A 61-year-old woman diagnosed with multifocal, poorly differentiated epithelial mesothelioma in September 2002 went into sustained spontaneous remission within months. She was completely disease-free within 6 months, and remained so 5 years later. This case demonstrates that this tumour may, very rarely, regress spontaneously, with no recurrence for many years. A greater knowledge of the underlying immune mechanisms would aid future management of this and other tumours.
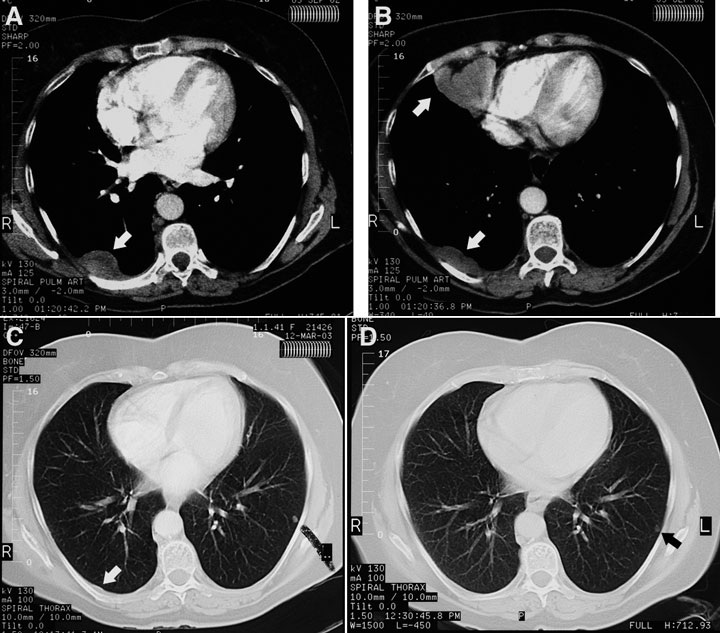
A chest radiograph taken before referral showed a pleural density measuring 10 cm × 2 cm overlying the posterior aspect of the right lower lobe, with no pleural plaques or other stigmata of asbestos exposure. A CT pulmonary angiogram performed the same day to exclude pulmonary embolism showed three pleural masses in the right side of the chest — the first corresponding to the opacity visible on the chest radiograph in the right costovertebral gutter at the level of the tracheal bifurcation, the second having a diameter of 6 cm and located in the right cardiophrenic angle (not of fatty attenuation), and the third in the right posteromedial costophrenic recess, just above the diaphragm (Box 1A and Box 1B). The lungs, mediastinum and upper abdomen (including the pancreas and para-aortic nodes) were normal.
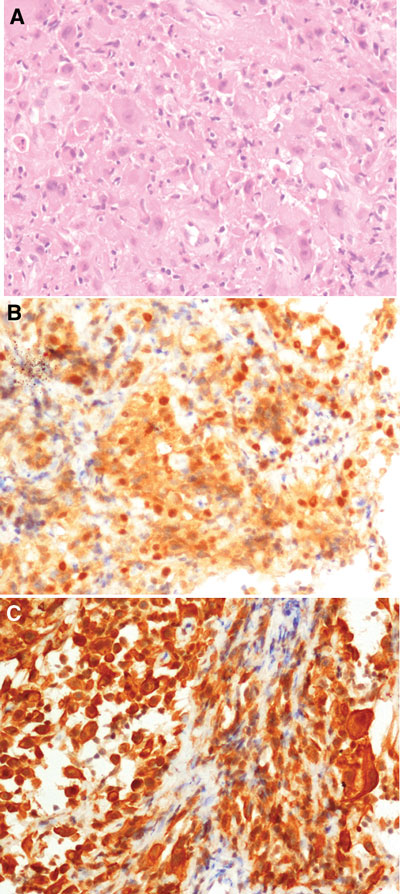
There is a proliferation of poorly cohesive large cells many of which had vesicular nuclei, prominent nucleoli and abundant eosinophilic cytoplasm. Occasional binucleate and multinucleate forms are present and there is a small amount of associated collagenous stroma with a mild chronic inflammatory cell infiltrate [Box 2A]. There is no evidence of mucin production, and immunoperoxidase stains for a variety of keratins are strongly positive, along with positive staining for calretinin [Box 2B] and cytokeratin 5/6, both markers of mesothelial differentiation [Box 2C]. Stains for LCA and S100 protein are negative.
The patient opted for no treatment, as she felt well. By 30 December 2002, a repeat CT scan of the chest showed a decrease in the size of the large, right-sided pleural mass in the costovertebral gutter from 17 mm × 9 mm (CT chest scan, 29 November 2002) to 12 mm × 6 mm. The second pleural mass was now so small it was difficult to see. The third mass was not visible, and there were two small intrapulmonary nodules — one in the right middle lobe and the other in the left lower lobe. By March 2003, the first and second pleural masses were even smaller (Box 1C) and, by June 2003, they had disappeared (Box 1D). A CT chest scan in June 2004 was normal except for the two tiny intrapulmonary nodules that had not changed in size and were probably granulomas. The patient was last reviewed in June 2007 and was in good health, with no evidence of tumour, and was scheduled for next review in 6 months.
There has been one case report of a patient with malignant mesothelioma of the pleura that regressed spontaneously, but after 6 years there was a single recurrence, which was resected surgically, and the patient was followed up for a total of 12 years.1 This raises the question in such cases of the duration of follow-up. In the latter case, a prominent host response to tumour was seen in both the primary tumour and the recurrence. In another case, a patient had a spontaneous remission of a malignant peritoneal mesothelioma, and had high spiking fevers when the tumour recurred.2
A report from Western Australia described a woman whose tumour regressed spontaneously but who eventually died 20 months later.3 It was noted that the tumour tissue was infiltrated with mononuclear cells, and as the tumour recurred some malignant mesothelioma antigens disappeared.
Several aspects of our case should be noted.
Histopathology
The histopathological findings for our patient’s tumour were re-examined by another pathologist.
Not all three lesions were biopsied, as it was felt highly probable that the pathological findings for all three would be identical.
Author’s experience
I have considerable experience in the management of mesothelioma and benign asbestos-related conditions and currently see about 600 patients with this condition a year. I also act as an expert witness for the courts.
Mesothelioma is a relatively common condition in Australia at present and, as a result, our thoracic physicians, oncologists and pathologists have considerable experience in this area.
Course of the disease
The patient had markedly elevated inflammatory markers and is likely to have had the tumour for several months before diagnosis. It is suspected that her natural killer cells and cell-mediated immunity accounted for the regression of the tumour.
Evidence of the beginning of spontaneous regression was unusually rapid, occurring within months of diagnosis.
Exposure
Our patient’s exposure to asbestos seems to have been relatively mild and incidental, as often occurs with women who develop mesothelioma.
Self-treatment
The patient did not use any unusual therapies, such as alternative medicines, diets and faith healing, after the diagnosis.
2 Histological examination of a core biopsy specimen from one of the pleural masses (Box 1)
 |
|
A: Histological section of the core biopsy of the pleural mass in Box 1A showing sheet-like proliferation of pleomorphic epithelioid cells with abundant eosinophilic cytoplasm, representing a poorly differentiated epithelioid malignant mesothelioma. B: Core biopsy showing a positive result on immunoperoxidase staining for calretinin. C: Core biopsy showing a positive result on immunoperoxidase staining for cytokeratin 5/6. |
- 1. Pilling JE, Nicholson AG, Harmer C, Goldstraw P. Prolonged survival due to spontaneous regression and surgical excision of malignant mesothelioma. Ann Thorac Surg 2007; 83: 314-315.
- 2. Schwartz E, Maayan C, Mouallem M, et al. Malignant peritoneal mesothelioma: long-term spontaneous clinical remission. Med Pediatr Oncol 1991; 19: 325-328.
- 3. Robinson BW, Robinson C, Lake RA. Localised spontaneous remission in mesothelioma — possible immunological mechanism. Lung Cancer 2001; 32: 197-201.





I would like to thank Dr Rick Abraham, Medical Oncologist and Senior Lecturer in Medicine, University of Queensland, for his assistance in the management of this patient; Dr Ian LeFevre, Histopathologist, QML Pathology, Brisbane, and Dr Lyndie Clark, Histopathologist, Prince Charles Hospital, Brisbane, for reviewing the histological sections; and Dr Ian LeFevre and QML Pathology for supplying the photomicrographs.
None identified.