In 2005, an estimated 8.8 million new cases of tuberculosis (TB) worldwide caused 1.6 million deaths.1 However, Australia2 and other developed countries1,3 have relatively low TB incidence rates, with most new cases occurring in migrants. The most reliable predictor of TB incidence in Australia is the TB prevalence in countries from where migrants are accepted.4 TB incidence in people migrating to developed countries is highest in the first 2 years after migration, although it remains high for up to a decade after arrival.5-10
Control of TB in Australia is facilitated through entry screening of migrants using chest x-ray, and contact tracing of people with active TB.11 However, this strategy is inadequate for detecting latent and non-pulmonary disease and is unlikely to identify migrants whose TB will be diagnosed many years after their arrival.
We chose 10 years after arrival as the cut-off point for analysis, as TB may be less likely to be considered in the differential diagnosis in migrants a decade after arrival. This cut-off also allowed comparison with other studies.6,8-9
Time between arrival in Australia and diagnosis was examined for migrants from specific regions, and a multivariate logistic regression model was constructed to compare those diagnosed 10 or more years after arrival with those diagnosed within 10 years of arrival. Independent variables in the model were year of diagnosis, sex, age group, clinical site (pulmonary or non-pulmonary), drug resistance, and region of birth, all of which were associated with time to diagnosis at the univariate level (P < 0.1) (Box 1).
Data were analysed using Stata, version 8.0 (StataCorp, College Station, Tex, USA).
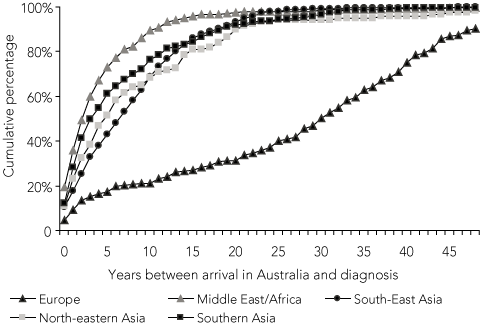
Time between arrival in Australia and diagnosis of TB was available for 1924 of the patients born overseas (83%). The median time to diagnosis was 6 years (interquartile range, 2–14 years), but 38% (734) of all migrants with complete data were diagnosed 10 or more years after arrival. Most patients born in the Middle East/Africa and Asia (86% and 71%, respectively) were diagnosed within 10 years of arrival, compared with 21% of Europeans (Box 2).
Logistic regression analysis indicated that, compared with migrants from South-East Asia (the largest group of migrants), migrants diagnosed 10 or more years after arrival were more likely to be diagnosed during 2000–2004, to be aged ≥ 35 years (with the risk increasing with age), and to be born in Europe. They were less likely to have been born in the Middle East/Africa, north-eastern Asia or southern Asia. There was no association between years to diagnosis and sex, site of infection or drug resistance (Box 1).
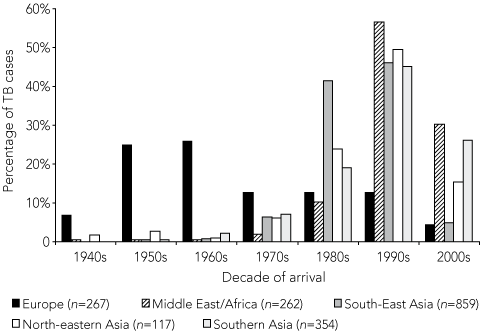
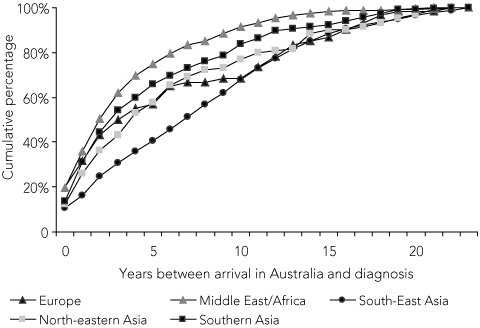
European patients in the study population have been arriving in Australia since the 1940s, when Europeans represented the majority of Australia’s immigrants (Box 3). By contrast, patients from other migrant groups have been arriving mostly since the 1970s or 1980s, when immigration policies changed in Australia. To determine whether these changes in immigration may explain the pattern of diagnosis for European migrants, we compared time between arrival and diagnosis for migrants by region of birth for the total study population (Box 2), and for only those migrants who arrived in Australia after 1980 (Box 4). Only 21% of European migrants in the total study population were diagnosed within a decade of arrival, compared with 73% of all other migrant groups combined (P < 0.001) (Box 2). However, of migrants arriving in Australia after 1980, 68% of Europeans were diagnosed within a decade of arrival, similar to the proportion of all other groups (76%) (P = 0.151) (Box 4).
We found that over a third of TB cases in migrants were diagnosed a decade or more after their arrival in Australia. This proportion represents an increase from the 20% previously reported for cases diagnosed between 1948 and 1992 in Victoria.9 This finding is consistent with the experience in other developed countries, where the risk of active TB continues beyond the immediate post-migration period.5-8,10
Overseas-born patients diagnosed a decade or more after arrival in Australia were more likely to be from Europe, mainly from countries in the EU and Western Europe. Similar to Australian-born TB patients2 and patients diagnosed in the EU/Western Europe in 2004,12 they were also more likely to be older. The older generations of European and Australian-born populations have a higher relative prevalence of latent infection because they were born at a time when the risk of TB infection was relatively high, and they have had a long cumulative probability of acquiring infection.3 This is also true for people born in North America.13
The second group of European migrants, those from Central or Eastern Europe, were younger at the time of diagnosis with active TB and formed the majority of European patients diagnosed within 10 years of arrival in Australia. This is similar to patients born in Asia and Africa and reflects the current higher incidence of TB in their home countries.13
Relatively little TB transmission from migrants to the broader community has been demonstrated in Australia and other low-incidence countries, and most TB diagnoses are due to reactivation of latent TB infection.4,14,15 In Victoria, genetic typing of all new TB isolates has occurred since 1993, and, apart from limited transmission within immigrant family groups and domestic contacts (including first-generation Australian-born children), most cases of TB in migrants who have been in Australia for 10 or more years have involved unique strains (D L, unpublished data).
Further efforts to improve detection and treatment of latent TB infection, along with the realisation that the risk of infection persists beyond the first decade after migration, may help to minimise the burden of TB in Australia. Additional strategies that include more sensitive screening techniques for latent TB, such as the Mantoux test and the second-generation interferon-γ release assays including QuantiFERON (Cellestis International, Melbourne, Vic) and enzyme-linked immunosorbent spot (ELISPOT) tests,16 should be introduced into the migrant screening program. Refugees and migrants from high-prevalence areas should be screened for latent TB on arrival, offered treatment as appropriate, and followed up beyond 10 years. A further control strategy, as recommended by the European framework for tuberculosis control and elimination in countries with low incidence,3 is a global investment to improve TB control in high-incidence countries and thus reduce the international transfer of risk.
1 Comparison between migrants diagnosed with tuberculosis less than 10 years after arrival with those diagnosed 10 or more years after arrival in Australia, Victoria, 1990–2004
2 Cumulative percentage of years between arrival in Australia and diagnosis of tuberculosis, by region of birth for all migrants (n = 1924)
- Michelle E McPherson1,2
- Heath Kelly3
- Mahomed S Patel2
- David Leslie3
- 1 Communicable Disease Control, Department of Human Services, Melbourne, VIC.
- 2 National Centre for Epidemiology and Population Health, Australian National University, Canberra, ACT.
- 3 Victorian Infectious Diseases Reference Laboratory, Melbourne, VIC.
We acknowledge the tuberculosis nurses at the Department of Human Services, especially Lynne Brown, for their work. We also acknowledge Dr Lily Yuen and Dr Paul Vinton for their contribution to earlier analyses of the epidemiology of laboratory-confirmed tuberculosis in Victoria.
None identified.
- 1. World Health Organization. Global tuberculosis control — surveillance, planning, financing. Geneva: WHO, 2007. http://www.who.int/tb/publications/global_report/en/ (accessed Mar 2008).
- 2. Roche P, Bastian I, Krause V; National Tuberculosis Advisory Committee. Tuberculosis notifications in Australia, 2005. Commun Dis Intell 2007; 31: 71-80.
- 3. Broekmans JF, Migliori GB, Rieder HL, et al. European framework for tuberculosis control and elimination in countries with low incidence. Recommendations of the World Health Organization, International Union Against Tuberculosis and Lung Disease and Royal Netherlands Tuberculosis Association Working Group. Eur Respir J 2002; 19: 765-775.
- 4. Watkins RE, Plant AJ. Predicting tuberculosis among migrant groups. Epidemiol Infect 2002; 129: 623-628.
- 5. Farah MG, Meyer HE, Selmer R, et al. Long-term risk of tuberculosis among immigrants in Norway. Int J Epidemiol 2005; 34: 1005-1011.
- 6. Cohen T, Murray M. Incidence tuberculosis among recent US immigrants and exogenous reinfection. Emerg Infect Dis 2005; 11: 725-728.
- 7. Lillebaek T, Anderson AB, Dirksen A, et al. Persistent high incidence of tuberculosis in immigrants in a low-incidence country. Emerg Infect Dis 2002; 8: 679-684.
- 8. Vos AM, Meima A, Verver S, et al. High incidence of pulmonary tuberculosis persists a decade after immigration, The Netherlands. Emerg Infect Dis 2004; 10: 736-739.
- 9. MacIntyre CR, Dwyer B, Streeton JA. The epidemiology of tuberculosis in Victoria. Med J Aust 1993; 159: 672-677.
- 10. Cowie RL, Sharpe JW. Tuberculosis among immigrants: interval from arrival in Canada to diagnosis. A 5-year study in southern Alberta. CMAJ 1998; 158: 599-602.
- 11. Victorian Government Department of Human Services. Management, control and prevention of tuberculosis. Guidelines for health care providers (2002–2005). Melbourne: DHS, 2002. http://www.health.vic.gov.au/ideas/diseases/tb_mgmt_guide#pdf (accessed Jan 2008).
- 12. EuroTB and the national coordinators for tuberculosis surveillance in the WHO European Region. Surveillance of tuberculosis in Europe. Report on tuberculosis cases notified in 2004. Saint-Maurice, France: Institut de Veille Sanitaire, 2006. http://www.eurotb.org/rapports/2004/table_cit04.pdf (accessed Mar 2008).
- 13. Dye C. Global epidemiology of tuberculosis. Lancet 2006; 367: 938-940.
- 14. Yuen LK, Leslie D, Coloe PJ. Bacteriological and molecular analysis of rifampin-resistant Mycobacterium tuberculosis strains isolated in Australia. J Clin Microbiol 1999; 37: 3844-3850.
- 15. Dasgupta K, Menzies D. Cost-effectiveness of tuberculosis control strategies among immigrants and refugees. Eur Respir J 2005; 25: 1107-1116.
- 16. Connell TG, Rangaka MX, Curtis N, Wilkinson RJ. QuantiFERON-TB Gold: state of the art for the diagnosis of tuberculosis infection? Expert Rev Mol Diagn 2006; 6: 663-677.





Abstract
Objective: To examine the risk of tuberculosis (TB) in migrants a decade after their arrival in Australia.
Design, setting and patients: Retrospective review of laboratory-confirmed cases of TB in migrants diagnosed between 1990 and 2004 by the state TB reference laboratory in Victoria, analysed by a multivariate model comparing migrants diagnosed 10 or more years after arrival with those diagnosed within 10 years of arrival.
Main outcome measures: Time to diagnosis; characteristics of migrants diagnosed with TB, including age, sex, region of birth, site of infection, and drug resistance.
Results: A third of migrants (734/1924) were diagnosed with TB 10 or more years after arrival in Australia. This group was more likely to be European-born (adjusted odds ratio [AOR], 3.4; 95% CI, 2.4–5.0) and older than 34 years (35–49 years: AOR, 3.8; 95% CI, 2.0–7.0), reflecting the longer time European migrants have been in Australia. There were two distinct European groups: European Union (EU)/Western and Central/Eastern. The Central/Eastern group were from countries with current high TB rates and, compared with the EU/Western group, were younger (mean age, 50 v 64 years) and more likely to be diagnosed within 10 years of arrival (47% v 14%; P < 0.001).
Conclusion: European migrants were more likely to be diagnosed a decade or more after arrival in Australia. Once migrants from the currently high TB incidence areas of Asia and Africa have been in Australia for a similar period of time, their timing of diagnosis may resemble that for migrants from Europe. The current screening policy should be complemented with more sensitive techniques to detect latent TB.