Paracetamol is the most widely used over-the-counter analgesic agent in the world. It is involved in a large proportion of accidental paediatric exposures and deliberate self-poisoning cases and is the leading pharmaceutical agent responsible for calls to poisons information centres in Australia and New Zealand. Paracetamol is also the single most commonly taken drug in overdoses that lead to hospital presentation and admission.1 Hepatic failure and death are uncommon outcomes,2,3 although paracetamol remains the most important single cause of acute fulminant hepatic failure in Western countries.4
Acute deliberate self-poisoning, accidental paediatric exposure and inadvertent repeated supratherapeutic ingestions all require specific approaches to risk assessment and management. This complexity is compounded by the existence of several different treatment nomograms in Australasia. Existing recommendations do not deal with extended-release paracetamol, and previously distributed industry-generated guidelines (from GlaxoSmithKline Consumer Healthcare [GSK]) no longer reflect current clinical toxicology practice or poison centre recommendations.To rectify this problem, new consensus guidelines have been drafted by a panel of Australian and New Zealand clinical toxicologists, with the objective of supporting clinicians in poison centres, primary care and hospital practice with concise and meaningful clinical management guidelines for presentations related to paracetamol overdose. Here, we summarise the rationale for the recommendations made in the guidelines (which are available in full at http://www.toxinz.com).
Paracetamol is rapidly absorbed from the small intestine. Peak serum concentrations occur within 1–2 hours for standard tablet or capsule formulations and within 30 minutes for liquid preparations. Peak serum concentrations after therapeutic doses do not usually exceed 130 μmol/L (20 mg/L). Twenty per cent of the ingested dose undergoes first-pass metabolism in the gut wall (sulphation). Distribution is usually within 4 hours of ingestion for standard preparations and 2 hours for liquid preparations. Volume of distribution is 0.9 L/kg.2
Further elimination occurs by hepatic biotransformation. After therapeutic doses, the elimination half-life is 1.5–3 hours. About 90% is metabolised to inactive sulphate and glucuronide conjugates that are excreted in the urine. Metabolism of the remainder is via cytochrome P450 (chiefly 2E1 and 3A4) and results in the highly reactive intermediary compound N-acetyl-p-benzoquinone imine (NAPQI). In normal conditions, NAPQI is immediately bound by intracellular glutathione and eliminated in the urine as mercapturic adducts.3
With increased paracetamol doses, greater production of NAPQI may deplete glutathione stores. When glutathione depletion reaches a critical level (thought to be about 30% of normal stores), NAPQI binds to other proteins, causing damage to the hepatocyte. Glutathione depletion itself may also be injurious.3
Immediate threats to the airway, breathing and circulation are extremely rare in isolated paracetamol overdose. In exceptional cases, massive ingestion causing extremely high serum paracetamol levels (ie, > 5000 μmol/L or 800 mg/L) may be associated with an early decrease in level of consciousness and with lactic acidosis.5 Supportive management is appropriate in such cases, with N-acetylcysteine administered in routine doses, although prolonged infusions may be required. Haemodialysis has been described in this setting, but indications for its use have not been defined. Recovery is usual with supportive care.
The dosing threshold at which hepatic injury occurs after supratherapeutic paracetamol ingestion appears to be subject to wide interindividual variation and depends on the dosing context (Box 1). Serum paracetamol levels should be used to assess the need for N-acetylcysteine administration in all patients with deliberate paracetamol self-poisoning, regardless of the stated dose.
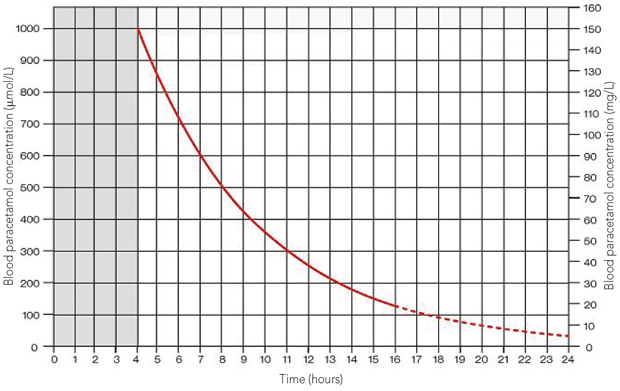
Paracetamol treatment nomograms have been used for many years in Australasia. The nomogram used appears to be a local decision,6 but they are often derived from nomograms devised overseas.7,8
The Prescott nomogram was based on a cohort of patients in Ed-inburgh and extends from 1320 μmol/L (200 mg/L) at 4 hours to 200 μmol/L (30 mg/L) at 15 hours.7 The Rumack–Matthew nomogram is based on the same data, but extrapolated to 24 hours. It also uses a “treatment line” that is plotted 25% lower (1000 μmol/L [150 mg/L] at 4 hours) to comply with a United States Food and Drug Administration requirement to provide a “safety buffer” for research and clinical purposes.8
The efficacy and safety of dosing N-acetylcysteine according to the Rumack–Matthew nomogram has been demonstrated in a study of more than 11 000 patients, with no deaths among patients who were treated within 15 hours.8 In contrast, while use of the higher Prescott line has been demonstrated to be usually safe in smaller cohorts of patients,7,9 there have been occasional reports of people who were not treated because they had concentrations below the line, but who subsequently died from acute hepatic failure.10 In addition, both these nomograms use a log scale, making it difficult to plot levels accurately.7,8
Several variables (such as chronic ethanol misuse, use of enzyme-inducing drugs, prolonged fasting, and dehydration) are hypothesised to increase the risk of hepatic injury. These “risk factors” are thought to increase the metabolism of paracetamol to the toxic metabolite NAPQI via induction of mixed function oxidases, or decrease hepatocellular glutathione stores, or both. However, the relevance of these risk factors in clinical practice remains controversial, with no consensus among clinical toxicologists.11,12 Many guidelines have recommended an arbitrary further lowering of the nomogram line (ie, to 660 μmol/L [100 mg/L] at 4 hours) for patients with such risk factors.13 This introduces further complexity into clinical risk assessment. It is our experience that clinical assessment of risk factors is done in a haphazard fashion and the “high-risk” line on the nomogram is often misinterpreted, with some clinicians treating everyone above this line, and some never using it at all.6
The aim of the new treatment nomogram, shown in Box 2, is to reconcile the hypotheses and evidence regarding risk factors while using a single nomogram line to simplify decision making. The new nomogram lowers the old Australian nomogram line13 by 25%, meaning that it starts at 1000 μmol/L (150 mg/L) at 4 hours. This provides both a margin of safety for patients who may possess risk factors and a small margin of error for estimation of time of ingestion, and avoids the need for potentially confusing additional lines. This change simplifies management guidelines, remains conservative and poses minimal inherent risk for misinterpretation and error. It is also the treatment threshold with the most clinical data to support its efficacy and safety.8
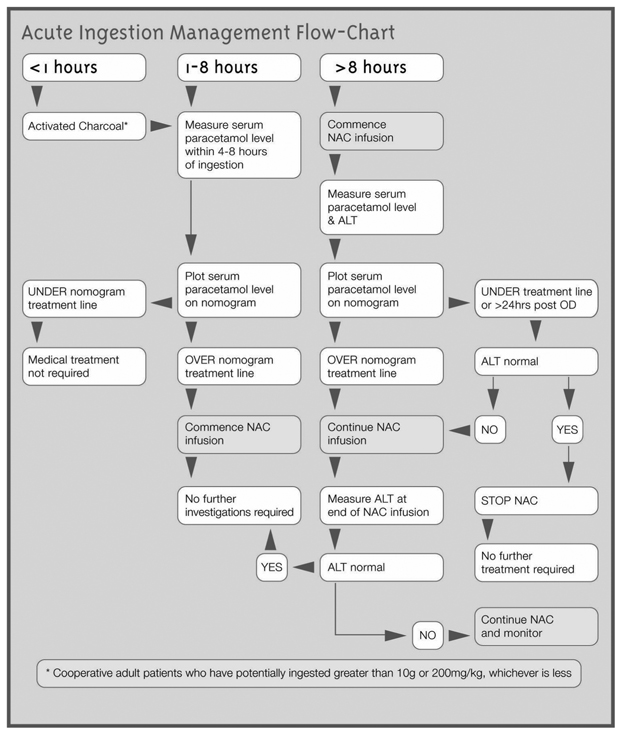
In adults, activated charcoal administered within 1–2 hours of ingestion reduces the absorbed paracetamol dose and the likelihood that N-acetylcysteine will subsequently be required.14 Nevertheless, if activated charcoal cannot be administered, treatment with N-acetylcysteine within 8 hours guarantees survival in any case. Therefore, activated charcoal alone is not a life-saving treatment that may be imposed under a duty-of-care principle. We recommend administration of 50 g activated charcoal only in cooperative adults who can receive the dose within 1–2 hours of paracetamol ingestion.
Treatment with N-acetylcysteine guarantees survival if administered within 8 hours of paracetamol ingestion, and outcome is the same regardless of when treatment is given within this 8-hour window. Beyond 8–10 hours after ingestion, efficacy decreases with increasing delay to treatment.8 If a patient presents within 8 hours of ingestion, N-acetylcysteine administration may be delayed until a serum paracetamol level plotted on the nomogram confirms it is indicated (as long as treatment can still be commenced within the 8-hour window if it is required).
In patients who present 8 or more hours after ingestion, N-acetylcysteine should be commenced immediately if the reported dose exceeds the threshold for possible toxicity (Box 1) or the patient shows clinical signs suggestive of paracetamol toxicity (nausea, vomiting, right upper quadrant pain or tenderness). Evaluation of serum paracetamol and alanine aminotransferase (ALT) levels should then be performed as soon as possible. If the serum paracetamol level is subsequently found to be below the nomogram line, N-acetylcysteine may be ceased; if above the line, it should be continued. The baseline serum ALT level assists risk assessment and provides useful baseline data if N-acetylcysteine is indicated. Similarly, if N-acetylcysteine is commenced, baseline international normalised ratio and platelet count provide additional data to inform later risk assessments (eg, for risk of death from hepatic failure).
Box 3 summarises the steps for management of acute paracetamol exposure with known time of ingestion.
If the time of ingestion is unknown, or the treating clinician is not confident of the history of ingestion, it is safest to treat the patient as a delayed presentation. Thus, the recommendation is to follow the > 8 hours scenario in Box 3.
The kinetics of sustained-release paracetamol preparations after deliberate self-poisoning have not been defined. Studies in volunteers confirm there is a potential for slow absorption and thus a delayed peak serum paracetamol concentration above the nomogram line.15
In this scenario, if it has been less than 8 hours since the first dose, the patient can safely be treated as per the 1–8 hours scenario in Box 3. The rationale for this is that due to the rapid absorption of paracetamol, any later doses will only lead to overestimation of the risk. However, if it has been more than 8 hours since the first dose, treat the patient as per the > 8 hours scenario in Box 3.
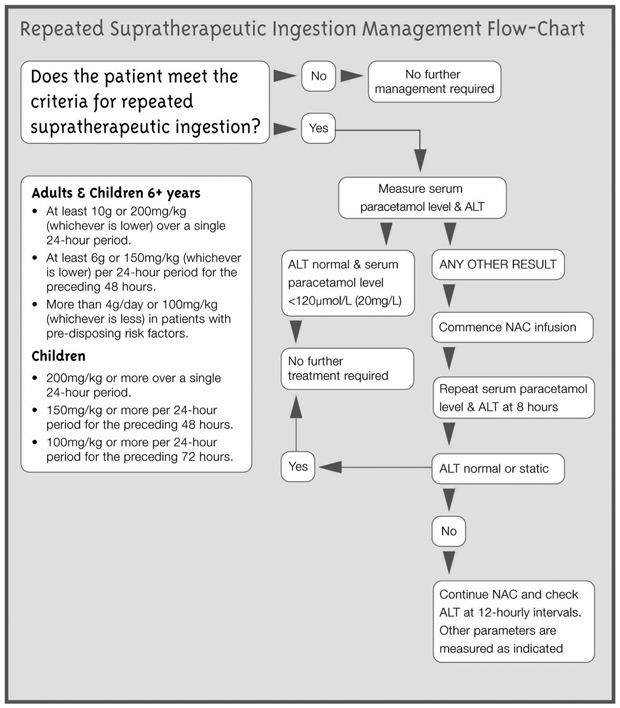
There is little evidence to guide risk assessment for repeated ingestion of high doses of paracetamol. The margin of safety has for many years been assumed to be high;16 however, recent data suggest that minor subclinical elevations of serum aminotransferase levels may be quite common with prolonged therapy.17 Conversely, studies of “high-risk” patients who have taken supratherapeutic doses over 3–4 days have suggested significant hepatotoxicity is uncommon.18 Therefore, the threshold for the reported dose that causes toxicity has been made deliberately and conservatively low (see Box 1 and Box 4). However, there is evidence that the combination of a low paracetamol level and normal alanine and aspartate aminotransferase levels at any time indicates there is no risk of subsequent hepatotoxicity.10 In most cases, this rule precludes the need for prolonged treatment in this group.
Anaphylactoid reactions manifested by rash, wheeze or mild hypotension occur in 10%–50% of patients during the first two N- acetylcysteine infusions.1,8 Management is supportive, with temporary halting or slowing of the infusion, and administration of antihistamines.19 The occurrence of an anaphylactoid reaction does not preclude the use of N-acetylcysteine on another occasion if indicated. Severe life-threatening reactions are very rare, but may occur in predisposed individuals, such as patients with asthma.
N-acetylcysteine reduces mortality if commenced late in patients with established paracetamol-induced fulminant hepatic failure, although mechanisms of action in this later period may be different. In this setting, N-acetylcysteine reduces inotrope requirements, decreases cerebral oedema and increases the rate of survival by about 30%.20
When risk assessment indicates that N-acetylcysteine is required, it is administered as a three-stage infusion (Box 6). Each stage contains different doses, totalling 300 mg/kg over 20–21 hours.7 If hepatic injury is suspected after the three infusion stages, N-acetylcysteine is continued at the rate of the last infusion stage (100 mg/kg each 16 hours or 150 mg/kg/24 hours) until there is clinical and biochemical evidence of improvement.
N-acetylcysteine is packaged for intravenous infusion in 10 mL ampoules, each containing 2000 mg (20%). Prescription of N-acetylcysteine requires a two-stage calculation to compute the appropriate weight-based dose and then the volume required. Calculation or transcription errors may lead to potentially fatal dosing errors.21 It is recommended that dosing tables providing the required volume of 20% N-acetylcysteine by weight categories be used to chart the volume required in each infusion. This precludes the need for calculations and decreases the potential for error. Such tables are found in the N-acetylcysteine product information and have also been reproduced in the new guidelines (Box 7).
Calculation of N-acetylcysteine doses is based on estimated lean bodyweight to the nearest 10 kg. A formula is provided to calculate N-acetylcysteine volume in each infusion for patients weighing more than 90 kg. For children, the dose of N-acetylcysteine is calculated in the same way, but with the volume reduced appropriately (Box 8).
Only a small proportion of patients who present late develop severe hepatotoxicity and fulminant hepatic failure.1,8 This complex subject was considered beyond the scope of the new guidelines. Clinicians should consult a specialist liver unit for advice on the management of patients with liver failure or signs that indicate a poor prognosis.22-24
1 Paracetamol dosing that may be associated with hepatic injury*
3 Management flow chart for acute paracetamol exposure with known time of ingestion
 | |||||||||||||||
|
ALT = alanine aminotransferase. NAC = N-acetylcysteine. OD = overdose. | |||||||||||||||
5 Recommended investigations according to time from paracetamol ingestion to N-acetylcysteine treatment
Time after paracetamol ingestion |
|||||||||||||||
7 Volume of N-acetylcysteine to be charted for each infusion, based on lean bodyweight*
Volume (mL) of N-acetylcysteine to be added to: |
|||||||||||||||
*Adapted from product information (Mayne Pharma Ltd, Melbourne, Vic). | |||||||||||||||
8 N-acetylcysteine administration applicable to children, based on bodyweight
- Frank F S Daly1,2,3
- John S Fountain4
- Lindsay Murray5
- Andis Graudins6,3
- Nicholas A Buckley3,7
- 1 Emergency Medicine, Royal Perth Hospital, Perth, WA.
- 2 University of Western Australia, Perth, WA.
- 3 NSW Poisons Information Centre, The Children’s Hospital at Westmead, Sydney, NSW.
- 4 National Poisons Centre, University of Otago, Dunedin, New Zealand.
- 5 Emergency Medicine, Sir Charles Gairdner Hospital, Perth, WA.
- 6 Emergency Medicine, Prince of Wales Hospital, Sydney, NSW.
- 7 Professorial Medical Unit, Prince of Wales Hospital Clinical School, University of New South Wales, Sydney, NSW.
We would like to thank Hazel Palmer of Scius Solutions for assistance in the preparation of this manuscript; her contribution was funded by an unrestricted educational grant from GlaxoSmithKline Consumer Healthcare Australia (GSK). GSK provided funding to enable the development of the new guidelines poster only; they had no role in the preparation, review or approval of this manuscript.
Frank Daly, John Fountain and Lindsay Murray each received a small honorarium from GSK for attending a half-day round-table meeting to discuss the content and scope of the poster (“Guidelines for the management of paracetamol overdose”) upon which this article is based.
- 1. Buckley N, Eddleston M. Paracetamol (acetaminophen) poisoning. Clin Evid 2005; (14): 1738-1744.
- 2. Dart RC, Erdman AR, Olson KR, et al. Acetaminophen poisoning: an evidence-based consensus guideline for out-of-hospital management. Clin Toxicol (Phila) 2006; 44: 1-18.
- 3. Linden CH, Rumack BH. Acetaminophen overdose. Emerg Med Clin North Am 1984; 2: 103-119.
- 4. Ostapowicz G, Fontana RJ, Schiødt FV, et al. Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Ann Intern Med 2002; 137: 947-954.
- 5. Flanagan RJ, Mant TG. Coma and metabolic acidosis early in severe acute paracetamol poisoning. Hum Toxicol 1986; 5: 179-182.
- 6. Reid D, Hazell W. Paracetamol poisoning: which nomogram should we use? Emerg Med (Fremantle) 2003; 15: 486-496.
- 7. Prescott LF, Illingworth RN, Critchley JA, et al. Intravenous N-acetylcystine: the treatment of choice for paracetamol poisoning. Br Med J 1979; 2: 1097-1100.
- 8. Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose. Analysis of the national multicenter study (1976 to 1985). N Engl J Med 1988; 319: 1557-1562.
- 9. Buckley NA, Whyte IM, O’Connell DL, Dawson AH. Oral or intravenous N-acetylcysteine: which is the treatment of choice for acetaminophen (paracetamol) poisoning? J Toxicol Clin Toxicol 1999; 37: 759-767.
- 10. Bridger S, Henderson K, Glucksman E, et al. Deaths from low dose paracetamol poisoning. BMJ 1998; 316: 1724-1725.
- 11. Dargan PI, Jones AL. Should a lower treatment line be used when treating paracetamol poisoning in patients with chronic alcoholism?: a case against. Drug Saf 2002; 25: 625-632.
- 12. Buckley NA, Srinivasan J. Should a lower treatment line be used when treating paracetamol poisoning in patients with chronic alcoholism?: a case for. Drug Saf 2002; 25: 619-624.
- 13. Rossi S, editor. Australian medicines handbook. Adelaide: Australian Medicines Handbook, 2006.
- 14. Buckley NA, Whyte IM, O’Connell DL, Dawson AH. Activated charcoal reduces the need for N-acetylcysteine treatment after acetaminophen (paracetamol) overdose. J Toxicol Clin Toxicol 1999; 37: 753-757.
- 15. Tan C, Graudins A. Comparative pharmacokinetics of Panadol Extend and immediate-release paracetamol in a simulated overdose model. Emerg Med Australas 2006; 18: 398-403.
- 16. Prescott LF. Therapeutic misadventure with paracetamol: fact or fiction? Am J Ther 2000; 7: 99-114.
- 17. Watkins PB, Kaplowitz N, Slattery JT, et al. Aminotransferase elevations in healthy adults receiving 4 grams of acetaminophen daily: a randomized controlled trial. JAMA 2006; 296: 87-93.
- 18. Daly FF, O’Malley GF, Heard K, et al. Prospective evaluation of repeated supratherapeutic acetaminophen (paracetamol) ingestion. Ann Emerg Med 2004; 44: 393-398.
- 19. Prescott LF, Park J, Ballantyne A, et al. Treatment of paracetamol (acetaminophen) poisoning with N-acetylcysteine. Lancet 1977; 2: 432-434.
- 20. Keays R, Harrison PM, Wendon JA, et al. Intravenous acetylcysteine in paracetamol induced fulminant hepatic failure: a prospective controlled trial. BMJ 1991; 303: 1026-1029.
- 21. Little M, Murray L, McCoubrie D, Daly FFS. A potentially fatal prescribing error in the treatment of paracetamol poisoning. Med J Aust 2005; 183: 535-536. <MJA full text>
- 22. Bernal W, Wendon J. More on serum phosphate and prognosis of acute liver failure. Hepatology 2003; 38: 533-534.
- 23. Bernal W, Donaldson N, Wyncoll D, Wendon J. Blood lactate as an early predictor of outcome in paracetamol-induced acute liver failure: a cohort study. Lancet 2002; 359: 558-563.
- 24. Bernal W, Wendon J. Acute liver failure; clinical features and management. Eur J Gastroenterol Hepatol 1999; 11: 977-984.





Abstract
Paracetamol is involved in a large proportion of accidental paediatric exposures and deliberate self-poisoning cases, although subsequent hepatic failure and death are both uncommon outcomes.
The optimal management of most patients with paracetamol overdose is usually straightforward. However, several differing nomograms and varying recommendations regarding potential risk factors for hepatic injury introduce complexity.
In order to reconcile management advice with current Australasian clinical toxicology practice, revised guidelines have been developed by a panel of clinical toxicologists consulting to the poisons information centres in Australia and New Zealand using a workshop and consultative process.
This article summarises the rationale for the recommendations made in these new guidelines.