, University of New South Wales, Sydney, NSW.
- 5 Discipline of Paediatrics and Child Health, University of Sydney, Sydney, NSW.
- 6 Children’s Hospital at Westmead, Sydney, NSW.
- 7 Australian Paediatric Surveillance Unit, Sydney, NSW.
- 8 Department of Immunology, Sydney Children’s Hospital, Sydney, NSW.
- 9 School of Women’s and Children’s Health, University of New South Wales, Sydney, NSW.
The activities of the APSU are supported by the Australian Government Department of Health and Ageing (National Health and Medical Research Council [NHMRC] Enabling Grant No. 402784 and NHMRC Practitioner Fellowship No. 457084 [Elizabeth Elliott]); Discipline of Paediatrics and Child Health, University of Sydney; and the Royal Australasian College of Physicians.
Sharon Lewin has received funding to attend meetings by Gilead, Pfizer, Roche and Boehringer. She received educational grants from Roche and has sat on advisory boards for Roche and Pfizer.
- 1. National Centre in HIV Epidemiology and Clinical Research. HIV/AIDS, viral hepatitis and sexually transmissible infections in Australia Annual Surveillance Report 2006. Sydney: NCHECR; Australian Institute of Health and Welfare, 2006.
- 2. Brocklehurst P. Interventions for reducing the risk of mother-to-child transmission of HIV infection Cochrane Database Syst Rev 2002; (1): CD000102.
- 3. Ziegler JB, Cooper DA, Johnson RD, et al. Postnatal transmission of AIDS-associated retrovirus from mother to infant. Lancet 1985; 1: 896-898.
- 4. Connor EM, Sperling RS, Gelber R, et al. Reduction of maternal-infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. N Engl J Med 1994; 331: 1173-1180.
- 5. The European Mode of Delivery Collaboration. Elective caesarean-section versus vaginal delivery in prevention of vertical HIV-1 transmission: a randomized clinical trial. Lancet 1999; 353: 1035-1039.
- 6. The International Perinatal HIV Group. The mode of delivery and the risk of vertical transmission of human immunodeficiency virus type 1. N Engl J Med 1999; 340: 977-987.
- 7. McDonald AM, Cruickshank M, Ziegler JB, et al. Perinatal exposure to HIV in Australia, 1982–1994. Med J Aust 1997; 166: 77-80.
- 8. McDonald AM, Li Y, Cruickshank MA, et al. Use of interventions for reducing mother-to-child transmission of HIV in Australia. Med J Aust 2001; 174: 449-452.
- 9. Ioannidis JPA, Abrams EL, Ammann A, et al. Perinatal transmission of human immunodeficiency virus type 1 by pregnant women with RNA virus loads < 1000 copies/mL. J Infect Dis 2001; 183: 539-545.
- 10. Perinatal HIV Guidelines Working Group. Public Health Service Task Force recommendations for use of antiretroviral drugs in pregnant HIV-1-infected women for maternal health and interventions to reduce perinatal HIV-1 transmission in the United States. November 2, 2007. Washington, DC: United States Department of Health and Human Services, 2007.
- 11. British HIV Association. Guidelines for the management of HIV infection in pregnant women and the prevention of mother-to-child transmission of HIV. London: BHIVA, 2005. http://www.bhiva.org/files/file1030325.pdf (accessed Mar 2008).
- 12. Centers for Disease Control and Prevention (CDC). Achievements in public health: reduction in perinatal transmission of HIV infection — United States, 1985–2005. MMWR Morb Mortal Wkly Rep 2006; 55: 592-597.
- 13. Dominguez KL, Lindegren ML, D’Almada PJ, et al. Increasing trend of cesarean deliveries in HIV-infected women in the United States from 1994 to 2000. J Acquir Immune Defic Syndr 2003; 33: 232-238.
- 14. Lyall EG, Stainsby C, Taylor GP, et al. Review of uptake of interventions to reduce mother to child transmission of HIV by women aware of their HIV status. BMJ 1998; 316: 268-270.
- 15. Hall SM, Nicoll A. The British Paediatric Surveillance Unit — a pioneering method for investigating the less common disorders of childhood. Report of a seminar held in June 1995. Child Care Health Dev 1998; 24: 129-143.
- 16. Knowles RL, Smith A, Lynn R, Rahi JS; British Paediatric Surveillance Unit. Using multiple sources to improve and measure case ascertainment in surveillance studies: 20 years of the British Paediatric Surveillance Unit. J Public Health (Oxf) 2006; 28: 157-165.





Abstract
Objective: To analyse the uptake of interventions known to reduce the risk of perinatal HIV transmission among Australian women with HIV infection (who knew their HIV status before delivery), and identify predictors of uptake.
Design: Retrospective analysis of perinatal HIV surveillance data in Australia.
Patients: Women reported as having HIV infection and having given birth to a child (1982–2005) were identified through three mechanisms: an informal network of clinicians (1982–1993); an active surveillance program through paediatricians (since 1993); and state health department reports of children born to women newly diagnosed with HIV (since 1995).
Main outcome measures: Uptake of interventions — avoidance of breastfeeding (after 1985), use of zidovudine during pregnancy (after 1994), and elective caesarean section (after 1999). Factors associated with uptake of these interventions were identified by univariate and multivariate analyses.
Results: 367 live births were reported in 291 women with HIV infection. Among the subgroup diagnosed with HIV infection before delivery, 4/255 (1.6%) elected to breastfeed (post 1985), 44/185 (24%) did not receive zidovudine (after 1994), and 41/118 (35%) did not have an elective caesarean section (after 1999). In multivariate analysis, there were significant differences in uptake of zidovudine and elective caesarean section according to year of birth and state in which the birth took place.
Conclusion: In Australia between 1982 and 2005, uptake of interventions to reduce mother-to-child transmission of HIV was high. There were significant differences associated with use of zidovudine and mode of delivery according to location of delivery and year of birth.