The recommended use of aspirin for the primary prevention of cardiovascular disease is based on absolute cardiovascular event risk. Age is the greatest determinant of absolute risk, and yet few participants in major primary prevention trials have been elderly. ASPirin in Reducing Events in the Elderly (ASPREE) is a double-blind, randomised, placebo-controlled study of low-dose aspirin for the primary prevention of major adverse and cardiovascular events in older people. The research questions tackle areas of major public health importance.1 The rationale for the conduct of ASPREE and its protocol have been published elsewhere.2,3
GP co-investigators were purposefully sampled from those who had participated in the Second Australian National Blood Pressure Study (ANBP2) in Melbourne.4 GPs were excluded if they did not have Medical Director clinical software (Health Communications Network, Sydney, NSW), as research nurses were trained to prescreen practice databases by participant inclusion and exclusion criteria on this clinical software. GPs vetted these databases to exclude deceased patients and those they considered unsuitable or not their usual patients.
The identification and enrolment of participants is illustrated in Box 1. The initial step of using additional search criteria in computerised medical records enabled the exclusion of ineligible patients (Box 2). For example, 27% of the general practice population screened were identified as taking aspirin or another anticoagulant. Baseline characteristics of the final 209 participants are shown in Box 3. In general, this was an active, healthy group of men and women aged 70 years and over.
Box 4 shows clinical measurements, neuropsychological testing, and quality-of-life measurements at baseline and at the 12-month follow-up visit, both overall and for participants randomly allocated to receive aspirin or placebo. After 12 months of treatment, there were significant increases in systolic and diastolic blood pressures and GDS scores, and significant reductions in haemoglobin and blood glucose levels, and in waist circumference in the aspirin group. There were no differences between aspirin and placebo groups in changes from baseline in the levels of cognitive function or independent activities. Haemoglobin and glucose levels were significantly lower (P < 0.05) and blood pressures higher (P < 0.05) in the aspirin-treated participants compared with the placebo-treated group. Health behaviour measures at baseline and at 12 months are shown in Box 5.
There are a number of important findings from this pilot study that suggest that conducting an 18 000-participant general-practice-based study in Australia is feasible. Firstly, computer-based querying of general practice records to identify potential study participants was highly efficient. In our ASPREE feasibility study, instant identification of age-eligible patients and the exclusion of 41% of these by searching for further exclusion criteria (Box 2) significantly reduced recruitment costs. In our previous large-scale general practice initiative (ANBP2), all databases were generated by hand, and age was the only criterion on which letters of invitation were sent.6 Secondly, the 23% response rate to the letter of invitation is similar to that reported in ANBP2, and reflects a willingness of older people to participate in clinical research. Finally, and most importantly, 86% of those who entered the run-in phase were randomly allocated to receive aspirin or placebo in this ASPREE pilot study. On this basis, and allowing for lower response rates, about 30 000 people aged 70 years and older would need to be entered into the run-in phase to achieve the recruitment target in the main trial (Box 1). Given that 53 242 participants entered into the screening phase of ANBP2 within the 2.5-year recruitment period, the screening target of 30 000 is highly likely to be achievable within a 2-year recruitment phase in the main ASPREE study.
Low-dose aspirin therapy has been shown to reduce the risk of vascular events in those with manifest atherosclerotic disease.7 In primary prevention, atherosclerotic events can also be reduced.3,8-13 In addition, there is increasing evidence of the potential, as yet unproven, of low-dose aspirin therapy to reduce the rate of intellectual decline in older people.14-16 However, part of the benefit of aspirin therapy may be offset by adverse effects, including gastrointestinal and intracranial bleeding. Epidemiological modelling suggests that the routine use of aspirin in people aged 70 years and over who do not have manifest disease has an uncertain balance of risk and benefit, and a trial such as ASPREE is therefore warranted.17 Because the risks and benefits have not been established in older participants, aspirin is not widely used for primary prevention in Australian general practice. In our screened population, only 27% of patients were currently taking aspirin or another anticoagulant (Box 2). This population estimate needs to be interpreted with caution, as it included patients taking aspirin for both primary and secondary prevention, and aspirin is available over the counter, and hence may not be recorded consistently in GPs’ records. The use of aspirin in the community is also likely to increase, given its cardioprotective effects and the recent evidence of cardiotoxicity of other non-selective and selective non-steroidal anti-inflammatory drugs.18
The 80% compliance level at 12 months observed in our ASPREE pilot study is consistent with the 81% at 1 year of those randomly assigned to receive aspirin in the United Kingdom general-practice-based Primary Prevention Project (PPP).13 Experience from PPP and other primary prevention trials suggests that participants stop taking trial medication predominantly in the first year, with relatively low rates of cessation thereafter. The significant reduction in haemoglobin level observed in the active treatment arm of ASPREE was also found in the Prevention with low-dose Aspirin of Cardiovascular disease in the Elderly study.19 Such an outcome may be trivial in younger, more robust populations, but is likely to be important in older people in whom comorbidity is common. For example, occult blood loss may lead to the development of cardiac failure.20 Nearly one in five of the cohort of our ASPREE feasibility study were aged 80 or more years. While these numbers will be underpowered for subgroup analysis by age band, it is likely to give trends if the benefits and harms are different in the very old.
The observed significant increase in systolic blood pressure in the aspirin group may be explained as an association between aspirin and hypertension, particularly in women.21 However, no effect was reported in our own previous pilot study, in any of the six main primary prevention trials or their meta-analyses, or in the Antithrombotic Trialists Group secondary prevention meta-analysis.8-13,22,23
Expert groups have also called for the conduct of a trial such as ASPREE.24,25 In December 2003, the United States Food and Drug Administration (FDA) Cardiovascular and Renal Advisory Committee voted against a labelling indication for aspirin that was based on assessment of absolute risk of coronary heart disease, for which age is the major determinant.26 The FDA Committee called for a trial to provide the evidence for aspirin use in individuals at moderate cardiovascular risk.
1 Flow chart of screening and random allocation of participants for the ASPirin in Reducing Events in the Elderly pilot study
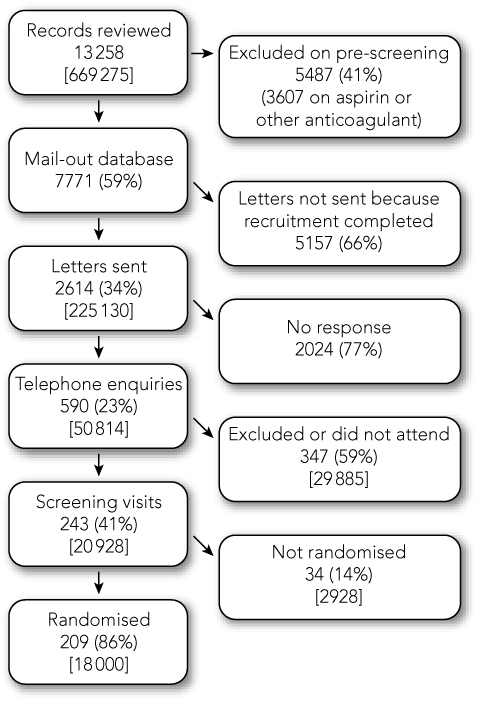 | |||||||||||||||
|
Figures in square parentheses are projected numbers for the main study. | |||||||||||||||
2 Pre-screening for exclusion criteria on general practice computer databases
Patients excluded at pre-screening on exclusion criteria (below) |
|||||||||||||||
Patients excluded at pre-screening because they were taking aspirin or anticoagulants |
|||||||||||||||
4 Clinical measurements, neuropsychological and quality-of-life test scores* at baseline and 12 months, overall and by treatment group for the 192 participants who returned for 12-month follow-up
Received 2 December 2007, accepted 27 April 2008
- Mark R Nelson1
- Christopher M Reid2
- David Ames3
- Lawrence J Beilin4
- Geoffrey A Donnan5
- Peter Gibbs6
- Colin I Johnston7
- Henry Krum2
- Elsdon Storey8
- Andrew Tonkin2
- Rory Wolfe2
- Robyn Woods2
- John J McNeil2
- 1 Menzies Research Institute, University of Tasmania, Hobart, TAS.
- 2 Department of Epidemiology and Preventive Medicine, Monash University, Melbourne, VIC.
- 3 Department of Psychiatry, University of Melbourne, Melbourne, VIC.
- 4 Department of Internal Medicine, University of Western Australia, Perth, WA.
- 5 Department of Neurology, University of Melbourne, Melbourne, VIC.
- 6 Ludwig Institute for Cancer Research, Melbourne, VIC.
- 7 Baker Heart Research Institute, Melbourne, VIC.
- 8 Van Cleef/Roet Centre for Nervous Diseases, Monash University, Melbourne, VIC.
This feasibility study received support from a grant of the National Heart Foundation of Australia. ASPREE has also received funding from the National Health and Medical Research Council (NHMRC) and is endorsed by the National Stroke Foundation, the National Heart Foundation of Australia, the Australian Divisions of General Practice, and Alzheimer’s Australia. Bayer HealthCare is supplying enteric-coated low-dose aspirin and placebo. We would also like to acknowledge Robyn Woods, Executive Officer of the ASPREE study, Louise Shiel for database management, and research nurses Kay Murphy, Anne Bruce, and Anne Lyons. We would also like to thank the AusDiab investigators for use of their data for community absolute cardiovascular event risk estimations. We also acknowledge the people on the following committees and our general practitioner co-investigators:
Steering committee: David Ames, Geoffrey A Donnan (University of Melbourne); Lawrence J Beilin (University of Western Australia); Colin I Johnston (Baker Heart Research Institute); Henry Krum, John J McNeil, Christopher M Reid, Elsdon Storey, Andrew Tonkin, Robyn Woods (Monash University); Mark R Nelson (University of Tasmania).
International advisory committee: Colin Baigent (University of Oxford); John CS Breitner (University of Washington); Christopher J Bulpitt (Imperial College London); Barry R Davis, Robert G Hart (University of Texas); Charles H Hennekens (University of Miami); Christophe Tzourio (Hôpital de la Salpêtrière).
General practice advisory committee: Nigel Stocks (University of Adelaide); Michael Kidd (University of Sydney); Mark R Nelson (University of Tasmania); Leon Piterman (Monash University); Jonathon Emery (University of Western Australia); Claire Jackson (University of Queensland).
Endpoint committee: Francis Dudley, Elsdon Storey (Monash University); Graham Giles (The Cancer Council Victoria); Trefor Morgan, Geoffrey A Donnan (University of Melbourne).
Data monitoring and safety committee: Lindon M H Wing (Flinders University); Graeme Hankey (University of Western Australia); Philip Ryan (University of Adelaide); Charles H Hennekens (University of Miami).
General practitioner co-investigators: Peter Arndt, Andrew Batty, Robert Brack, Peter Campbell, David Court, Vince Galteri, Robert Gingold, Abby Hechtman, Leon Lewi, Christopher McHardy, Margaret McNiff, Peter Meyer, Paul O’Hanlon and Stephen Parker.
Mark Nelson, Christopher Reid and Andrew Tonkin have received travel support from Bayer HealthCare, who are providing the aspirin and placebo for this trial. Bayer HealthCare has also provided an educational grant to support the roll-out of the main trial.
- 1. McNeil JJ, Nelson MR, Tonkin AM. Public funding of large-scale clinical trials in Australia. Med J Aust 2003; 179: 519-520. <MJA full text>
- 2. Nelson MR, Reid CM, Beilin LJ, et al; ASPREE Study Group. Rationale for a primary prevention trial of low dose aspirin for major adverse cardiovascular events and vascular dementia in the elderly: ASPirin in Reducing Events in the Elderly (ASPREE). Drugs Aging 2003; 20: 897-903.
- 3. McNeil JJ, Woods R. ASPirin in Reducing Events in the Elderly. Protocol. http://www.med.monash.edu.au/epidemiology/downloads/aspree-protocol-060308.pdf (accessed Jun 2008).
- 4. Reid C, Ryan P, Nelson M, et al. General practitioner participation in the Second Australian National Blood Pressure Study (ANBP2). Clin Exp Pharmacol Physiol 2001; 28: 663-667.
- 5. D’Elia LF, Satz P, Uchiyama CL, et al. Color Trails Test. Odessa, Fla: Psychology Assessment Resources, 1999.
- 6. Wing LM, Reid CM, Ryan P, et al. A comparison of outcomes with angiotensin-converting-enzyme inhibitors and diuretics for hypertension in the elderly. N Engl J Med 2003; 348: 583-592.
- 7. Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 2002; 324: 71-86.
- 8. Peto R, Gray R, Collins R, et al. Randomised trial of prophylactic daily aspirin in British male doctors. Br Med J (Clin Res Ed) 1988; 296: 313-316.
- 9. Steering Committee of the Physicians’ Health Study Research Group. Final report on the aspirin component of the ongoing Physicians’ Health Study. N Engl J Med 1989; 321: 129-135.
- 10. Hansson L, Zanchetti A, Carruthers SG, et al. Effects of intensive blood-pressure lowering and low dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. Lancet 1998; 351: 1755-1762.
- 11. The Medical Research Council’s General Practice Research Framework. Thrombosis prevention trial: randomised trial of low-intensity oral anticoagulation with warfarin and low-dose aspirin in the primary prevention of ischaemic heart disease in men at increased risk. Lancet 1998; 351: 233-241.
- 12. Hart RG, Halperin JL, McBride R, et al. Aspirin for the primary prevention of stroke and other major vascular events: meta-analysis and hypotheses. Arch Neurol 2000; 57: 326-332.
- 13. Collaborative Group of the Primary Prevention Project (PPP). Low-dose aspirin and vitamin E in people at cardiovascular risk: a randomised trial in general practice. Lancet 2001; 357: 89-95.
- 14. McGeer PL, Schulzer M, McGeer EG. Arthritis and anti-inflammatory agents as possible protective factors for Alzheimer’s disease; a review of 17 epidemiologic studies. Neurology 1996; 47: 425-432.
- 15. Broe GA, Grayson DA, Creasey HM, et al. Anti-inflammatory drugs protect against Alzheimer disease at low doses. Arch Neurol 2000; 57: 1586-1591.
- 16. in ’t Veld BA, Ruitenberg A, Hofman A, et al. Nonsteroidal antiinflammatory drugs and the risk of Alzheimer’s disease. N Engl J Med 2001; 345: 1515-1521.
- 17. Nelson MR, Liew D, Bertram M, Vos T. Epidemiological modelling of routine use of low dose aspirin for the primary prevention of coronary heart disease and stroke in those aged 70. BMJ 2005; 330: 1306-1308.
- 18. Nelson MR, Tonkin AM, Cicuttini FM, McNeil JJ. COX-2 inhibitors: exemplars of the drug-safety conundrum [editorial]. Med J Aust 2005; 182: 262-263. <MJA full text>
- 19. Silagy CA, McNeil JJ, Donnan GA, et al. Adverse effects of low-dose aspirin in a healthy elderly population. Clin Pharmacol Ther 1993; 54: 84-89.
- 20. Silverberg DS, Wexler D, Sheps D, et al. The effect of correction of mild anemia in severe, resistant congestive heart failure using subcutaneous erythropoietin and intravenous iron: a randomized controlled study. J Am Coll Cardiol 2001; 37: 1775-1780.
- 21. Dedier J, Stampfer MJ, Hankinson SE, et al. Nonnarcotic analgesic use and the risk of hypertension in US women. Hypertension 2002; 40: 604-608.
- 22. Hayden M, Pignone M, Phillips C, Mulrow C. Aspirin for the primary prevention of cardiovascular events: a summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2002; 136: 161-172.
- 23. Ridker PM, Cook NR, Lee I-M, et al. A randomized trial of low-dose aspirin in the primary prevention of cardiovascular disease in women. N Engl J Med 2005; 352: 1293-1304.
- 24. Patrono C, Garcia Rodriguez LA, Landolfi R, Baigent C. Low-dose aspirin for the prevention of atherothrombosis. N Engl J Med 2005; 353: 2373-2383.
- 25. Hennekens CH, Hollar D, Baigent C. Sex-related differences in response to aspirin in cardiovascular disease: an untested hypothesis. Nat Clin Pract Cardiovasc Med 2006; 3: 4-5.
- 26. United States of America Food and Drug Administration, Centre for Drug Evaluation and Research. Cardiovascular and Renal Drugs Advisory Committee meeting. 8 Dec 2003. http://www.fda.gov/ohrms/dockets/ac/03/transcripts/4012T1.htm (accessed Jun 2008).





Abstract
Aim: To determine the feasibility of performing a large clinical trial of the use of aspirin for the primary prevention of cardiovascular disease in older participants — the ASPirin in Reducing Events in the Elderly (ASPREE) trial.
Design and participants: A randomised double-blind placebo-controlled pilot trial of 100 mg of enteric-coated aspirin tablets daily, in men and women aged 70 years and over who did not have overt cardiovascular disease, and who were followed for 12 months. Participants were identified from the computer databases of general practitioners who were co-investigators in a previous trial.
Setting: The Melbourne metropolitan area between March 2003 and June 2005.
Main outcome measures: The level of response to participation by GPs; the level of response from potential trial participants; the screening-to-randomisation rate to ensure the recruitment target could be achieved; and the retention of participants in the trial after 12 months.
Results: Forty-two GPs (23% of 180 mailed) expressed interest in participating in the pilot trial. Nineteen became co-investigators, of whom six were not required to meet recruitment targets. Letters were sent to 2614 patients, of whom 243 were screened and 209 (86%) were randomly allocated to receive aspirin or placebo. At 12 months,192 (92%) returned for follow-up, and 153 of these (80%) were still taking trial medication. There was a significant reduction in mean haemoglobin level in those taking aspirin.
Conclusions: The recruitment strategy for ASPREE, based on methods developed for the conduct of a previous large-scale trial conducted in general practice, was successfully redeployed in this pilot study, with improved efficiency resulting from computerised database searching, telephone pre-screening, a simpler run-in phase and participant familiarity with the trial drug. We conclude that conducting ASPREE in Australian general practice with 18 000 participants is feasible.
Trial registration: International Standard Randomised Controlled Trial Number Register ISRCTN83772183.